Solve the the data table to fill it Table 1. Raw and Processed Data for the Determination
Fantastic news! We've Found the answer you've been seeking!
Question:
Solve the the data table to fill it

Transcribed Image Text:
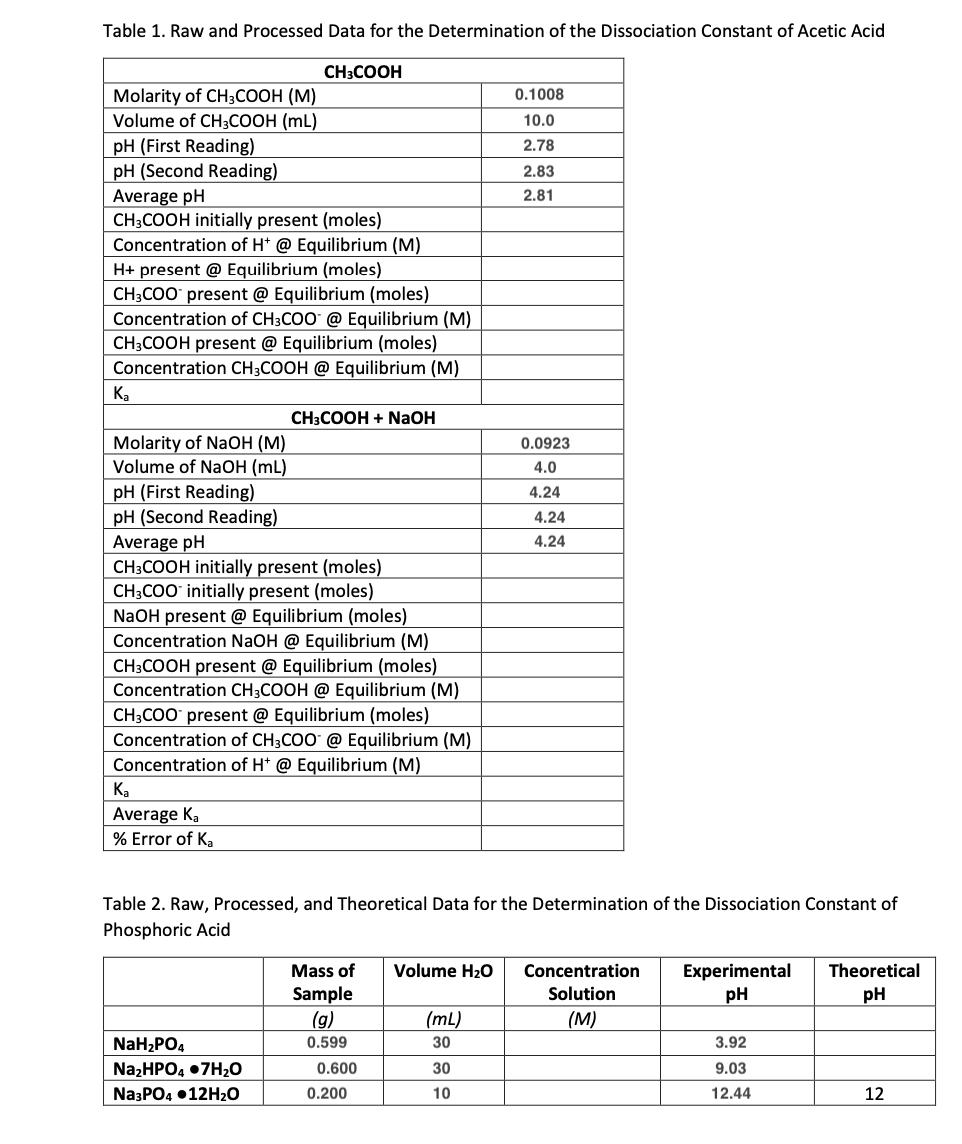
Table 1. Raw and Processed Data for the Determination of the Dissociation Constant of Acetic Acid Molarity of CH3COOH (M) Volume of CH3COOH (ML) pH (First Reading) pH (Second Reading) Average pH CH3COOH initially present (moles) Concentration of H+ @ Equilibrium (M) H+ present @ Equilibrium (moles) CH3COO present @ Equilibrium (moles) Concentration of CH3COO @ Equilibrium (M) CH3COOH present @ Equilibrium (moles) Concentration CH3COOH @ Equilibrium (M) Ka Molarity of NaOH (M) Volume of NaOH (mL) Ka Average Ka % Error of Ka CH3COOH pH (First Reading) pH (Second Reading) Average pH CH3COOH initially present (moles) CH3COO initially present (moles) NaOH present @ Equilibrium (moles) Concentration NaOH @ Equilibrium (M) CH3COOH present @ Equilibrium (moles) Concentration CH3COOH @ Equilibrium (M) CH3COO present @ Equilibrium (moles) Concentration of CH3COO @ Equilibrium (M) Concentration of H+ @ Equilibrium (M) NaH₂PO4 Na₂HPO4.7H₂O CH3COOH + NaOH Na3PO4 12H₂O Table 2. Raw, Processed, and Theoretical Data for the Determination of the Dissociation Constant of Phosphoric Acid Sample (g) 0.599 0.600 0.200 0.1008 10.0 2.78 Mass of Volume H₂O Concentration Experimental Theoretical pH pH Solution (M) (mL) 30 2.83 2.81 30 10 0.0923 4.0 4.24 4.24 4.24 3.92 9.03 12.44 12 Table 1. Raw and Processed Data for the Determination of the Dissociation Constant of Acetic Acid Molarity of CH3COOH (M) Volume of CH3COOH (ML) pH (First Reading) pH (Second Reading) Average pH CH3COOH initially present (moles) Concentration of H+ @ Equilibrium (M) H+ present @ Equilibrium (moles) CH3COO present @ Equilibrium (moles) Concentration of CH3COO @ Equilibrium (M) CH3COOH present @ Equilibrium (moles) Concentration CH3COOH @ Equilibrium (M) Ka Molarity of NaOH (M) Volume of NaOH (mL) Ka Average Ka % Error of Ka CH3COOH pH (First Reading) pH (Second Reading) Average pH CH3COOH initially present (moles) CH3COO initially present (moles) NaOH present @ Equilibrium (moles) Concentration NaOH @ Equilibrium (M) CH3COOH present @ Equilibrium (moles) Concentration CH3COOH @ Equilibrium (M) CH3COO present @ Equilibrium (moles) Concentration of CH3COO @ Equilibrium (M) Concentration of H+ @ Equilibrium (M) NaH₂PO4 Na₂HPO4.7H₂O CH3COOH + NaOH Na3PO4 12H₂O Table 2. Raw, Processed, and Theoretical Data for the Determination of the Dissociation Constant of Phosphoric Acid Sample (g) 0.599 0.600 0.200 0.1008 10.0 2.78 Mass of Volume H₂O Concentration Experimental Theoretical pH pH Solution (M) (mL) 30 2.83 2.81 30 10 0.0923 4.0 4.24 4.24 4.24 3.92 9.03 12.44 12
Expert Answer:
Answer rating: 100% (QA)
Answer Total answers posted by the expert is 1183 Acetic acid is a weak acid and ionizes as follows ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
Fill in the blanks in the table below and find the necessary sample sizes. One or Two Samples Type of Data Bound, Solve This B Margin of Error Sample Size Inequality p or o Binomial
-
Fill in the blanks in the table below and find the necessary sample sizes. One or Two Samples Margin of Error Type of Data Bound, Solve This Inequality Sample Size <4 n = n 2. s.10 n, = n2. p or o...
-
Using raw javascript and HTML create a webpage that has a digital clock and an analog clock. be sure to add a feature to switch between the clocks. the clock should show real time (eastern time)
