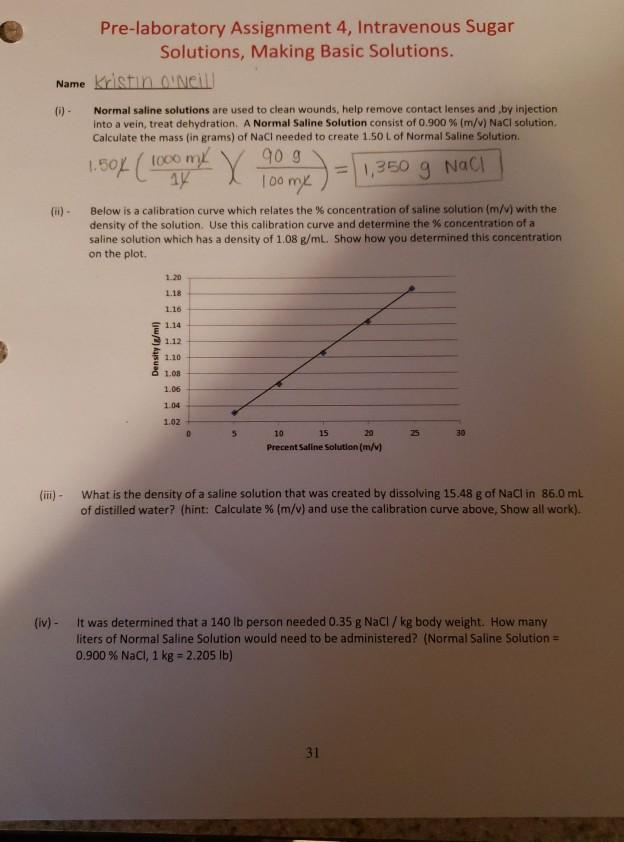
Pre-laboratory Assignment 4, Intravenous Sugar Solutions, Making Basic Solutions. Name Kristin O'Neill (i) - Normal saline...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Pre-laboratory Assignment 4, Intravenous Sugar Solutions, Making Basic Solutions. Name Kristin O'Neill (i) - Normal saline solutions are used to clean wounds, help remove contact lenses and by injection into a vein, treat dehydration. A Normal Saline Solution consist of 0.900 % (m/v) NaCl solution. Calculate the mass (in grams) of NaCl needed to create 1.50 L of Normal Saline Solution. = 1,350 g NaCl 1.50% (1000 m X 90 g 100 mx Below is a calibration curve which relates the % concentration of saline solution (m/v) with the density of the solution. Use this calibration curve and determine the % concentration of a saline solution which has a density of 1.08 g/mL. Show how you determined this concentration on the plot. Density (g/ml) 1.20 1.18 1.16 1.14 1.12 1.10 1.08 1.06 1.04 1.02 5 10 15 20 Precent Saline Solution (m/v) 30 (iii) - What is the density of a saline solution that was created by dissolving 15.48 g of NaCl in 86.0 mL of distilled water? (hint: Calculate % (m/v) and use the calibration curve above, Show all work). (iv) - It was determined that a 140 lb person needed 0.35 g NaCl / kg body weight. How many liters of Normal Saline Solution would need to be administered? (Normal Saline Solution = 0.900 % NaCl, 1 kg = 2.205 lb) 31 Pre-laboratory Assignment 4, Intravenous Sugar Solutions, Making Basic Solutions. Name Kristin O'Neill (i) - Normal saline solutions are used to clean wounds, help remove contact lenses and by injection into a vein, treat dehydration. A Normal Saline Solution consist of 0.900 % (m/v) NaCl solution. Calculate the mass (in grams) of NaCl needed to create 1.50 L of Normal Saline Solution. = 1,350 g NaCl 1.50% (1000 m X 90 g 100 mx Below is a calibration curve which relates the % concentration of saline solution (m/v) with the density of the solution. Use this calibration curve and determine the % concentration of a saline solution which has a density of 1.08 g/mL. Show how you determined this concentration on the plot. Density (g/ml) 1.20 1.18 1.16 1.14 1.12 1.10 1.08 1.06 1.04 1.02 5 10 15 20 Precent Saline Solution (m/v) 30 (iii) - What is the density of a saline solution that was created by dissolving 15.48 g of NaCl in 86.0 mL of distilled water? (hint: Calculate % (m/v) and use the calibration curve above, Show all work). (iv) - It was determined that a 140 lb person needed 0.35 g NaCl / kg body weight. How many liters of Normal Saline Solution would need to be administered? (Normal Saline Solution = 0.900 % NaCl, 1 kg = 2.205 lb) 31
Expert Answer:
Answer rating: 100% (QA)
0900 mv Nall sol of Nall is present in Since In 100m... View the full answer

