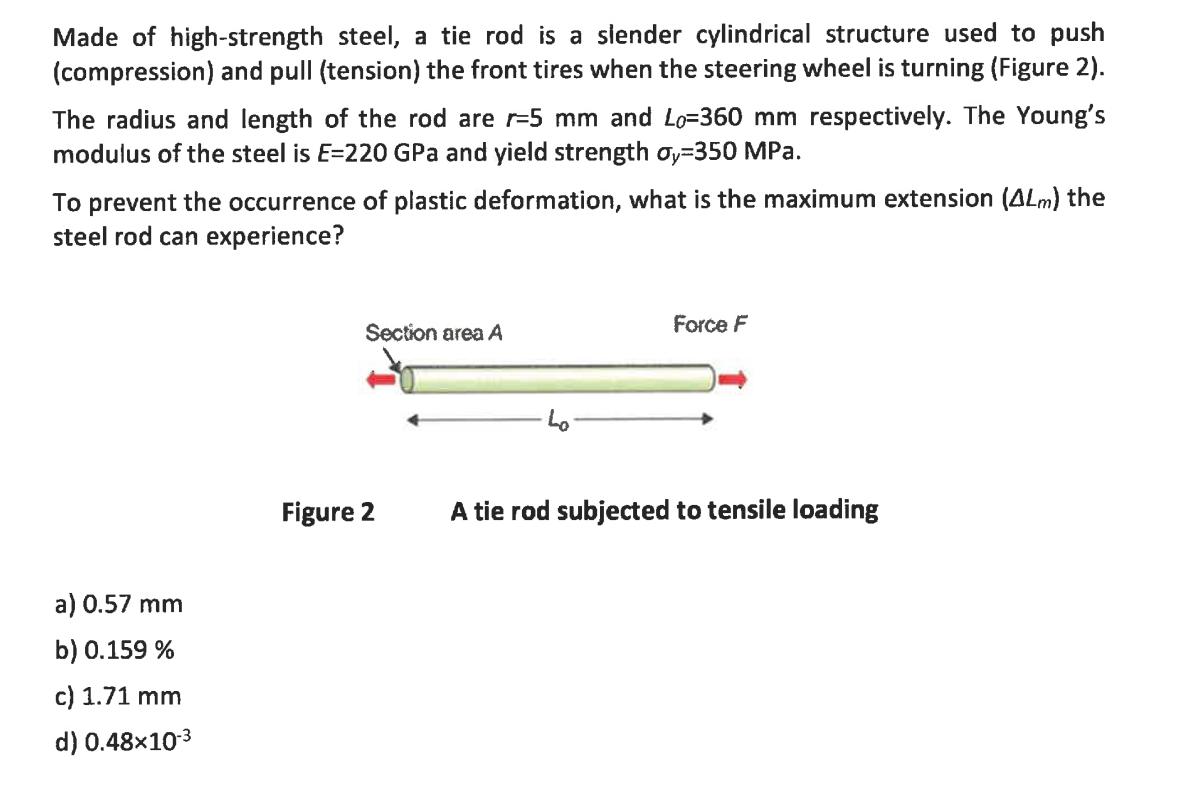
Made of high-strength steel, a tie rod is a slender cylindrical structure used to push (compression)...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Made of high-strength steel, a tie rod is a slender cylindrical structure used to push (compression) and pull (tension) the front tires when the steering wheel is turning (Figure 2). The radius and length of the rod are r-5 mm and Lo=360 mm respectively. The Young's modulus of the steel is E=220 GPa and yield strength oy-350 MPa. To prevent the occurrence of plastic deformation, what is the maximum extension (4Lm) the steel rod can experience? a) 0.57 mm b) 0.159 % c) 1.71 mm d) 0.48x10-3 Section area A Figure 2 Force F A tie rod subjected to tensile loading Please select the FALSE statement a) In a crystalline metallic structure, the atoms are arranged in a three-dimensional array called a lattice. b) A BCC unit cell consists of eight atoms at the corners of a cube and one atom at the body centre of the cube. c) Hydrogen bond is a type of bond, which occurs due to the ease with which hydrogen atoms are willing to give up an electron to atoms of oxygen, fluorine, or nitrogen (x) d) FCC crystal structures are more close-packed than BCC crystal structures. Made of high-strength steel, a tie rod is a slender cylindrical structure used to push (compression) and pull (tension) the front tires when the steering wheel is turning (Figure 2). The radius and length of the rod are r-5 mm and Lo=360 mm respectively. The Young's modulus of the steel is E=220 GPa and yield strength oy-350 MPa. To prevent the occurrence of plastic deformation, what is the maximum extension (4Lm) the steel rod can experience? a) 0.57 mm b) 0.159 % c) 1.71 mm d) 0.48x10-3 Section area A Figure 2 Force F A tie rod subjected to tensile loading Please select the FALSE statement a) In a crystalline metallic structure, the atoms are arranged in a three-dimensional array called a lattice. b) A BCC unit cell consists of eight atoms at the corners of a cube and one atom at the body centre of the cube. c) Hydrogen bond is a type of bond, which occurs due to the ease with which hydrogen atoms are willing to give up an electron to atoms of oxygen, fluorine, or nitrogen (x) d) FCC crystal structures are more close-packed than BCC crystal structures.
Expert Answer:
Answer rating: 100% (QA)
To find the maximum extension L without plastic deformation we can use Hookes Law which states that ... View the full answer

