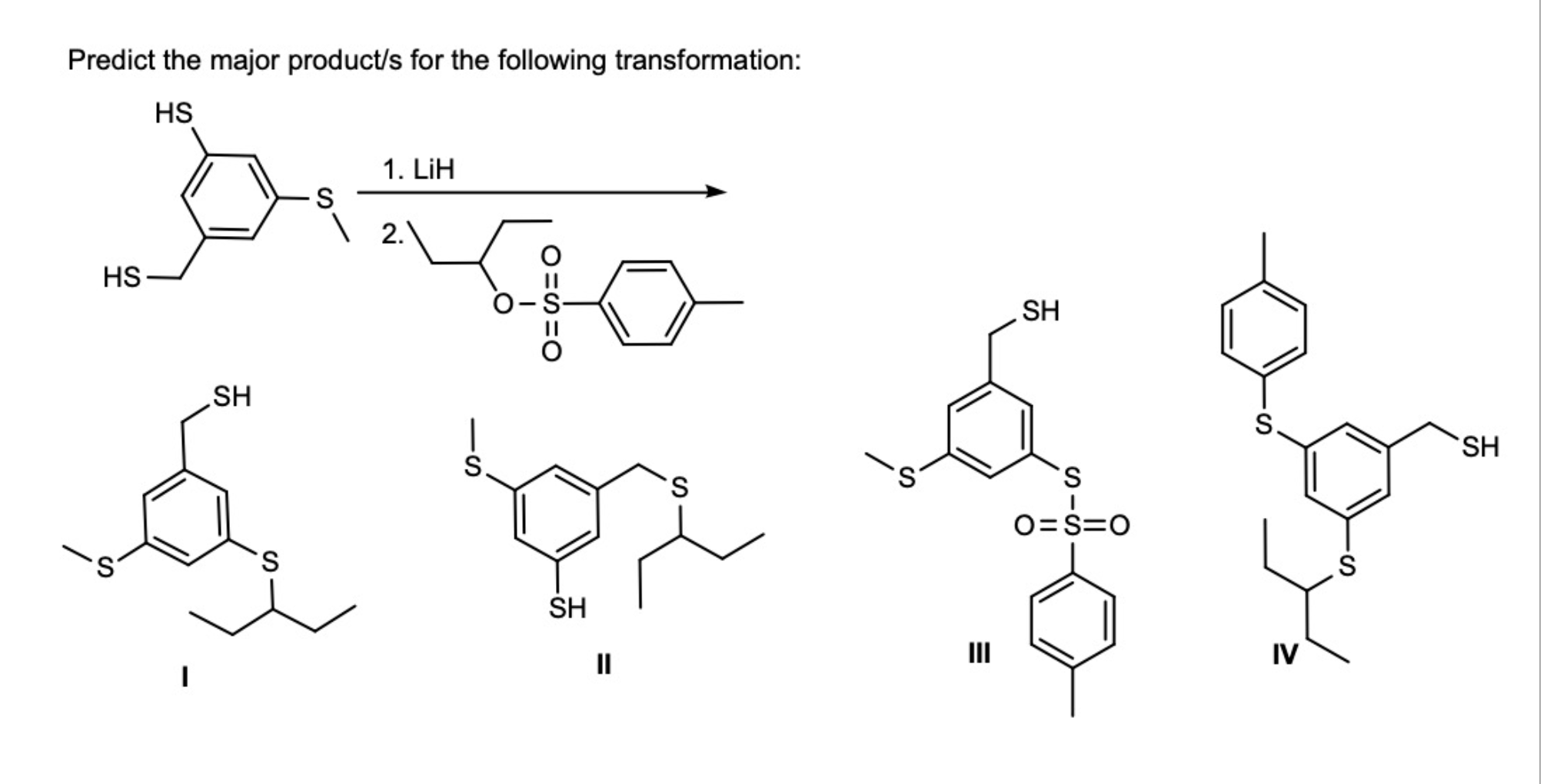
Predict the major product/s for the following transformation: HS HS .S SH...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
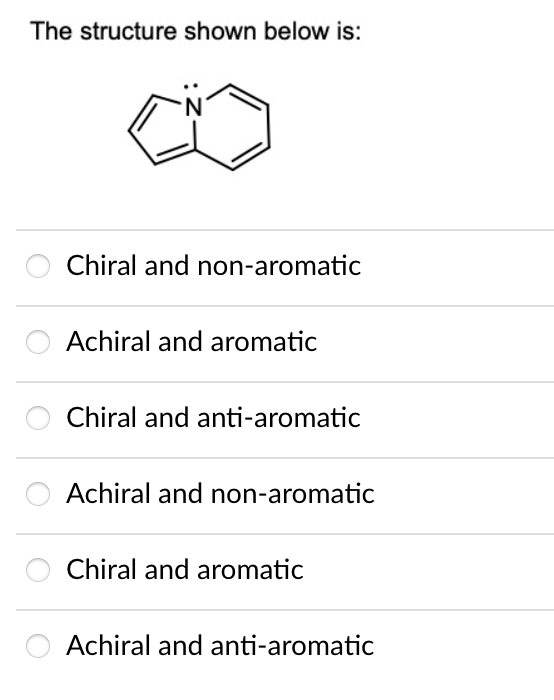
Predict the major product/s for the following transformation: HS مه ہو و لکه HS .S SH S S 1. LiH 2. S SH II S S SH S 0=S=0 S. IV S SH If benzyl phenoxide is mixed with large excess of hydrobromic acid, what is/are the product/s of interest obtained from this reaction? a) benzene. b) toluene. c) phenol. d) benzyl alcohol. e) benzyl bromide. f) bromobenzene. g) bromine. h) xylene. The structure shown below is: Chiral and non-aromatic Achiral and aromatic Chiral and anti-aromatic Achiral and non-aromatic Chiral and aromatic Achiral and anti-aromatic Predict the major product/s for the following transformation: HS مه ہو و لکه HS .S SH S S 1. LiH 2. S SH II S S SH S 0=S=0 S. IV S SH If benzyl phenoxide is mixed with large excess of hydrobromic acid, what is/are the product/s of interest obtained from this reaction? a) benzene. b) toluene. c) phenol. d) benzyl alcohol. e) benzyl bromide. f) bromobenzene. g) bromine. h) xylene. The structure shown below is: Chiral and non-aromatic Achiral and aromatic Chiral and anti-aromatic Achiral and non-aromatic Chiral and aromatic Achiral and anti-aromatic
Expert Answer:

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
Predict the major product (or products) that would be obtained when each of the following compounds is nitrated: (a) (b) (c) OH CF CN SO3H OCH3 NO2
-
Predict the major product (or products) formed when each of the following reacts with a mixture of concentrated HNO3 and H2SO4. (a) (b) (c) 4-Chlorobenzoic acid (d) 3-Chlorobenzoic acid (e)...
-
For each compound, predict the major product of free-radical bromination. Remember that bromination is highly selective, and only the most stable radical will be formed. (a) Cyclo-hexane (b)...
-
Gerald De Beer is a 38-year-old vibrant, outgoing young man who has been employed as a digital artist at Innovate Tech Solutions (Pty) Ltd ("Innovate Tech") since 1 May 2019. Details of Geralds...
-
It would also be possible to use a block design in this study. Sort the before CFU counts into three blocks (representing low, medium, and high CFU counts). Each block should have 10 units. Analyze...
-
Conduct an internet search and find the balance sheet of a publicly traded company in its financials. Analyze the assets section and explain in which accounts the company's main asset strength lies....
-
McKeller Company would like to start a new venture. The company is currently in the 24% marginal tax bracket and uses a 5% discount factor. The company projects that the venture will produce...
-
Spectra, Inc., produces semiconductors of which part no. 200 is a subassembly. Spectra, Inc., currently produces part no. 200 in its own shop. The Alta Company offers to supply it at a cost of $200...
