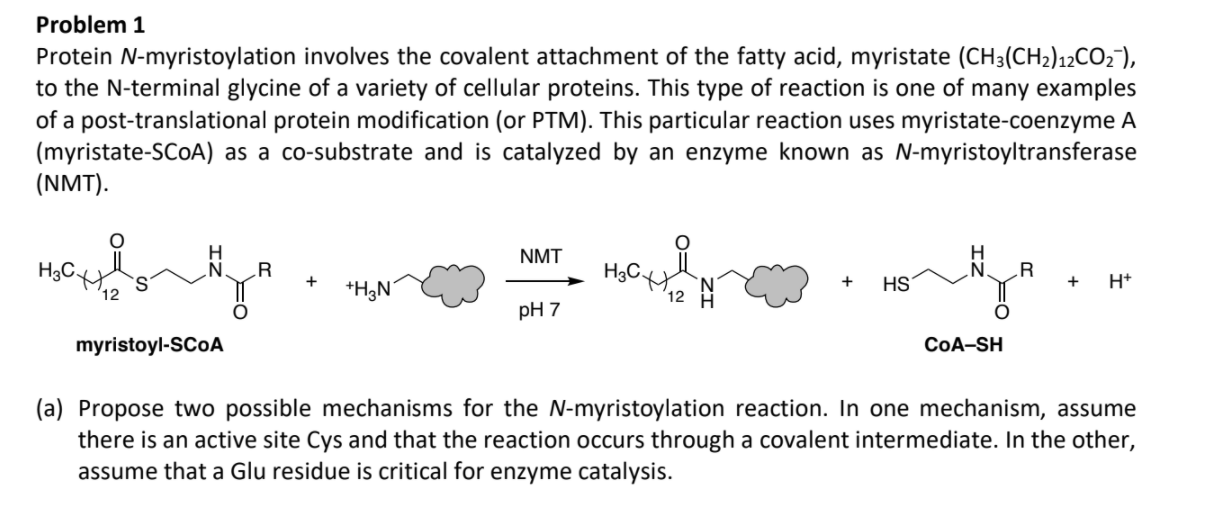
Problem 1 Protein N-myristoylation involves the covalent attachment of the fatty acid, myristate (CH3(CH)12CO), to the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
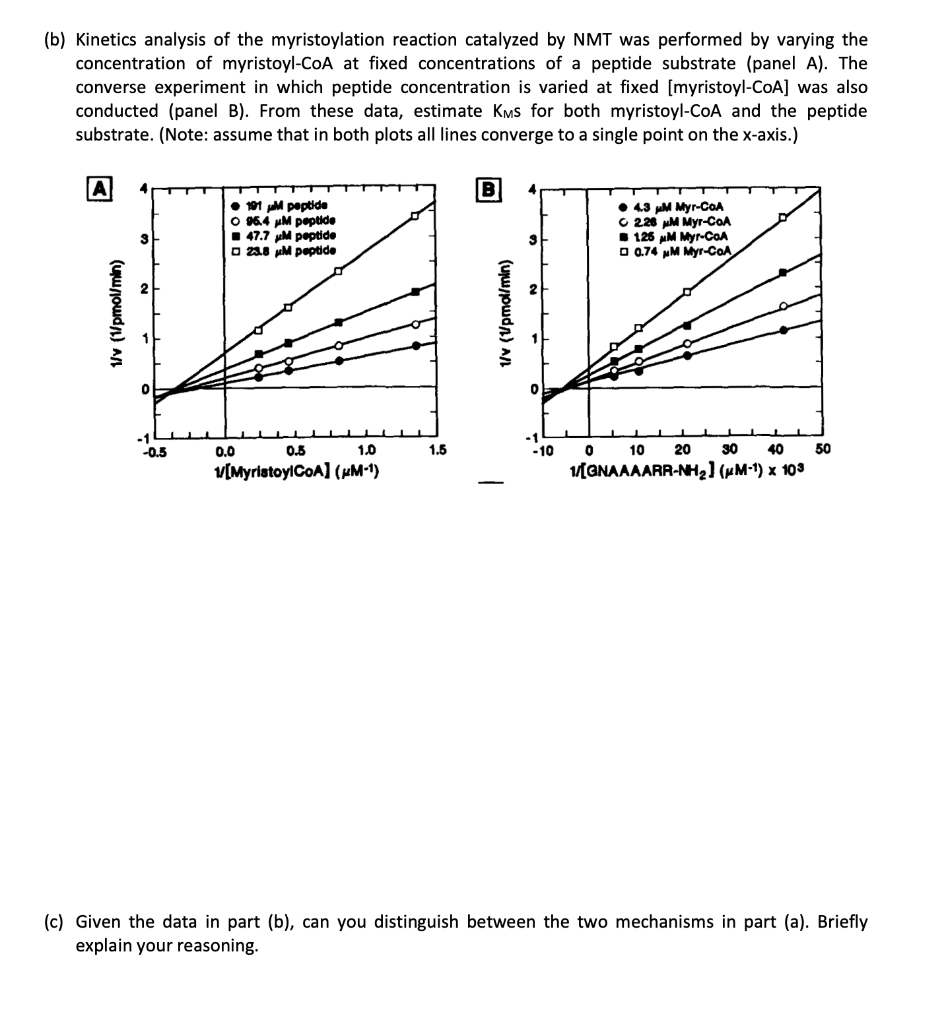
Problem 1 Protein N-myristoylation involves the covalent attachment of the fatty acid, myristate (CH3(CH₂)12CO₂¯), to the N-terminal glycine of a variety of cellular proteins. This type of reaction is one of many examples of a post-translational protein modification (or PTM). This particular reaction uses myristate-coenzyme A (myristate-SCOA) as a co-substrate and is catalyzed by an enzyme known as N-myristoyltransferase (NMT). ارسی یا H myristoyl-SCOA R +H3N NMT pH 7 요 12 N + HS H CoA-SH R + H+ (a) Propose two possible mechanisms for the N-myristoylation reaction. In one mechanism, assume there is an active site Cys and that the reaction occurs through a covalent intermediate. In the other, assume that a Glu residue is critical for enzyme catalysis. (b) Kinetics analysis of the myristoylation reaction catalyzed by NMT was performed by varying the concentration of myristoyl-CoA at fixed concentrations of a peptide substrate (panel A). The converse experiment in which peptide concentration is varied at fixed [myristoyl-CoA] was also conducted (panel B). From these data, estimate Kms for both myristoyl-CoA and the peptide substrate. (Note: assume that in both plots all lines converge to a single point on the x-axis.) A 1/v (1/pmol/min) -0.5 191 μM peptide O 96.4 μM peptide 47.7 μM peptide □23.8 μM peptide 0.5 1.0 1/[MyristoylCoA] (µM-1) 0.0 1.5 B 1/v (1/pmol/min) 0 ● 4.3 μM Myr-CoA 228 μM Myr-CoA 125 μM Myr-CoA 0.74 μM Myr-CoA -10 0 10 1/[GNAAAARR-NH₂) (μM-1) x 103 20 30 40 