Related Book For 

Posted Date:
Students also viewed these programming questions
-
Irene Pypypiv has been working as a dishwasher at a bakery, while attending pastry arts school. As a graduation present, Irenes parents give her a graduation present of $40,000, which matches the...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Coffin Corporation appropriately uses the installment-sales method of accounting to recognize income in its financial statements. The following information is available for 2014 and 2015....
-
Describe how span of control, centralization, and formalization have changed at Merritt's Bakery over the years. Is the company's organizational structure today more mechanistic or organic? Are these...
-
In Problem perform the indicated operations and simplify. (m - n) (m + n)
-
Assume the same information as in question 4. Also assume that beginning work in process had \($6,000\) in conversion cost and that \($84,000\) in conversion is added during this period. What is the...
-
Weighted-average method. Ashworth Handcraft is a manufacturer of picture frames for large retailers. Every picture frame passes through two departments: the assembly department and the finishing...
-
What is Online transaction processing and Online Analytical Processing? How are they different?
-
How would the legal realists (e.g., Karl Llewellyn and Oliver Wendell Holmes) view the current relationship between social psychology and the law?
-
Airbus has found an 80% learning curve applies to direct labor for new production items. The first set of a new wing designed for a plane required 1,000 direct labor hours. How many total direct...
-
On the first day of the year 2019, a company issues a $50,000,000, 6%, 10-year bond that pays semi-annual interest, receiving cash of $52,000,000. Journalize the entry to record the first interest...
-
What are common allocation methods of pipeline imbalances in energy accounting?
-
What are the enterprise risks and opportunities for a company that has adopted a cryptocurrency strategy (e.g., Bitcoin)?
-
Washington Company has two divisions: the Adams Division and the Jefferson Division. The following information pertains to last year's results: Adams Division Jefferson Division Net (after-tax)...
-
Write a program that read a sequence of integer inputs and print (20 Points) a) The smallest and largest of the inputs. b) The number of even and odd inputs. c) Cumulative totals. For example, if the...
-
Assume that this is the beginning of year 2014 and you have joined a retail giant in France, a few days ago, as a marketing manager. You have been tasked with forecasting the sales for year 2014....
-
Aztec Furnishings makes hand-crafted furniture for sale in its retail stores. The furniture maker has recently installed a new assembly process, including a new sander and polisher. With this new...
-
Go to the Web site (http://www.umich.edu/~elements/6e/08chap/iclicker_ch8_q1.html) and view at least five i>clicker questions. Choose one that could be used as is, or a variation thereof, to be...
-
The elementary gas reaction 2A + B C is carried out isothermally in a PFR with no pressure drop. The feed is equal molar in A and B, and the entering concentration of A is 0.1 mol/dm 3 . Set up a...
-
What if you were asked to give an everyday example that demonstrates the principles discussed in this chapter? (Would sipping a teaspoon of Tabasco or other hot sauce be one?)
-
A candlestick chart is similar to a bar chart except that the candlestick chart: A. Represents upward movements in price with Xs. B. Also graphically shows the range of the periods highs and lows. C....
-
What considerations might encourage a market analyst to rely more on a top-down or bottom-up forecast of S&P 500 operating earnings?
-
Which of the following advantages listed by Ryan with respect to the earnings-based approach studied by Atkinson is most likely correct? The model A. Can be used for non-U.S. equity markets. B....

Study smarter with the SolutionInn App


