Problem 1 (20 points) a. What is the mass in grams of a molecule of uranyl...
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
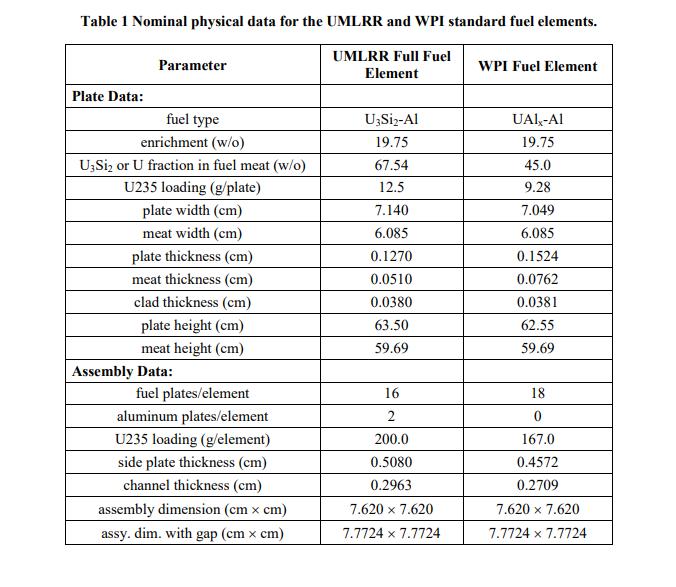
Problem 1 (20 points) a. What is the mass in grams of a molecule of uranyl sulfate, UO₂SO4? What is this mass in amu? b. Dry air at normal temperature and pressure has a density of about 0.0012 g/cm³ with a mass fraction of oxygen of 0.23 (i.e. 0.23 g of O per g of air). What is the atom density of 018 in dry air at nominal conditions? c. A sample of uranium is enriched to 3.20 atom percent (a/o) in U235 with the remainder being U238. What is the U235 enrichment in weight percent (w/o)? Note: Enrichment is usually given in w/o. However, this problem wants to highlight the difference between a/o and w/o. Also, for this problem, use the molecular weights to 5 significant figures (since the difference between a/o and w/o is small for this case). d. How many atoms of deuterium are in 2 kg of ordinary water? Problem 2 (10 points) Look up the material composition and physical density of stainless steel alloy 304 (i.e. SS304) and estimate the atom densities of its primary constituents. Problem 3 (20 points) During the summer of 2011, UMass-Lowell obtained several essentially fresh fuel assemblies from the recently decommissioned WPI reactor (the fuel was only very slightly used). The existing UMLRR fuel elements and the WPI fuel assemblies are quite similar in overall size and shape, but there are enough key differences so that a formal comparison and safety evaluation for their combined use is required. In particular, Table 1 highlights many of these similarities and differences. For example, both assemblies have 18 plates within the two side plates -- but the WPI element has 18 fuel plates, whereas the UMLRR element has 16 fuel plates and two aluminum end plates for a total of 18 plates. The fuel meat for the WPI plate is UAI,-Al with a U235 loading of about 9.3 g, whereas the UMLRR full fuel plate contains U3Si2-Al fuel with 12.5 g of U235 per plate. In addition, although the nominal cladding thicknesses are essentially identical, the WPI fuel meat is slightly thicker than the UMLRR fuel -- thus, the overall fuel plate thickness is a little larger, with a slightly decreased channel thickness for the WPI element. Before any physics analysis can be performed for the WPI assembly, one first must determine the region and assembly-averaged material compositions associated with this fuel element -- and this will be the goal of this HW Problem. In particular, based on the UMLRR example done in class, the general geometry layout given in the Lecture Notes, and the specific WPI design data highlighted in Table 1, your goal is to compute assembly-averaged densities for all five isotopes that make up the WPI fuel element (U235, U238, Al, H, and O). All the needed information for the fuel region is given in Table 1 and you can assume nominal densities for the pure aluminum and water regions (2.7 g/cm³ and 1.0 g/cm³, respectively, at room temperature). Note that the assembly basis in the axial direction should only include the active fuel region (i.e. the fuel meat height) -- and, since all the structures have the same height, the volume fraction calculations can really be done using area fractions (as done in the class examples). Note that this problem requires a number of computations -- so be sure to organize your work so that a reviewer can easily follow your assumptions/analyses. Also clearly tabulate appropriate intermediate results (such as the region densities and volume fractions), so that it is easy to see how the final assembly-homogenized densities are developed. Thus, at a minimum, you should have three summary tables of results -- one for the fuel meat, structure, and water region densities, one for the fuel meat, structure, and water volume fractions, and a final table of densities averaged over the entire fuel assembly. It is this final table that contains the desired homogenized densities for the WPI fuel assembly... Good luck -- and be sure to do your work neatly and to clearly explain your overall solution logic! Table 1 Nominal physical data for the UMLRR and WPI standard fuel elements. UMLRR Full Fuel Element Plate Data: Parameter fuel type enrichment (w/o) U3Si₂ or U fraction in fuel meat (w/o) U235 loading (g/plate) plate width (cm) meat width (cm) plate thickness (cm) meat thickness (cm) clad thickness (cm) plate height (cm) meat height (cm) Assembly Data: fuel plates/element aluminum plates/element U235 loading (g/element) side plate thickness (cm) channel thickness (cm) assembly dimension (cm x cm) assy. dim. with gap (cm x cm) U3Si₂-Al 19.75 67.54 12.5 7.140 6.085 0.1270 0.0510 0.0380 63.50 59.69 16 2 200.0 0.5080 0.2963 7.620 × 7.620 7.7724 x 7.7724 WPI Fuel Element UAIX-AI 19.75 45.0 9.28 7.049 6.085 0.1524 0.0762 0.0381 62.55 59.69 18 0 167.0 0.4572 