50 (c) Given the data in part (b), can you distinguish between the two mechanisms in part (a). Briefly explain your reasoning. Problem 1 Protein N-myristoylation involves the covalent attachment of the fatty acid, myristate (CH3(CH₂)12CO₂¯), to the N-terminal glycine of a variety of cellular proteins. This type of reaction is one of many examples of a post-translational protein modification (or PTM). This particular reaction uses myristate-coenzyme A (myristate-SCOA) as a co-substrate and is catalyzed by an enzyme known as N-myristoyltransferase (NMT). ارسی یا H myristoyl-SCOA R +H3N NMT pH 7 요 12 N + HS H CoA-SH R + H+ (a) Propose two possible mechanisms for the N-myristoylation reaction. In one mechanism, assume there is an active site Cys and that the reaction occurs through a covalent intermediate. In the other, assume that a Glu residue is critical for enzyme catalysis. (b) Kinetics analysis of the myristoylation reaction catalyzed by NMT was performed by varying the concentration of myristoyl-CoA at fixed concentrations of a peptide substrate (panel A). The converse experiment in which peptide concentration is varied at fixed [myristoyl-CoA] was also conducted (panel B). From these data, estimate Kms for both myristoyl-CoA and the peptide substrate. (Note: assume that in both plots all lines converge to a single point on the x-axis.) A 1/v (1/pmol/min) -0.5 191 μM peptide O 96.4 μM peptide 47.7 μM peptide □23.8 μM peptide 0.5 1.0 1/[MyristoylCoA] (µM-1) 0.0 1.5 B 1/v (1/pmol/min) 0 ● 4.3 μM Myr-CoA 228 μM Myr-CoA 125 μM Myr-CoA 0.74 μM Myr-CoA -10 0 10 1/[GNAAAARR-NH₂) (μM-1) x 103 20 30 40 50 (c) Given the data in part (b), can you distinguish between the two mechanisms in part (a). Briefly explain your reasoning.
