Question 4 Pure caffeine has a literature melting point of 238C. If you performed a series...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
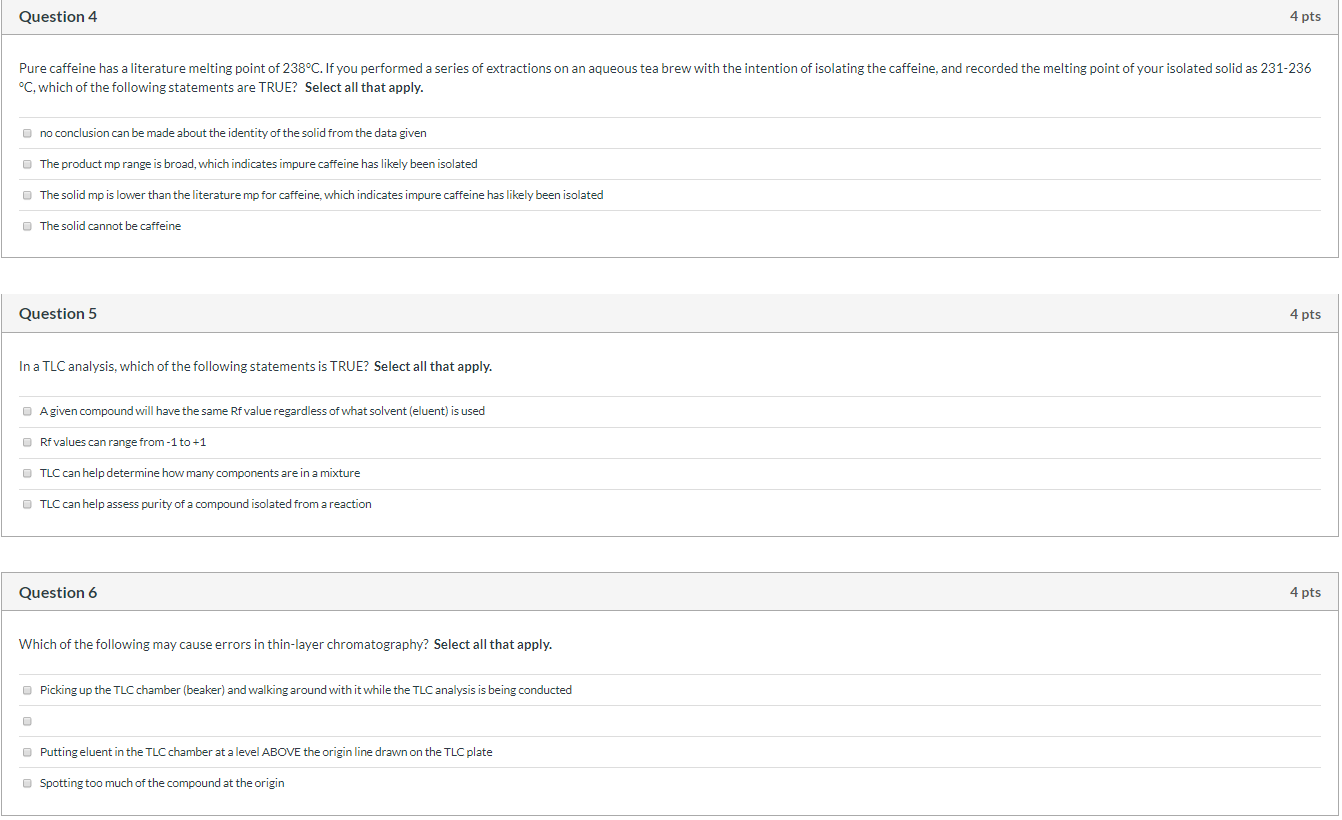
Question 4 Pure caffeine has a literature melting point of 238°C. If you performed a series of extractions on an aqueous tea brew with the intention of isolating the caffeine, and recorded the melting point of your isolated solid as 231-236 °C, which of the following statements are TRUE? Select all that apply. no conclusion can be made about the identity of the solid from the data given The product mp range is broad, which indicates impure caffeine has likely been isolated The solid mp is lower than the literature mp for caffeine, which indicates impure caffeine has likely been isolated The solid cannot be caffeine Question 5 In a TLC analysis, which of the following statements is TRUE? Select all that apply. A given compound will have the same Rf value regardless of what solvent (eluent) is used ⒸRf values can range from-1 to +1 TLC can help determine how many components are in a mixture TLC can help assess purity of a compound isolated from a reaction Question 6 Which of the following may cause errors in thin-layer chromatography? Select all that apply. Picking up the TLC chamber (beaker) and walking around with it while the TLC analysis is being conducted 4 pts Putting eluent in the TLC chamber at a level ABOVE the origin line drawn on the TLC plate Spotting too much of the compound at the origin 4 pts 4 pts Question 4 Pure caffeine has a literature melting point of 238°C. If you performed a series of extractions on an aqueous tea brew with the intention of isolating the caffeine, and recorded the melting point of your isolated solid as 231-236 °C, which of the following statements are TRUE? Select all that apply. no conclusion can be made about the identity of the solid from the data given The product mp range is broad, which indicates impure caffeine has likely been isolated The solid mp is lower than the literature mp for caffeine, which indicates impure caffeine has likely been isolated The solid cannot be caffeine Question 5 In a TLC analysis, which of the following statements is TRUE? Select all that apply. A given compound will have the same Rf value regardless of what solvent (eluent) is used ⒸRf values can range from-1 to +1 TLC can help determine how many components are in a mixture TLC can help assess purity of a compound isolated from a reaction Question 6 Which of the following may cause errors in thin-layer chromatography? Select all that apply. Picking up the TLC chamber (beaker) and walking around with it while the TLC analysis is being conducted 4 pts Putting eluent in the TLC chamber at a level ABOVE the origin line drawn on the TLC plate Spotting too much of the compound at the origin 4 pts 4 pts
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below 4 Answer no conclusion c... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemistry questions
-
1. conditions. a) reaction: H Calculate the molar volume of H, gas under experimental b) -0.421 g Zn x _Lmol Zn Lmol H, 0.00644 mol 65.39 g Zn 1 mol Zn Second, the molar volume under experimental...
