Predict major product(s) in each of the following Friedel-Crafts reactions. If the reaction does not occur, offer
Fantastic news! We've Found the answer you've been seeking!
Question:
Predict major product(s) in each of the following Friedel-Crafts reactions. If the reaction does not occur, offer a reasonable explanation for that fact.
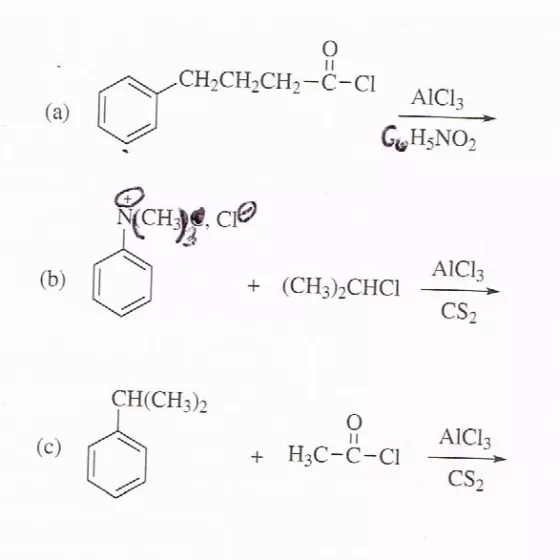
Predict major product(s) in each of the following Friedel-Crafts reactions. If the reaction does not occur, offer a reasonable explanation for that fact.
Transcribed Image Text:
II CH2CH2CH2-C-CI AICI3 (а) GH;NO2 CH CIO AICI3 (b) + (CH):СHCI CS2 CH(CH3)2 AIC13 (с) + H3C-C-CI CS2 II CH2CH2CH2-C-CI AICI3 (а) GH;NO2 CH CIO AICI3 (b) + (CH):СHCI CS2 CH(CH3)2 AIC13 (с) + H3C-C-CI CS2
Expert Answer:
Posted Date:
Students also viewed these chemistry questions
-
Predict the major product obtained when each of the following compounds is treated with Birch conditions. a. b. c. d. e. f. OH
-
Predict the major product obtained when each of the following compounds is treated with bromine (Br 2 ) together with sodium hydroxide (NaOH) followed by aqueous acid (H 3 O + ). (a) (b) (c)
-
Predict the major organic product in each of the following reactions: (a) (b) (c) (d) (e) (f) Cl NO2 CH3 Cl H,NNH triethylene glycol NO2 Cl CI L. HNO, H SO. 120C 2. NH, ethylene glycol, 140C CF3 L...
-
An excise tax of $1.00 per gallon of gasoline placed on the suppliers of gasoline would shift the supply curve Multiple Choice O down by $1.00. down by more than $1.00. up by $1.00. up by less than...
-
Gene Horita was choosing a major in his sophomore year. He enjoyed the accounting classes at his university, but also found the information systems classes to be interesting. Genes father is a public...
-
Find I1, I2 and V1 in the circuit in figure. 3 A V1 402 60 w-
-
Indicate whether the following items, a through j, are (1) typical characteristics of open-book management or (2) not typical characteristics of open-book management. a. Considered a large size...
-
The following information is available for Shanika Company for 2016: Instructions 1. Prepare the 2016 statement of cost of goods manufactured. 2. Prepare the 2016 income statement. Inventories...
-
PAPER TOPIC: Seasonal Employees & Cybersecurity Provide written critiques for these short papers written by your peers. Your critiques of classmates' work should be constructive and offer 3 or more...
-
1. Using the spreadsheet model from Case 2.1 as a starting point, use Solver to find the optimal set of projects to approve. The solution should maximize the total NPV from the approved projects, and...
-
The number of real solutions of the equation i+1 sin' (3) 2 i=1 i=1 lying in the interval 2 cos-x assume values in - COS (-2) (Here, the inverse trigonometric functions sin x and is (1) i=1 22...
-
Two subtracted from five times a number is 103. What is the A) Translate the statement above into an equation that you can solve to answer this question. Do not solve it yet. Use x as your variable....
-
How can Google Cloud compete and gain market share against AWS and Azure?
-
y-5 B) Enter the expression 5y Your input: I Basic Funcs Trig X
-
Find the distance between the points ( 1 5 , 1 5 ) and ( 4 , 7 ) . Also find the midpoint of the line segment joining the two points.
-
Explain what is Model, Strategy, Algorithm in math teaching? How is it similar/compare to 'algorithm' language of borrow or carry over in subtraction
-
900 0000 2002ENG - Fluid Mechanics and Hydraulics Provide detailed calculation for one submerged case. Case 3: 300g, Fully submerged Hydrostatic Pressure Experiment
-
1. True or False. Pitfalls to consider in a statistical test include nonrandom samples, small sample size, and lack of causal links. 2. Because 25 percent of the students in my morning statistics...
-
What is the Fickian diffusivity of chlorobenzene in liquid bromobenzene at \(300 \mathrm{~K}\) when the mole fraction of chlorobenzene is 0.0332 ? Assume that the diffusivity follows an Arrhenius...
-
a. Estimate the Fickian diffusivity of a binary mixture of benzene and air at \(298.2 \mathrm{~K}\) and \(1.0 \mathrm{~atm}\) pressure using Chapman-Enskog theory and Table 15-2. b. Compare your...
-
Water at \(60^{\circ} \mathrm{C}\) and 0.95 bar is evaporating into a \(12.0 \mathrm{~cm}-1\) ong tube (also at \(60^{\circ} \mathrm{C}\) ) and diffusing through a stagnant layer of air. The device...

Study smarter with the SolutionInn App