Related Book For 

Probability & Statistics for Engineers & Scientists
ISBN: 978-0130415295
7th Edition
Authors: Ronald E. Walpole, Raymond H. Myers, Sharon L. Myers, Keying
Posted Date:
Students also viewed these accounting questions
-
Calculate the mass in grams of the following. a. 0.15 mol Na b. 0.594 mol S c. 2.78 mol CH2Cl2 d. 38 mol (NH4)2S
-
Calculate the mass in grams of the following. a. 0.205 mol Fe b. 0.79 mol F c. 5.8 mol CO2 d. 48.1 mol K2CrO4
-
Calculate the mass in grams of iodine (I2) that will react completely with 20.4 g of aluminum (Al) to form aluminum iodide (AlI3).
-
1. What is the present value (PV") of an offer of $15,000 two years from now if the Opportunity Cost of Capital (OCC) is $12% per annum? 2. What is the PV of an offer to receive $12,000 three years...
-
Worksheet data for DeSousa Company are presented in E4-2. In E4-2, The adjusted trial balance columns of the worksheet for DeSousa Company are as follows. Instructions (a) Journalize the closing...
-
Many people who earn incomes below some level receive food stamps from the government. Economists argue that these people would be better off if the government gave them the cash equivalent of the...
-
Rods \(A B\) and \(A C\) have a diameter of \(20 \mathrm{~mm}\) and are made of 6061-T6 aluminum alloy. They are connected to the rigid collar \(A\) which slides freely along the vertical guide rod....
-
Rob's Shameless Self-Promotion Sales (RSS-PS) Inc. is a Canadian-controlled private corporation located in London, Ontario. For its fiscal year ended December 31, 2012, the corporation had correctly...
-
Graph the equation y 83 +4 by plotting points.
-
Determine the force in members GF, CF, and CD of the roof truss and indicate if the members are in tension or compression. 1.5 kN 1.70 m 2 kN 1.5 m 0,8 m G. -1 m- 2 m 2 m
-
X 21 Let f : (0, ) R and f : (0, ) R be defined by f(x) = f(t-j)' dt, x>0 and 0 =1 f(x) = 98(x - 1)50-600(x - 1)4 +2450, x > 0, where, for any positive integer n and real numbers a, a2, ...., ans;...
-
How do leaders cultivate resilience and emotional intelligence within their teams, equipping individuals with the psychological tools necessary to navigate ambiguity and embrace change as a catalyst...
-
Consider a Venn diagram, where P(E) = .10, P(E2) = .05, P(E3) = P(E4) = .2, P(Es) = .06, P(E) .3, P(E7) = .06, and P(Eg) = .03. E1 E3 A E7 E E4 Es B Find the following probabilities: a) P(A) b) P(B)...
-
How do change leaders effectively manage complexity and navigate the interplay of interconnected systems and subsystems within organizations to orchestrate holistic and enduring transformational...
-
Use supply & demand curves to explain what might happen in the market for bicycles if the price of gasoline increases. ps. (not gas powered bike)( make supply and demand curve to show what happens to...
-
How do change agents leverage data analytics and predictive modeling to anticipate potential barriers and proactively design interventions that facilitate smoother transitions and minimize disruption...
-
The merchandise inventory was destroyed by fire on December 13. The following data were obtained from the accounting records: Jan. 1 Merchandise inventory $350,000 Jan. 1-Dec. 31 Purchases (net)...
-
1. What is the semi-annually compounded interest rate if $200 accumulates to $318.77 in eight years? Answer in percentage with two decimal places. 2. What is the quarterly compounded interest rate if...
-
Repeat Sample Problem39-6 for the Balmer series of the hydrogen atom.
-
Referring to the random variables whose joint density function is given in Exercise 3.41, find the covariance between the weight of the creams and the weight of the toffees in these boxes of...
-
Suppose the diagram of an electrical system is given in Figure. What is the probability that the system works? Assume the components fail independently. 0.7 0.95 0.9 0.8
-
Develop a value stream map for the process you flowcharted in question 27 to identify the valueadded and non-value-added activities. How can you estimate costs and/or revenue for the process steps?...
-
Given the information in problem 36, what do you recommend if Worthington Machining can buy folded metal panels of identical size at a price of \($27.25\) per panel? Problem 36 Worthington Machining...
-
Bluegrass Irrigation Services (BIS) in Lexington, Kentucky, provides a wide variety of services to meet customer needs. These services include installation of lawn and tree irrigation equipment for...

Study smarter with the SolutionInn App