-
Please help with this code all the requirements are in the attached image. Add the requirements into the code provided below. HTML: My Movie Library Title: Genre: Action Adventure Comedy Documentary...
-
1) The speed of light in a specific medium is 0.8 c where c is the speed of light in vacuum. The refractive index of this medium is: 2) A light ray is incident upon a plane interface between two...
-
Creativity and Innovation Management (Feb. 2008) published an article on identifying the social network characteristics of lead users of children's computer games. Data were collected for n = 326...
-
Journalize the following transactions in the accounts of Powerplay Co., which operates a riverboat casino: Mar. 1. Received a $45,000, 60-day, 6% note dated March 1 from Pynn Co. on account. 18....
-
Mandel Manufacturing, Inc. has a manufacturing machine that needs attention. The company is considering two options. Option 1 is to refurbish the current machine at a cost of $1,100,000. If...
-
3. When Walton Weiner interviewed for a job with McGraw-Hill, Inc., he was assured that the company would not terminate an employee without "just cause." Weiner also signed a contract specifying that...
-
Kwikeze Company set the following standard costs for one unit of its product. Direct materials (4.5 Ibs. @ $6 per Ib.) . . . . . . . $27.00 Direct labor (1.5 hrs. @ $12 per hr.) . . . . . . . . ....
-
Need answer with solution. Thank you. The Skateboard Company uses a process cost system for wheels. Materials are ad of the process and conversion costs are uniformly incurred. At the beginning of Se...
-
a) A venturimeter is in an installation of 100 mm diameter pipe carrying water. The maximum range available in water-mercury differential pressure gauge is 50 cm of mercury deflection. Find the...
-
Two students are discussing environmentalists cl aims that trade harms the environment. One student says, Sure, there may be pollution effects, but theyre a small price to pay for a higher standard...
-
T/F: Stable analysis patterns (SAPs) separate the core concepts of the domain from business-specific concepts.
-
Can you list different applications of the word anxiety?
-
T/F: Analysis patterns are conceptual models that can be used to model and share domain knowledge.
-
What could be IOs for AnyGain? List any two.
-
T/F: A traditional analysis pattern has a very limited reuse.
-
Hawkins Corporation began construction of a motel on March 31, 2024. The project was completed on April 30, 2025. No new loans were required to fund construction. Hawkins does have the following two...
-
Use nodal analysis to determine voltages v1, v2, and v3 in the circuit Fig. 3.76. Figure 3.76 4 S 3i, 2 A 4A
-
In June of 2012, Maureen's house is vandalized during a long-term power failure after a hurricane hit the city. The president of the United States declares Maureen's city a disaster area as a result...
-
Ann hires a nanny to watch her two children while she works at a local hospital. She pays the 19-year-old nanny $125 per week for 48 weeks during the current year. a. What is the employer's portion...
-
How much of each of the following prizes or awards is taxable? a. Cheline received a $50,000 gift bag at the Oscars in 2012. b. Jon received a gold watch worth $350 for 25 years of service to his...
-
The following transactions relate to topics in this chapter. a. Purchased inventory on account, terms \(2 / n, n / 30\), accounted for using the perpetual method. b. Collected sales taxes on a...
-
Atlas Inc. issues short-term interest-bearing notes and uses the proceeds to purchase inventory. The company's profits for the year remain unchanged. Required Indicate how the use of the notes would...
-
List and briefly explain the primary characteristics of long-term debt securities. What are the primary distinctions between a debt security and an equity security?

Study smarter with the SolutionInn App


