Percent Error At 25 C, the density of water is 0.997044 g/mL. Use this value to...
Fantastic news! We've Found the answer you've been seeking!
Question:
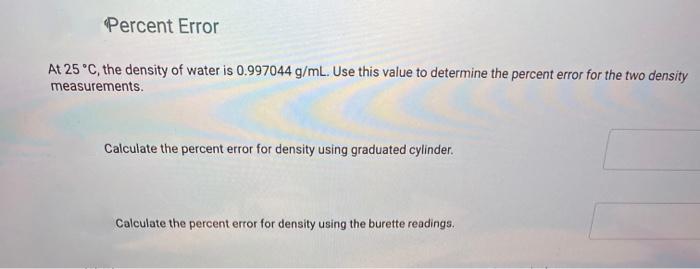
Transcribed Image Text:
Percent Error At 25 °C, the density of water is 0.997044 g/mL. Use this value to determine the percent error for the two density measurements. Calculate the percent error for density using graduated cylinder. Calculate the percent error for density using the burette readings. Percent Error At 25 °C, the density of water is 0.997044 g/mL. Use this value to determine the percent error for the two density measurements. Calculate the percent error for density using graduated cylinder. Calculate the percent error for density using the burette readings.
Expert Answer:
Answer rating: 100% (QA)
Density of water from graduated cylinder 0994 gmol D... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The density of water for 0C < T < 100C is given in Table A-1. Fit this data to a least-squares parabola, = a + bT + cT2, and test its accuracy vis-a-vis Table A-1.Finally, compute at T = 45C and...
-
The density of water is 1.00 g/mL at 48C. How many water molecules are present in 2.56 mL of water at this temperature?
-
The density of water at 4oC is 1.00 X 103 kg/m3. What is waters density at 94oC?
-
Assume your company shows the market values of equity and debt at the level of $175373 and $224626, respectively. The rate of return on assets is 33 percent and its volatility is 45 percent. The...
-
Altman and Bland report the survival times for patients with active hepatitis, half treated with prednisone and half receiving no treatment. The survival times (in months) (Exercise 1.25 and EX0125)...
-
Studies indicate that the income elasticity of demand for servants in the United States exceeds 1. Nevertheless, the number of servants has been decreasing during the last 75 years, while incomes...
-
A stock price is governed by geometric Brownian motion with \(\mu=.20\) and \(\sigma=.40\). The initial price is \(S(0)=1\). Evaluate the four quantities E[In S(1)], E[S(1)], stdev[In S(1)]...
-
A bond trader purchased each of the following bonds at a yield to maturity of 8%. Immediately after she purchased the bonds, interest rates fell to 7%. What is the percentage change in the price of...
-
Duddy Kravitz owns the Saint Viateur Bagel store. His world famous bagels are hand rolled, boiled and then baked in a wood - burning oven. Uncle Benjy thinks that the wood oven should be replaced by...
-
In order to prepare the statement of cash flows for Tinker Toys Corporation for 2001, The accountant has compiled the following data regarding cash flows. ANSWER THE FOLLOWING QUESTIONS. Cash paid to...
-
The following were taken from the books of Satellite Company. January 1 P 335,000 March 31 P 208,750 Raw materials Work in process Finished goods Direct materials used Direct labor Factory overhead...
-
Why are we attracted to certain people, and what progression do social relationships follow?
-
Can love be studied scientifically? Is there an elusive quality to love that makes it at least partially unknowable? How would you define falling in love? How would you study it?
-
Par Ltd. purchased 100% of the voting shares of Sub Ltd. for $1,400,000 on October 1, 2006. The balance sheet of Sub Ltd. at that date was: Required: Prepare the eliminating entry (or entries)...
-
How does the reality of late adulthood differ from the stereotypes about that period?
-
__________therapies assume that people should take responsibility for their lives and the decisions they make.
-
The stress state at point P 0x C B toy 02 A = 100 MPa y = -40 MPa Oz 80 MPa Txy = Tyz = Tzx = 0 MPa Determine: (i) maximum values of share stresses and associated normal stresses. (ii) octahedral...
-
Find an equation of the given line. Slope is -2; x-intercept is -2
-
A chemist in the nineteenth century prepared an unknown substance. In general, do you think it would be more difficult to prove that it is an element or a compound? Explain.
-
When heated, lithium reacts with nitrogen to form lithium nitride: What is the theoretical yield of Li3N in grams when 12.3 g of Li are heated with 33.6 g of N2? If the actual yield of Li3N is 5.89...
-
Since 1998, elements 112, 114, and 116 have been synthesized. Element 112 was created by bombarding 208Pb with 66Zn; element 114 was created by bombarding 244Pu with 48Ca; element 116 was created by...
-
Evaluate the shape factor \(\phi_{B}^{f}\) for strength-limited design in bending of a square box section of outer edge-length \(h=100 \mathrm{~mm}\) and wall thickness \(t=3 \mathrm{~mm}\). Is this...
-
Derive the expression for the shape-efficiency factor \(\phi_{B}^{e}\) for stiffness-limited design for a circular tube with outer radius \(5 t\) and wall thickness \(t\), loaded in bending (Fig....
-
Derive the expression for the shape-efficiency factor \(\phi_{B}^{e}\) for stiffness-limited design for a square box section of wall thickness \(t\), and height and width \(h_{1}=10 t\) bent about...

Study smarter with the SolutionInn App


