please write clearly Write the cell notation for an electrochemical cell consisting of an anode where Mg
Fantastic news! We've Found the answer you've been seeking!
Question:

 please write clearly
please write clearly
Transcribed Image Text:
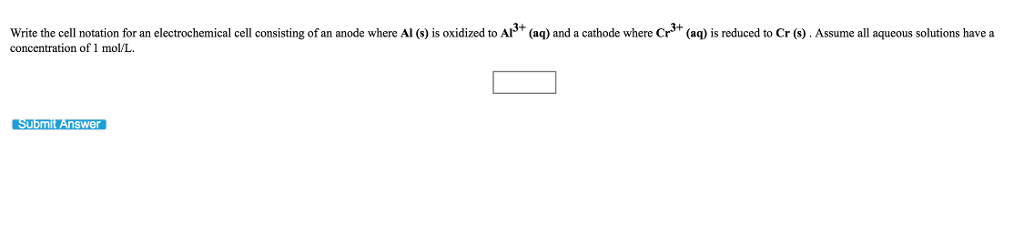
Write the cell notation for an electrochemical cell consisting of an anode where Mg (s) is oxidized to Mg2+ (aq) and a cathode where Fe³+ (aq) is reduced to Fe²+ (aq) at a platinum electrode. Assume all aqueous solutions have a concentration of 1 mol/L and gases have a pressure of 1 bar. Submit Answer 0 Write the cell notation for an electrochemical cell consisting of an anode where Al (s) is oxidized to Al³+ (aq) and a cathode where Cr³+ (aq) is reduced to Cr (s). Assume all aqueous solutions have a concentration of 1 mol/L. Submit Answer Write the cell notation for an electrochemical cell consisting of an anode where Mg (s) is oxidized to Mg2+ (aq) and a cathode where Fe³+ (aq) is reduced to Fe²+ (aq) at a platinum electrode. Assume all aqueous solutions have a concentration of 1 mol/L and gases have a pressure of 1 bar. Submit Answer 0 Write the cell notation for an electrochemical cell consisting of an anode where Al (s) is oxidized to Al³+ (aq) and a cathode where Cr³+ (aq) is reduced to Cr (s). Assume all aqueous solutions have a concentration of 1 mol/L. Submit Answer
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Write an equation that clearly shows the structure of the alcohol obtained from the sequential hydroboration and H2O2/OH oxidation of a. b.
-
Write an equation, clearly showing the stereochemistry of the starting material and the product, for the reaction of (S)-1-bromo-2-methylbutane with sodium iodide in acetone. What is the...
-
Write equations that clearly show the mechanism for the acid-catalyzed hydrolysis of a. Maltose to glucose b. Lactose to galactose and glucose c. Sucrose to fructose and
-
In Exercises confirm that the Integral Test can be applied to the series. Then use the Integral Test to determine the convergence or divergence of the series. n=1 2 3n + 5
-
a. Draw a budget line for Rafaella, who has a weekly income of $30. Assume that she buys chicken and eggs, and that chicken costs $5 per pound while eggs cost $1 each. Add an indifference curve for...
-
Mercury at 210F. Appendix D gives dynamic viscosity for a variety of fluids as a function of temperature. Using this appendix, give the value of the viscosity for the following fluids
-
Speegleville Marina needs to raise \(\$ 1\) million to expand. Speegleville's president is considering two plans: - Plan A: Issue \(\$ 1,000,000\) of \(8 \%\) bonds payable to borrow the money - Plan...
-
What SQL command(s) would you use to add the date on which an employee was hired to the EMPLOYEE table represented in Figure? Name this new attribute Employment_ Date. Assume that the employees were...
-
a share os stock sells for $35 today. the beta of the stock is 1.2 and the expected return on the market is 12%. the stock is expected to pay a dividend of $0.80 in one year. if the risk free rate is...
-
In addition to providing polarity data, like those you interpreted in Part B, most terrestrial lava flows contain minerals that can be precisely dated using radiometric age-dating techniques. The...
-
AIM: Write C program to access elements using pointer.
-
A company begins operations in March and has the following transactions. March 1 Issue common stock for $18,000. March 5 Obtain $8,400 loan from the bank by signing a note. March 10 Purchase...
-
You are planning a Teams Phone integration. The solution must meet the following requirements: Maintain an existing contract with a local PSTN carrier. Integrate with a local PBX. What should you...
-
1. Identify common risk factors for experiencing a childhood trauma and then developing trauma- or stressor-related disorders. Include biological and environmental factors such as home, family,...
-
Educational Tools Chart Part 1: Complete the following chart. Select tools that are appropriate for an early learning environment. Format citations according to APA guidelines. Name of Educational...
-
On January 1, 2019, Rand Corp. issued shares of its common stock to acquire all of the outstanding common stock of Spaulding Inc. Spaulding's book value was only $140,000 at the time, but Rand issued...
-
GDP between the two years and discuss the result. GDP in 2018; c) the rates of 2. Given the following data representing the goods market of an open economy: Marginal Propensity to consume Direct Tax...
-
In exchange for land, the company received a 12-month note on January 1. The face amount of the note is $1,000, and the stated rate of interest is 13%, compounded annually. The 13% rate is equal to...
-
The IO3 anion is called iodate. There are three related ions: IO, IO2, and IO4. Using what you have learned about similar groups of anions, write the name for each of the following compounds:...
-
An ideal gas with a density of 3.00 g/L has a pressure of 675 mmHg at 25oC. What is the root-mean-square speed of the molecules of this gas?
-
Write Lewis formulas for the following: a. XeF2 b. SeF4 c. TeF6 d. XeF5+
-
Examine Riverside's assets as follows: 1. What is Riverside's largest category of assets? List all 2020 assets in the largest category and their amounts as reported by Riverside. 2. What was...
-
Assume WestJet repaired one of its Boeing 737 aircraft at a cost of \(\$ 0.8\) million, which West Jet paid in cash. Further, assume that the WestJet accountant erroneously capitalized this cost as...
-
Assume that at the beginning of 2020, Porter Airlines purchased a Bombardier Q400 aircraft at a cost of \(\$ 25,000,000\). Porter expects the plane to remain useful for five years \((5,000,000...

Study smarter with the SolutionInn App