-
Prepare journal entries to record the issuance of 100,000 shares of common stock at $20 per share for each of the following independent cases: a. Jackson Corporation has common stock with a par value...
-
Assume inflation is 0.2% per month. Would you rather earn a nominal return of 0.75% per month, or a real return of 6.5% APR, compounded annually?
-
At the end of the current year, Cuppa Joes inventory account balance was \($12,600.\) A physical count of the inventory revealed that inventory on hand totaled \($12,200. What\) amount should Cuppa...
-
The following data relate to notes receivable and interest for Owens Co., a financial services company. (All notes are dated as of the day they are received.) Mar. 8. Received a $33,000, 5%, 60-day...
-
Grammer and Language, Culture, and Community Review Questions 1. What sign movement is being used to communicate that something is done regularly or habitually? 2. Look at Figures 5.3-5.6. How are...
-
The Foxridge Investment Group buys and sells rental income properties in southwest Virginia. Bill Hunter, president of Foxridge, has asked for your assistance in analyzing a small apartment building...
-
A program to convert INFIX into POSTFIX using char stack and operator precedence.
-
1) Using the data below and T-accounts, convert the cash basis income statement to accrual basis in appropriate professional Excel format (i.e., use cell referencing and formulas, no hard coding...
-
Tyler Tooling Company uses a job order cost system with overhead applied to products on the basis of machine hours. For the upcoming year, the company estimated its total manufacturing overhead cost...
-
Laker Incorporated s fiscal year - end is December 3 1 , 2 0 2 4 . The following is an adjusted trial balance as of December 3 1 . Accounts Debit Credit Cash $ 1 0 , 6 0 0 Supplies 3 2 , 0 0 0...
-
On March 1 , 2 0 2 3 , VisionTech Inc. s board of directors declared a 5 % share dividend when the market price per share was $ 2 8 . 0 0 . On November 1 5 , 2 0 2 3 , the board of directors declared...
-
Explain trojan horse and its types with example and diagram?
-
Q1: A 0.4 m rigid tank contains saturated mixture of R134a at 0.16 MPa. If the specific volume of the mixture is 0.1 m/kg. determine: (a) The total mass of the mixture. (b) The quality of the...
-
Distinguish among total-moisture content, free-moisture content, equilibrium-moisture content, unbound moisture, and bound moisture.
-
(a) Write an equation for the reaction in which H2C6H7O5 - (aq) acts as a base in H2O(l). (b) Write an equation for the reaction in which H2C6H7O5 - (aq) acts as an acid in H2O(l). (c) What is the...
-
Write the molecular and structural formulas for the compounds represented by the following models: (a) (b) (c) (d) Cl Cl
-
(a) What is meant by the term reaction rate? (b) Name three factors that can affect the rate of a chemical reaction? (c) Is the rate of disappearance of reactants always the same as the rate of...
-
The purpose of a link is to (a) transmit motion (b) guide other links (c) act as a support (d) all of the above.
-
Calculate the number of degrees of freedom of the mechanisms shown in Fig.1.53. (a) (b) Fig.1.53 Various types of mechanisms (c)
-
A kinematic chain requires at least (a) 2 links and 3 turning pairs (b) 3 links and 4 turning pairs (c) 4 links and 4 turning pairs (d) 5 links and 4 turning pairs.

Study smarter with the SolutionInn App


