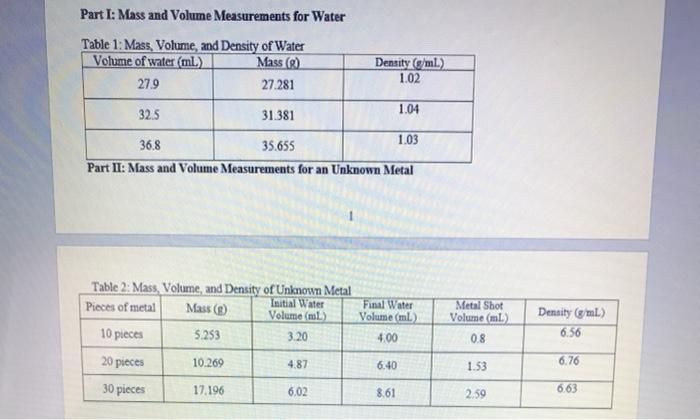
Part I: Mass and Volume Measurements for Water Table 1: Mass, Volume, and Density of Water...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
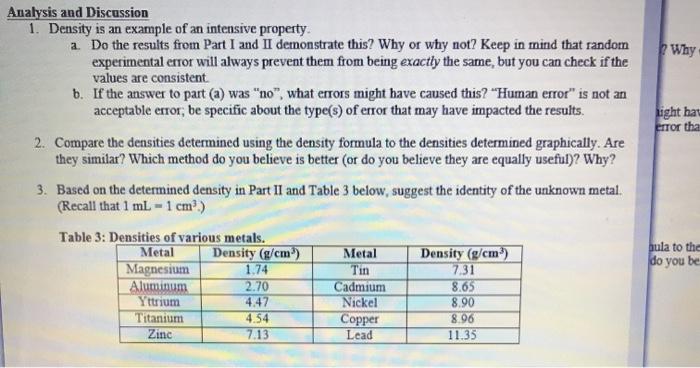
Part I: Mass and Volume Measurements for Water Table 1: Mass, Volume, and Density of Water Volume of water (ml) Mass (g) 27.9 27.281 32.5 31.381 36.8 35.655 Part II: Mass and Volume Measurements for an Unknown Metal Table 2: Mass, Volume, and Density of Unknown Metal Initial Water Pieces of metal Mass (g) Volume (ml) 10 pieces 5.253 3.20 20 pieces 30 pieces 10.269 17.196 4.87 6.02 Density (g/ml.) 1.02 6.40 1.04 Final Water Volume (ml) 4.00 8.61 1.03 Metal Shot Volume (ml) 0.8 1.53 2.59 Density (g/mL) 6.56 6.76 6.63 Calculations: Part I: Density of Water Average Density of Water: Figure 1: Mass vs. Volume of Water Mass (g) Volume (ml.) Best Fit Line: Y-0.9403x+0.9725 R-Value: 0.999 Density of water: Average Density of Metal: Figure 2: Mass vs. Volume of Unknown Metal Mass (g) Volume (ml) Best Fit Line: Linear R² Value Density of unknown metal: Analysis and Discussion 1. Density is an example of an intensive property. a. Do the results from Part I and II demonstrate this? Why or why not? Keep in mind that random experimental error will always prevent them from being exactly the same, but you can check if the values are consistent. b. If the answer to part (a) was "no", what errors might have caused this? "Human error" is not an acceptable error; be specific about the type(s) of error that may have impacted the results. 2. Compare the densities determined using the density formula to the densities determined graphically. Are they similar? Which method do you believe is better (or do you believe they are equally useful)? Why? 3. Based on the determined density in Part II and Table 3 below, suggest the identity of the unknown metal. (Recall that 1 mL-1 cm³.) Table 3: Densities of various metals. Metal Magnesium Aluminum Yttrium Titanium Zinc Density (g/cm³) 1.74 2.70 4.47 4.54 7.13 Metal Tin Cadmium Nickel Copper Lead Density (g/cm³) 7.31 8.65 8.90 8.96 11.35 Why ight hav error tha bula to the do you be 4. Assuming the metal was correctly identified, calculate the percent error in the determined density. Use the density determined graphically for this calculation. Percent Error: Theoretical Value - Experimental Value Theoretical Value -x 100% 5. Water has a known density of 0.997 g/mL at room temperature. Calculate the percent error in the determined density. Use the density determined graphically for this calculation. Part I: Mass and Volume Measurements for Water Table 1: Mass, Volume, and Density of Water Volume of water (ml) Mass (g) 27.9 27.281 32.5 31.381 36.8 35.655 Part II: Mass and Volume Measurements for an Unknown Metal Table 2: Mass, Volume, and Density of Unknown Metal Initial Water Pieces of metal Mass (g) Volume (ml) 10 pieces 5.253 3.20 20 pieces 30 pieces 10.269 17.196 4.87 6.02 Density (g/ml.) 1.02 6.40 1.04 Final Water Volume (ml) 4.00 8.61 1.03 Metal Shot Volume (ml) 0.8 1.53 2.59 Density (g/mL) 6.56 6.76 6.63 Calculations: Part I: Density of Water Average Density of Water: Figure 1: Mass vs. Volume of Water Mass (g) Volume (ml.) Best Fit Line: Y-0.9403x+0.9725 R-Value: 0.999 Density of water: Average Density of Metal: Figure 2: Mass vs. Volume of Unknown Metal Mass (g) Volume (ml) Best Fit Line: Linear R² Value Density of unknown metal: Analysis and Discussion 1. Density is an example of an intensive property. a. Do the results from Part I and II demonstrate this? Why or why not? Keep in mind that random experimental error will always prevent them from being exactly the same, but you can check if the values are consistent. b. If the answer to part (a) was "no", what errors might have caused this? "Human error" is not an acceptable error; be specific about the type(s) of error that may have impacted the results. 2. Compare the densities determined using the density formula to the densities determined graphically. Are they similar? Which method do you believe is better (or do you believe they are equally useful)? Why? 3. Based on the determined density in Part II and Table 3 below, suggest the identity of the unknown metal. (Recall that 1 mL-1 cm³.) Table 3: Densities of various metals. Metal Magnesium Aluminum Yttrium Titanium Zinc Density (g/cm³) 1.74 2.70 4.47 4.54 7.13 Metal Tin Cadmium Nickel Copper Lead Density (g/cm³) 7.31 8.65 8.90 8.96 11.35 Why ight hav error tha bula to the do you be 4. Assuming the metal was correctly identified, calculate the percent error in the determined density. Use the density determined graphically for this calculation. Percent Error: Theoretical Value - Experimental Value Theoretical Value -x 100% 5. Water has a known density of 0.997 g/mL at room temperature. Calculate the percent error in the determined density. Use the density determined graphically for this calculation.
Expert Answer:
Answer rating: 100% (QA)
Density Mass Volume Mass Density x Volume This equation is in the form of y mx Therefore if we plot ... View the full answer