-
Fellow Design Sdn Bhd (FD) intends to incorporate machine learning (ML) components into their eCommerce platform. Their primary objective is to offer furniture for sale, accompanied by personalized...
-
Gold has a density of 19.3 g/mL. Suppose you have 100.0 glonkins of gold. What volume in liters will the gold occupy? Here are some conversion factors to help you: 0.911 ounce per glonkin and 28.35 g...
-
Discuss the definition of externality, explain why government intervention is usually required to address the economic failure that results, and describe how taxes are used to fund this.
-
Which of the following statements is not required to be presented for an Investment Trust Fund? a. Statement of net assets. b. Statement of changes in net assets. c. Statement of cash flows. d. All...
-
Restricted net assets for a governmental college or university are commonly subclassified for a. designated net assets. b. reserved net assets. c. nonexpendable net assets. d. expendable net assets....
-
Which of the following must be prepared for Agency Funds as part of a governments Basic Financial Statements? a. Statement of changes in net assets. b. Statement of revenues, expenditures, and...
-
Government colleges and universities solely engaged in business-type activities would present the following classes of equity except a. invested in capital assets, net of related debt. b. restricted...
-
How should government college and university revenues and expenses be classified for external financial reporting purposes?
-
Subject: Accounting Answer the following with complete and detailed solution. Use excel for better understanding and find the balances needed then prepare financial position and notes to financial...
-
Use this circle graph to answer following Exercises. 1. What fraction of areas maintained by the National Park Service are designated as National Recreation Areas? 2. What fraction of areas...
-
Which atoms in each of the following molecules are chirality centers? (a) (b) (c) (d) OH OH Lactic acid Glyceraldehyde OH HO 0 0 OH Ascorbic acid (vitamin C) OH HO Estradiol (an estrogen)
-
(a) When cis-1-bromo-2-methylcyclohexane undergoes an E2 reaction, two products (cycloalkenes) are formed. What are these two cycloalkenes, and which would you expect to be the major product? Write...
-
Consider these reactions: The intermediate A is a covalently bonded compound that has typical 1H NMR signals for aromatic ring hydrogens and only one additional signal at δ 1.21, with an...
-
What are the functions of regulatory bodies in safety assessment of nuclear reactors?
-
Explain the difference between DEC-A and DEC-B?
-
Explain the deterministic safety assessment (DSA) of nuclear reactors and development growth of this methodology over the years.

Study smarter with the SolutionInn App