0.2709 7.620 × 7.620 7.7724 x 7.7724 Problem 1 (20 points) a. What is the mass in grams of a molecule of uranyl sulfate, UO₂SO4? What is this mass in amu? b. Dry air at normal temperature and pressure has a density of about 0.0012 g/cm³ with a mass fraction of oxygen of 0.23 (i.e. 0.23 g of O per g of air). What is the atom density of 018 in dry air at nominal conditions? c. A sample of uranium is enriched to 3.20 atom percent (a/o) in U235 with the remainder being U238. What is the U235 enrichment in weight percent (w/o)? Note: Enrichment is usually given in w/o. However, this problem wants to highlight the difference between a/o and w/o. Also, for this problem, use the molecular weights to 5 significant figures (since the difference between a/o and w/o is small for this case). d. How many atoms of deuterium are in 2 kg of ordinary water? Problem 2 (10 points) Look up the material composition and physical density of stainless steel alloy 304 (i.e. SS304) and estimate the atom densities of its primary constituents. Problem 3 (20 points) During the summer of 2011, UMass-Lowell obtained several essentially fresh fuel assemblies from the recently decommissioned WPI reactor (the fuel was only very slightly used). The existing UMLRR fuel elements and the WPI fuel assemblies are quite similar in overall size and shape, but there are enough key differences so that a formal comparison and safety evaluation for their combined use is required. In particular, Table 1 highlights many of these similarities and differences. For example, both assemblies have 18 plates within the two side plates -- but the WPI element has 18 fuel plates, whereas the UMLRR element has 16 fuel plates and two aluminum end plates for a total of 18 plates. The fuel meat for the WPI plate is UAI,-Al with a U235 loading of about 9.3 g, whereas the UMLRR full fuel plate contains U3Si2-Al fuel with 12.5 g of U235 per plate. In addition, although the nominal cladding thicknesses are essentially identical, the WPI fuel meat is slightly thicker than the UMLRR fuel -- thus, the overall fuel plate thickness is a little larger, with a slightly decreased channel thickness for the WPI element. Before any physics analysis can be performed for the WPI assembly, one first must determine the region and assembly-averaged material compositions associated with this fuel element -- and this will be the goal of this HW Problem. In particular, based on the UMLRR example done in class, the general geometry layout given in the Lecture Notes, and the specific WPI design data highlighted in Table 1, your goal is to compute assembly-averaged densities for all five isotopes that make up the WPI fuel element (U235, U238, Al, H, and O). All the needed information for the fuel region is given in Table 1 and you can assume nominal densities for the pure aluminum and water regions (2.7 g/cm³ and 1.0 g/cm³, respectively, at room temperature). Note that the assembly basis in the axial direction should only include the active fuel region (i.e. the fuel meat height) -- and, since all the structures have the same height, the volume fraction calculations can really be done using area fractions (as done in the class examples). Note that this problem requires a number of computations -- so be sure to organize your work so that a reviewer can easily follow your assumptions/analyses. Also clearly tabulate appropriate intermediate results (such as the region densities and volume fractions), so that it is easy to see how the final assembly-homogenized densities are developed. Thus, at a minimum, you should have three summary tables of results -- one for the fuel meat, structure, and water region densities, one for the fuel meat, structure, and water volume fractions, and a final table of densities averaged over the entire fuel assembly. It is this final table that contains the desired homogenized densities for the WPI fuel assembly... Good luck -- and be sure to do your work neatly and to clearly explain your overall solution logic! Table 1 Nominal physical data for the UMLRR and WPI standard fuel elements. UMLRR Full Fuel Element Plate Data: Parameter fuel type enrichment (w/o) U3Si₂ or U fraction in fuel meat (w/o) U235 loading (g/plate) plate width (cm) meat width (cm) plate thickness (cm) meat thickness (cm) clad thickness (cm) plate height (cm) meat height (cm) Assembly Data: fuel plates/element aluminum plates/element U235 loading (g/element) side plate thickness (cm) channel thickness (cm) assembly dimension (cm x cm) assy. dim. with gap (cm x cm) U3Si₂-Al 19.75 67.54 12.5 7.140 6.085 0.1270 0.0510 0.0380 63.50 59.69 16 2 200.0 0.5080 0.2963 7.620 × 7.620 7.7724 x 7.7724 WPI Fuel Element UAIX-AI 19.75 45.0 9.28 7.049 6.085 0.1524 0.0762 0.0381 62.55 59.69 18 0 167.0 0.4572 0.2709 7.620 × 7.620 7.7724 x 7.7724
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the mass in grams of 1 molecule of H2O?