Related Book For 

Modeling the Dynamics of Life Calculus and Probability for Life Scientists
ISBN: 978-0840064189
3rd edition
Authors: Frederick R. Adler
Posted Date:
Students also viewed these physics questions
-
Explain how to apply the rules of Section (61)a to determine taxable items.
-
Refer to Table 10.1 in the text and look at the period from 1973 through 1978. a. Calculate the arithmetic average returns for common stocks and T-bills over this period. b. Calculate the standard...
-
Prove Theorem 9.7.1. Theorem 9.7.1 Principal Axes Theorem for ft3 Let ax2 + by2 + cz2 + 2dxy + 2exz + 2fyz + gx + hy + iz + j = 0 be the equation of a quadric Q, and let xTAx = ax2 + by2 + cz2 + 2dxy...
-
On June 12, 2015, Eastern Power and Electric Ltd.'s common shares had a market price of $48 per share. For the previous year, Eastern Power and Electric paid an annual dividend of $2.88. Compute the...
-
The trial balance of Biju Medical Supplies at the end of its fiscal year, August 31, 2020, includes these accounts (amounts in thousands): Inventory Rp17,200; Purchases Rp149,000; Sales Revenue...
-
XCBob, Inc. is an online motorcycle and ATV parts business from the owners home. XCBob entered into oral agreements with the defendant, Ed Tucker Distributers, for purchases of various materials to...
-
Information from the 2010 Form 990 and the 2010 annual report for the AmeriCares Foundation follows. Use the following information to complete the exercise. Required a. Compute the following...
-
New parents bring their 3-month-old to the clinic for a rash on the infants head. On examination, the skin affected by the rash is thickened, yellowish white in color, scaly, and looks waxy. In...
-
In alphabetical order below are balance sheet items for Mendoza Company at December 31, 2017. Kathy Mendoza is the owner of Mendoza Company. Prepare a balance sheet, following the format of...
-
A 1:5 step-up transformer has 120V across the primary and 600 ohms resistance across the secondary. Assuming 100% efficiency, the primary current equals (A) 0.2 Amp. (C) 10 Amps. (B) 5 Amps. (D) 20...
-
Your understanding of systems thinking. How the book Credibility address systems thinking? Does the book align with or challenge your understanding of systems thinking? Consider all that you are...
-
How (if at all) have your views on your own culture(s) changed since the beginning of this course? How have they changed in regards to cultures that differ from your own?2. After exploring areas of...
-
Identifying a Number to Multiply by When Using the Linear Combination Method atch each system to the number the first equation can be multiplied by to eliminate the terms when adding to the second...
-
Identify a company that is pursuing a growth strategy and explain how how they are doing well at it ? Energizing the Business Going Global GROWTH STRATEGIES Leveraging the Business Creating a New...
-
Imagine yourself as the executive manager of an organization. You are responsible for establishing the three levels of management and how each level functions in the organization. One of the...
-
Cars travelling to the George Washington Bridge from New Jersey to New York City must pay a toll. About 38 percent of the commuters use E-ZPass, which registers the toll E-ZPass customers go quickly...
-
Cobb Manufacturing Company uses a process cost system and average costing. The following production data is for the month of June 2011. Production Costs Work in process, beginning of the month:...
-
The Heaviside function (Section 2.3, Exercise 25), defined by The following functions all fail to be differentiable at x = 0. In each case, graph the function, see what happens if you try to compute...
-
The probability of a value greater than 11.0 drawn from a normal distribution with mean 13.0 and standard deviation 1.2. Use the cumulative distribution function for the standard normal, (z), to find...
-
The case in Exercise 1. For the following joint distributions, find the covariance of X and Y using the computational method, Cov(X, Y) = E(XY) - . X=0 0.14 0.21 X=1 0.26 0.39 Y=0 1
-
Powerhouse Ltd purchased machinery on 2 January 2019, at a cost of $800 000. The machinery is depreciated using the straightline method over a useful life of 8 years with a residual value of $80 000....
-
The purchases and sales of Big Flower Pty Ltd of one brand of lawn fertiliser for the year ended 31 December 2019 are contained in the schedule below. The selling price up to 30 June was $12 per unit...
-
In groups of four or five, consider the following information. On 1 July 2019, Stevenson Pty Ltd, a proprietary company with three shareholders, acquired some property by issuing 100 000 shares to...

Study smarter with the SolutionInn App


