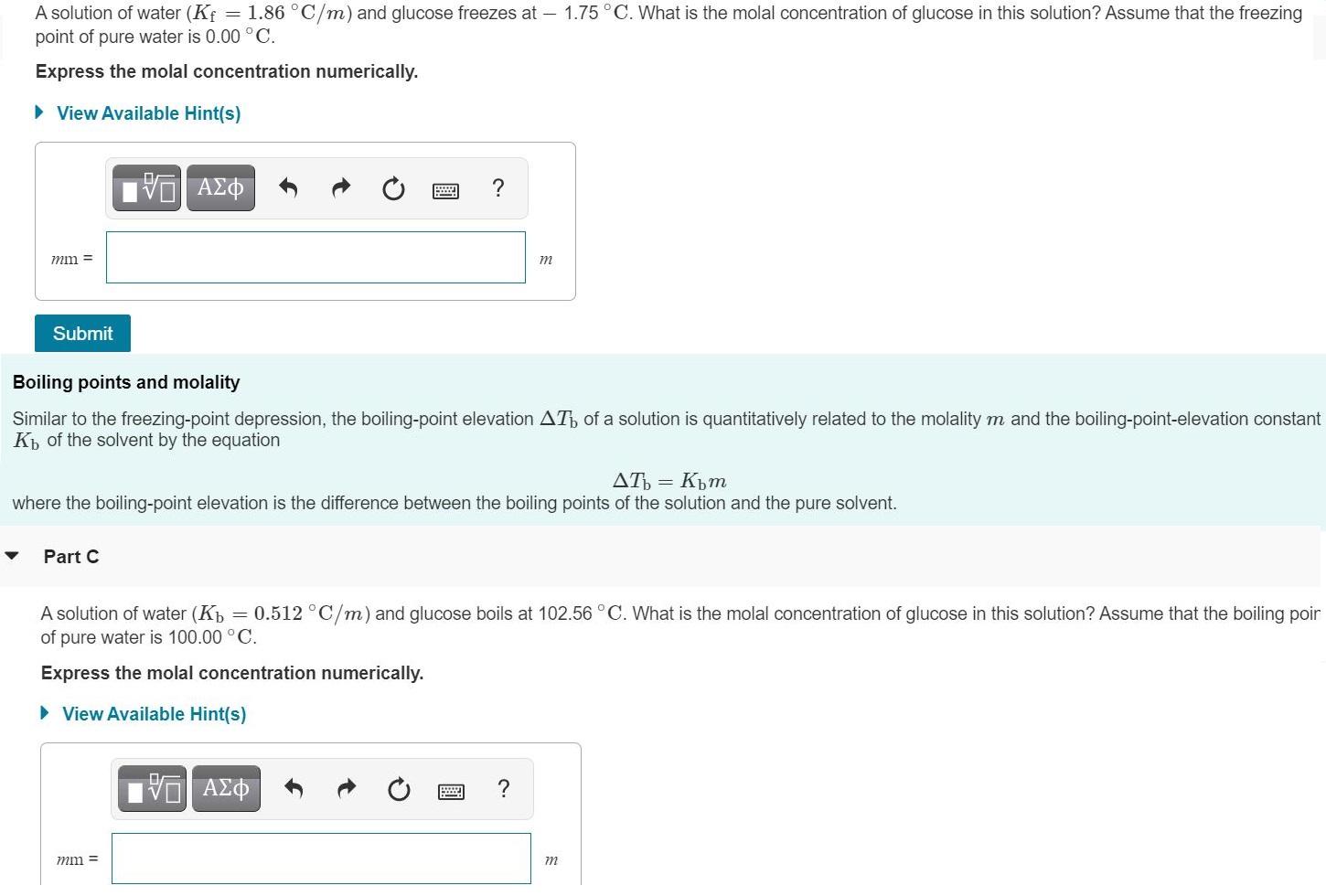
A solution of water (Kf point of pure water is 0.00 C. = 1.86 C/m) and...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A solution of water (Kf point of pure water is 0.00 °C. = 1.86 °C/m) and glucose freezes at – 1.75°C. What is the molal concentration of glucose in this solution? Assume that the freezing Express the molal concentration numerically. • View Available Hint(s) mm = m Submit Boiling points and molality Similar to the freezing-point depression, the boiling-point elevation AT of a solution is quantitatively related to the molality m and the boiling-point-elevation constant K of the solvent by the equation AT = Kpm where the boiling-point elevation is the difference between the boiling points of the solution and the pure solvent. Part C A solution of water (Kp = 0.512 °C/m) and glucose boils at 102.56°C. What is the molal concentration of glucose in this solution? Assume that the boiling poir of pure water is 100.00 °C. Express the molal concentration numerically. • View Available Hint(s) mm = m A solution of water (Kf point of pure water is 0.00 °C. = 1.86 °C/m) and glucose freezes at – 1.75°C. What is the molal concentration of glucose in this solution? Assume that the freezing Express the molal concentration numerically. • View Available Hint(s) mm = m Submit Boiling points and molality Similar to the freezing-point depression, the boiling-point elevation AT of a solution is quantitatively related to the molality m and the boiling-point-elevation constant K of the solvent by the equation AT = Kpm where the boiling-point elevation is the difference between the boiling points of the solution and the pure solvent. Part C A solution of water (Kp = 0.512 °C/m) and glucose boils at 102.56°C. What is the molal concentration of glucose in this solution? Assume that the boiling poir of pure water is 100.00 °C. Express the molal concentration numerically. • View Available Hint(s) mm = m
Expert Answer:

Related Book For 

Practical Management Science
ISBN: 978-1305250901
5th edition
Authors: Wayne L. Winston, Christian Albright
Posted Date:
Students also viewed these chemistry questions
-
Refer to Decision Maker, Purchase Manager, in this chapter. Assume that you are the motorcycle manufacturers managerial accountant. The purchasing manager asks you about preparing an estimate of the...
-
Refer to Decision Maker, Purchase Manager, in this chapter. Assume that you are the motorcycle manufacturers managerial accountant. The purchasing manager asks you about preparing an estimate of the...
-
For the problems in this exercise, assume that there are no pipeline stalls and that the breakdown of executed instructions is as follows: 1. In what fraction of all cycles is the data memory used?...
-
Preparing and interpreting a statement of cash flows using a T-account work sheet. Financial statement data for Dickerson Manufacturing Company for the current year appear in Exhibit 5.29. Additional...
-
A company has been asked by an important customer to demonstrate that its process capability ratio Cp exceeds 1.33. It has taken a sample of 50 parts and obtained the point estimate Cp = 152. Assume...
-
How is the value of a financial option affected by; (a) The current price of the underlying asset. (b) The exercise (or strike) price. (c) The risk-free rate. (d) The time until expiration (or...
-
\(\frac{44}{75} \div \frac{484}{285}\) Perform the indicated operations. Reduce to lowest terms.
-
Dilithium Batteries is a division of Enterprise Corporation. The division manufactures and sells a long-life battery used in a wide variety of applications. During the coming year, it expects to sell...
-
High-Low Method to Determine Fixed Cost and Variable Rate McGarvey Manufacturing Company had the following 12 months of data on purchasing cost and number of purchase orders. Number of Purchase Month...
-
Bradburn plc was formed 5 years ago through a public subscription of ordinary shares. Daniel Brown, who owns 15% of the ordinary shares, was one of the organizers of Bradburn and is its current...
-
Marigold's Sporting Stores Inc. reported the following cost and net realizable value information for inventory at December 31: Item Units Unit Cost Unit NRV Skates: Bauer 13 $247 $388 CCM 10 $400...
-
For this assignment, you will be demonstrating that you know (1) how to read the rhetorical situation of applying for a position, and (2) how to write to that situation by presenting yourself...
-
City of Johannesburg mayor Geoff Makhubo says the city will offer significant debt write-offs to qualifying ratepayers through its debt rehabilitation programme. In his state of the city address on...
-
Batibot, a resident citizen earned the ff incomes: Questions: 1. Income derived from sources WITHIN the Philippines 2. Income derived from sources OUTSIDE the Philippines 3. The TOTAL CAPITAL GAINS...
-
Factor the polynomial. 3 x +5x +5x+25
-
What are the isomorphic graphs? Determine whether following graphs G & H are isomorphic. a f b W x d h G g N y H
-
b - In Chapter 4, you modified the EventDemo class to demonstrate two Event objects. Now, modify that class again as follows: Instantiate three Event objects, and prompt the user for values for each...
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
A beer company has divided Bloomington into two territories. If the company spends x 1 dollars on promotion in territory 1, it can sell 60x 1 1/2 cases of beer there; and if it spends x 2 dollars on...
-
Use Solver Table to run a sensitivity analysis on the cost per assembling labor hour, letting it vary from $5 to $20 in increments of $1. Keep track of the computers produced in row 21, the hours...
-
Do the sales prices of houses in a given community vary systematically with their sizes (as measured in square feet)? Answer this question by estimating a simple regression equation where the sales...
-
1.12 Why Go. Why do firms become multinational?
-
1.13 Investment Motives of Firms. What is the differ- ence between proactive and defensive investment motives?
-
1.14 Aidan's Phases. What are the main phases that Aidan passed through as it evolved into a truly global firm? What are the advantages and disadvan- tages of each?

Study smarter with the SolutionInn App


