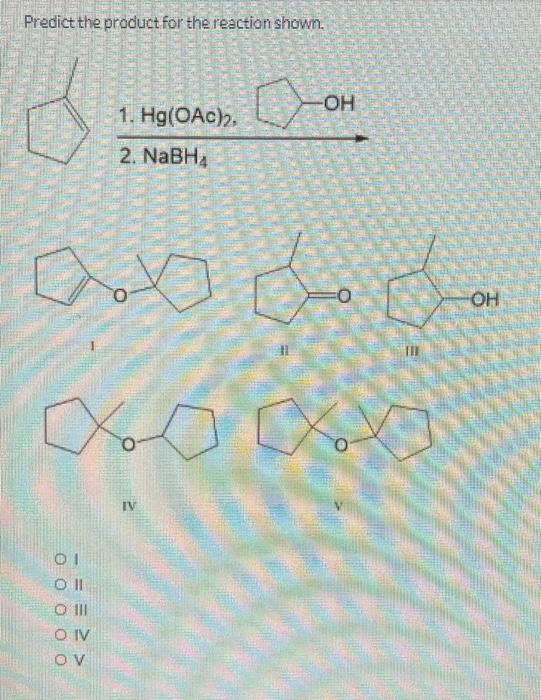
Predict the product for the reaction shown. 1. Hg(OAC), 2. NaBHA 01 Oll O III OIV...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
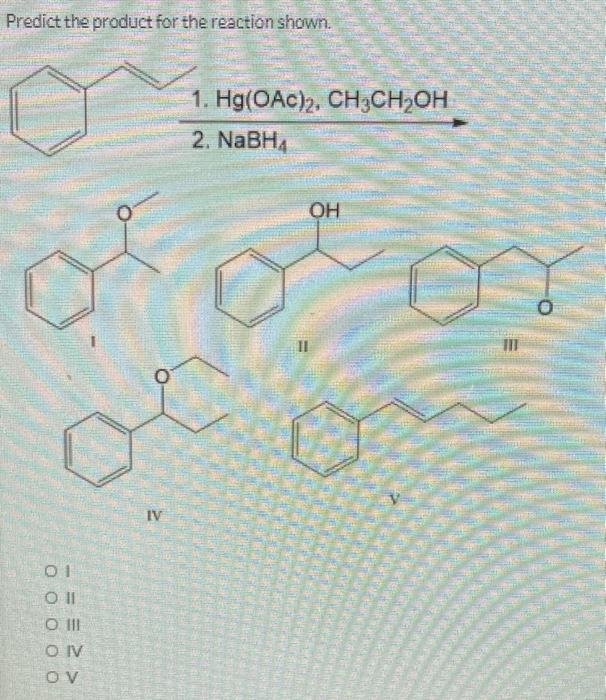
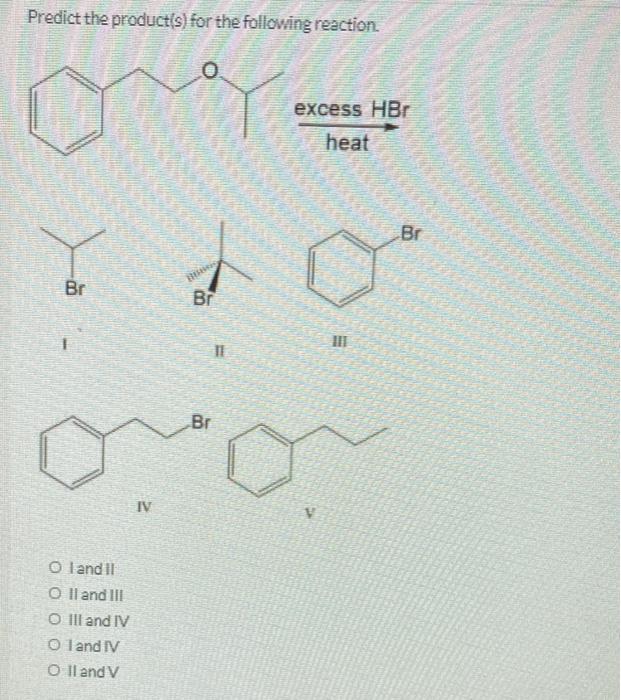
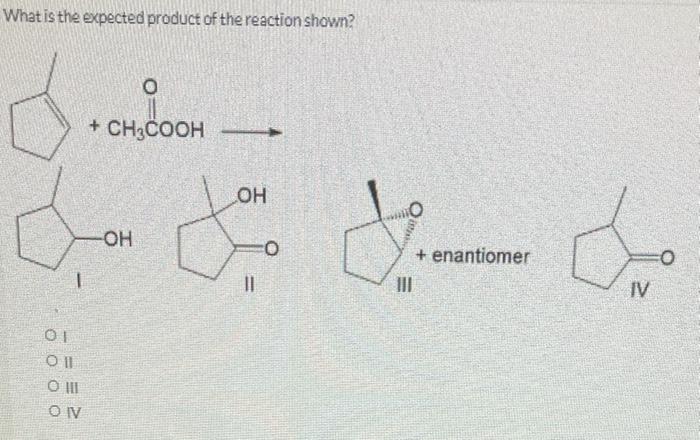
Predict the product for the reaction shown. 1. Hg(OAC), 2. NaBHA 01 Oll O III OIV OV من ا من 000 جلاه -OH مه ممه IV OH Predict the product for the reaction shown. 01 Oll CO III OIV OV 0 IV 1. Hg(OAc)2, CH3CH₂OH 2. NaBH₁ SALLE VENDARY OH II PRONTIER AUS Predict the product(s) for the following reaction. O Y Br Oland Il O II and III O III and IV Oland IV O II and V IV Br Br TI excess HBr heat Br What is the expected product of the reaction shown? O dondoom + CH3COOH 1 01 Coll O III CONV -OH OH fou 11 O + enantiomer IV Predict the product for the reaction shown. 1. Hg(OAC), 2. NaBHA 01 Oll O III OIV OV من ا من 000 جلاه -OH مه ممه IV OH Predict the product for the reaction shown. 01 Oll CO III OIV OV 0 IV 1. Hg(OAc)2, CH3CH₂OH 2. NaBH₁ SALLE VENDARY OH II PRONTIER AUS Predict the product(s) for the following reaction. O Y Br Oland Il O II and III O III and IV Oland IV O II and V IV Br Br TI excess HBr heat Br What is the expected product of the reaction shown? O dondoom + CH3COOH 1 01 Coll O III CONV -OH OH fou 11 O + enantiomer IV
Expert Answer:
Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
What is the expected rate of return for a stock that has a beta of 1.0 if the expected return on the market is 15%? a. 15%. b. More than 15%. c. Cannot be determined without the risk-free rate.
-
What is the expected value of perfect information?
-
What is the expected composition of the future diverse workforce by 2050?
-
Mr. Yap bought a bond having a face value of Php 1,000.00 for Php 970.00. The bond rate was 14% nominal and dividends were made to him semiannually for a total of 7 years. At the end of the seventh...
-
Assume the industry you are analyzing is in the fourth stage of the industrial life cycle. How would you react if your industry-economic analysis predicted that sales per share for this industry...
-
True or False: When using the Eastman hurdle rate calculator, if venture capital is being sought, the hurdle rate obtained will not be affected by ownership, plant site, or technology profiles.
-
In the benzene adsorber of Example 9.7, the flow rate is increased to \(0.25 \mathrm{~m}^{3} / \mathrm{s}\). Calculate the breakthrough time and the fraction of the bed adsorption capacity that has...
-
Raven Design is a website design and consulting firm. The firm uses a job cost system, in which each client is a different job. Raven Design traces direct labor, licensing costs, and travel costs...
-