-
What is the mass in grams of 1 atom of Al?
-
A weather balloon filled with helium has a diameter of 3.00 ft. What is the mass in grams of the helium in the balloon at 21oC and normal pressure? The density of helium under these conditions is...
-
International Accounting Standard No. 21, "The Effects of Changes in Foreign Exchange Rates," deals with foreign currency. It was originally issued in 1983 and was revised and reissued in 1993 as...
-
The Yankee Fork and Hoe Company is a leading producer of garden tools ranging from wheelbarrows, mortar pans, and hand trucks to shovels, rakes, and trowels. The tools are sold in four different...
-
Show that R3 is the direct sum of the xy-plane with each of these. (a) The z-axis (b) The line |zE R}
-
What is critical natural capital? Provide examples. For a company, who determines whether natural capital is a critical ecological resource? Is there a difference between other natural capital and...
-
A small rural county has experienced unprecedented growth over the past 6 years, and as a result, the local school district built the new 500-student North Park Elementary School. The district has...
-
How do advanced construction techniques, such as prefabrication and modular construction, revolutionize the efficiency and safety of building projects, while also addressing challenges related to...
-
The file AirportTraffic contains the number of total passengers and the annual rate of change in passenger traffic for 50 airports. a. Construct a 95% confidence interval estimate for the population...
-
Scotty Scooters plans to sell a motorized standard scooter for $ 4 5 and a motorized chrome scooter for $ 6 5 . Scotty Scooters purchases the standard scooter for $ 3 5 and the chrome scooter for $ 4...
-
What methods are used in (a) the preliminary review and (b) the completion of the review.
-
What are the basic audit differences between an E.DP and a manual system?
-
Imagine that you were the salesperson. How would you have conducted the sales interview?
-
What would be your marketing communications and sales promotional strategies for the company in the USA? More specifically, outline your sales message and the type of media you would use to...
-
Identify and briefly describe the principal types of (a) processing controls and (b) output controls.
-
A spacecraft is moving to the east, and its nozzle is pointing to the west. If the spacecraft needs to slow down, then it should make a 180-degree roll maneuver before it fires its main engines ...
-
You have accepted the engagement of auditing the financial statements of the C. Reis Company, a small manufacturing firm that has been your auditee for several years. Because you were busy writing...
-
A buffer is made by dissolving 13.0 g of sodium dihydrogen phosphate, NaH2PO4, and 15.0 g of disodium hydrogen phosphate, Na2HPO4, in a liter of solution. What is the pH of the buffer?
-
Thorium-230, which occurs in uranium minerals, decays by alpha emission to radium. Write the nuclear equation for this decay process.
-
Associate each type of solid in the left-hand column with two of the properties in the right hand column. Each property may be used more than once. a. Molecular solid .. low-melting b. Ionic solid ....
-
Typically most large scale commercial developments require an environmental impact assessment to be undertaken as part of the planning process. While more a regulatory requirement, such an assessment...
-
Use the data presented in 12.13. The probability of the monthly contribution from the new product exceeding 13 500 is: (a) 24.5% (b) 30.5% (c) 63.0% (d) 92.5% 12.13: A company expects to sell 1000...
-
Most businesses will try to minimize risk through a combination of risk assessment, risk management and, where possible, transfer the risk to another party. For businesses, risks like theft, fire...

Study smarter with the SolutionInn App


