Plot the polarization data shown below for an electrode of 1.00 cm of iron in an...
Fantastic news! We've Found the answer you've been seeking!
Question:
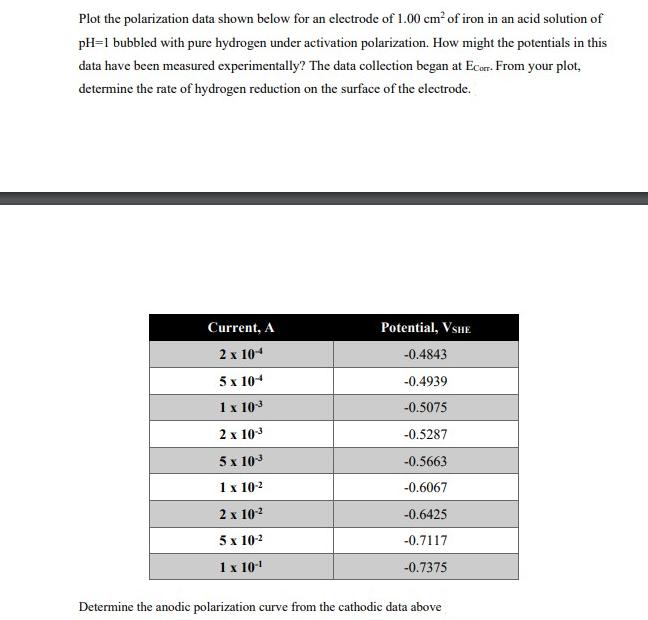
Transcribed Image Text:
Plot the polarization data shown below for an electrode of 1.00 cm² of iron in an acid solution of pH=1 bubbled with pure hydrogen under activation polarization. How might the potentials in this data have been measured experimentally? The data collection began at Ecorr. From your plot, determine the rate of hydrogen reduction on the surface of the electrode. Current, A 2 x 104 5 x 104 1 x 10-³ 2 x 10³ 5 x 10-3 1 x 10-² 2 x 10-² 5 x 10-² 1 x 10-¹ Potential, VSHE -0.4843 -0.4939 -0.5075 -0.5287 -0.5663 -0.6067 -0.6425 -0.7117 -0.7375 Determine the anodic polarization curve from the cathodic data above Plot the polarization data shown below for an electrode of 1.00 cm² of iron in an acid solution of pH=1 bubbled with pure hydrogen under activation polarization. How might the potentials in this data have been measured experimentally? The data collection began at Ecorr. From your plot, determine the rate of hydrogen reduction on the surface of the electrode. Current, A 2 x 104 5 x 104 1 x 10-³ 2 x 10³ 5 x 10-3 1 x 10-² 2 x 10-² 5 x 10-² 1 x 10-¹ Potential, VSHE -0.4843 -0.4939 -0.5075 -0.5287 -0.5663 -0.6067 -0.6425 -0.7117 -0.7375 Determine the anodic polarization curve from the cathodic data above
Expert Answer:
Answer rating: 100% (QA)
Solutions Step 1 First we need to find out the logarithmic of current from the data given be... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder, ? Ronald W. Rousseau, ? Lisa G. Bullard
Posted Date:
Students also viewed these chemical engineering questions
-
A company has two plants and three warehouses. Plant P1 can produce at most 100 units and Plant P2 can produce at most 200 units of the same product. The sales potential at Warehouse W1 is 150, 200...
-
A rope is wrapped exactly three times around a cylinder with a fixed axis of rotation at its centre. The cylinder has a mass of 250 kg and a diameter of 34.0 cm. The rope is pulled with a constant...
-
A record company needs to produce 100 gold records at one or more of its three studios. the cost of producing x records at studio 1 is 10 x; the cost of producing y records at studio 2 is 2y 2 ; the...
-
The following are the trial balance and other information related to Yorkis Perez, a consulting engineer. 1. Fees received in advance from clients R$6,000. 2. Services performed for clients that were...
-
A $ 1,000 face value corporate bond with a 6.75 percent coupon (paid semiannually) has 10 years left to maturity. It has had a credit rating of BB and a yield to maturity of 8.2 percent. The firm...
-
Armoring Military Planes During the Second World War, the U.S. military collected data on bullet holes found in B-24 bombers that returned from flight missions. The data showed that most bullet holes...
-
What are the advantages of establishing pay ranges, rather than specific pay levels, for each job? What are the drawbacks of this approach?
-
As of December 31 of the current year, Armani Company's records show the following. Cash Accounts receivable $ 11,800 10,800 Supplies 7,800 Equipment 6,800 Accounts payable 14,600 Common stock 15,800...
-
In June 2021, Karen transferred property with a $75,000 FMV and a $20,000 adjusted basis to Hal, her husband. Hal dies in March 2022; the property has appreciated to $85,000 in value by then. His...
-
Last year Janet purchased a $1,000 face value corporate bond with a 9% annual coupon rate and a 10-year maturity. At the time of the purchase, it had an expected yield to maturity of 9.15%. If Janet...
-
You plan to open a retirement account. Your employer will match 50 percent of your deposits up to a limit on the match of \($2\),500 per year. You believe the fund will earn 12 percent per year over...
-
On Juans twenty-sixth birthday, he invested $7,500 in a retirement account. Each year thereafter, he deposited 8 percent more than the previous deposit. The account paid annual compound interest of 5...
-
What equal monthly investment is required over a 40-year period to achieve a balance of $2,000,000 in an investment account that pays monthly interest of 3/4 percent?
-
A \($90\),000 investment is made. Over a 5-year period, a return of \($30\),000 occurs at the end of the first year. Each successive year yields a return that is 10 percent less than the previous...
-
You invest $10,000 in a fund that pays 7 percent per year for 5 years. How much is in the fund at the end of 5 years if (forgetting leap years and making convenient assumptions) a. compounding is...
-
Chec Sandy Bank, Inc., makes one model of wooden canoe. Partial information is given below. Required: 1. Complete the following table. 2. Suppose Sandy Bank sells its canoes for $590 each. Calculate...
-
From the choice of simple moving average, exponential smoothing, and linear regression analysis, which forecasting technique would you consider the most accurate? Why? please write it in word...
-
The level of toluene (a flammable hydrocarbon) in a storage tank may fluctuate between 10 and 400 cm from the top of the tank. Since it is impossible to see inside the tank, and open-end manometer...
-
An iron bar 2.00 cm x 3.00 cm x 10.0 cm at a temperature of 95C is dropped into a barrel of water at 25C. The barrel is large enough so that the water temperature rises negligibly as the bar cools....
-
A liquid stream consisting of 12.5 mole% n-butane and the balance a heavy nonvolatile hydrocarbon is fed to the top of a stripping column, where it is contacted with an upward-flowing stream of...
-
Give an example of a requirement on a computer printer.
-
Briefly describe the distinction between requirements and specification.
-
Give an example of a requirement on a digital still camera.

Study smarter with the SolutionInn App