Consider the acceleration of a good sports car in real life . (1) Pick a certain car model, which could be the one you are driving, or the one you wish you were --- and search online for its...
-
Fokine Research Institute has three departments: biology, chemistry, and physics. The institute's controller wants to estimate the cost of operating each department. He has identified several...
-
Susan Price, from Arkansas, sues Stuff-Mart in state court in Arkansas. Stuff-Mart is incorporated in Arkansas, and its principle place of business is in Arkansas. Price's lawsuit alleges that...
-
When a landlord sells leased premises to a third party, any existing leases terminate automatically. (True/False)
-
Mercer Management is a consulting firm that operates an information technology (IT) center to bring greater efficiencies to other businesses and thereby help those companies cut costs. In Mercers IT...
-
Good Healthcare, Inc., initiates an attempt to purchase enough shares in Home Health Aides Corporation to control it. This process is a corporate a. consolidation. b. merger. c. liquidation. d....
-
All employers are subject to Title VII of the Civil Rights Act of 1964 regardless of the number of their employees. (True/False)
-
An agent can perform legal acts that bind the principal. (True/False)
-
For each of the following accounts, TO THE LEFT place the letter of the financial statement where the account is found. TO THE RIGHT place the letter DR for a normal debit balance or CR for a normal...
-
6. (Potential Energy and Conservation of Energy) What should be the spring constant k of a spring designed to bring a 1200-kg car to rest from a speed of 95 km/h so that the occupants undergo a...
-
Using the rules found in this chapter, give systematic names for the following or indicate that more rules need to be provided: (a) (b) (c) (d) Cl CI H C-B Br Br
-
Write the structures of two chair conformations of 1-tert-butyl-1-methylcyclohexane. Which conformation is more stable? Explain your answer.
-
The tert-butyl group can be used as a blocking group in certain syntheses of aromatic compounds. (a) How would you introduce a tert-butyl group? (b) How would you remove it? (c) What advantage might...
-
Rewrite the Gross-Pitaevskii equation and the mean field energy, see equations (11.2.21) and (11.2.23), for an isotropic harmonic oscillator trap with frequency \(\omega_{0}\) in a dimensionless form...
-
The energy levels of an imperfect Fermi gas in the presence of an external magnetic field \(\boldsymbol{H}\), to the first order in \(a\), may be written as...
-
Solve the Gross-Pitaevskii equation (11.2.23) in a harmonic trap for the case when the scattering length \(a\) is zero. Show that this reproduces the properties of the ground state of the...

Study smarter with the SolutionInn App



