Please fill in the following table. Thanks for all the help! Give the pKa and pKb of
Question:
Please fill in the following table. Thanks for all the help!
Give the pKa and pKb of weak acids and bases. For each solution, calculate the pH of a 0.1000 M solution, and calculate the volume of 0.1000 M NaOH or HCl that would be required to titrate a 10.00 mL sample to the equivalence point. To fill-in the table below, answer the subsequent questions.
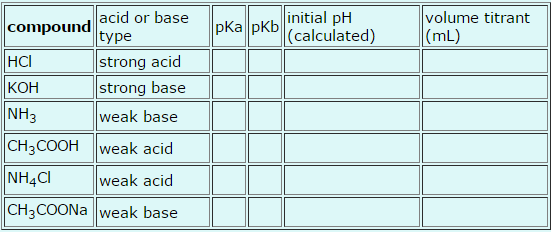
pKb(NH3) =
pKa(CH3COOH) =
pKa(NH4Cl) =
pKb(CH3COONa) =
pH(0.1000 M HCl) =
pH(0.1000 M KOH) =
pH(0.1000 M NH3) =
pH(0.1000 M CH3COOH) =
ptspH(0.1000 M NH4Cl) =
pH(0.1000 M CH3COONa) =
Volume of 0.1000 M NaOH required to titrate 10.00 mL of 0.1000 M HCl =
Volume of 0.1000 M HCl required to titrate 10.00 mL of 0.1000 M KOH =
Volume of 0.1000 M HCl required to titrate 10.00 mL of 0.1000 M NH3 =
Volume of 0.1000 M NaOH required to titrate 10.00 mL of 0.1000 M CH3COOH =
Volume of 0.1000 M NaOH required to titrate 10.00 mL of 0.1000 M NH4Cl =
Volume of 0.1000 M HCl required to titrate 10.00 mL of 0.1000 M CH3COONa =

Foundations of Financial Management
ISBN: 978-1259194078
15th edition
Authors: Stanley Block, Geoffrey Hirt, Bartley Danielsen