Expert Answer:

Related Book For 

Fundamentals of biochemistry Life at the Molecular Level
ISBN: 978-0470547847
4th edition
Authors: Donald Voet, Judith G. Voet, Charlotte W. Pratt
Posted Date:
Students also viewed these chemistry questions
-
Propose two possible mechanisms for the following reaction. (Consider the possible sites of protonation in the molecule and the mechanistic consequences of each.) Devise an isotope-labeling...
-
Propose two possible mechanisms for the following reaction. (Consider the possible sites of protonation in the molecule and the mechanistic consequences of each.) Devise an isotope-labeling...
-
An enzyme contains an active site aspartic acid with a pKa = 5.0, which acts as a general acid catalyst. On the template below, draw the curve of enzyme activity (reaction rate) versus pH for the...
-
Marcus is the HR manager for United Airlines, an Illinois-based company. One of his employees has recently become disabled and is unable to fulfill the essential functions of his current position,...
-
The Shilling Company analyzed repair costs by month using linear regression analysis. The equation fit took the following form: The results were TRC = $20,000 $0.75x Average monthly repair costs have...
-
LuAnn Bean will receive $ 7,000 in seven years. Required: What is the present value at 7 percent compounded annually?
-
If Elizabeth could earn a 17 percent return on an investment in Hoffman Company stock (or some other company), what would you advise her to do? Explain your reasoning.
-
Reizenstein Technologies (RT) has just developed a solar panel capable of generating 200% more electricity than any solar panel currently on the market. As a result, RT is expected to experience a...
-
Select 2 companies in the same industry listed on the stock market - highlight which stock market. Analysis based on the latest 2 years annual report with financial statements. Income Statement,...
-
The Wilson Company's marketing manager has determined that the price elasticity of demand for its product equals - 2.2. According to studies she carried out, the relationship between the amount spent...
-
I am an auditor who has to test prepaid expenses. I need to find the list of procedures to perform for as required by the current audit. Where do I look for these procedures?
-
Simplify the expression. Enter the exact answer. 108x4+ 27x4
-
Solve the equation: 6x+7x= 3. There are two solutions to this equation. Enter the two solutions and round to two decimal places. Do not enter fractions.
-
ASSIGNMENT 1 - FILING OF CAPITAL GAINS TAX RETURN ON SALE OF UNLISTED SHARES OF STOCKS Instructions: 1. Download file of BIR Form 1707 Version 2021 at www.bir.gov.ph. 2. Input ALL the necessary...
-
Comparative financial statements for Weller Corporation, a merchandising company, for the year ending December 31 appear below. The company did not issue any new common stock during the year. A total...
-
Estrella Corporation purchased 5,000 outstanding shares (which had already been issued previously) from its shareholders and paid $250,000.00 for them. Some time after this transaction, he resold...
-
27 16 206 Required information The following information applies to the questions displayed below.) Cardinal Company is considering a five-year project requiring a $2,860,000 investment in equipment...
-
What is a content filter? Where is it placed in the network to gain the best result for the organization?
-
The subunit composition of an oligomeric protein can be determined by treating the protein with a cross-linking agent (a bifunctional molecule that reacts with and links groups in two different...
-
At pH 0, 1/2 O2 + 2H+ + 2e- H2 O................E = 1.23 V Is oxygen reduction more favored at pH 0 or at pH 7? Explain in electrochemical terms as well as in terms of chemical equilibria.
-
The last two columns of the table (labeled 'pH of environment' and '[A-]/[HA]') have been partially completed for you. The 'pH of environment' column lists a hypothetical pH at which the weak acid is...
-
Question: To ensure that its employees did not use illegal drugs in or outside of the workplace, Rain Co. required all employees to take a lie detector test. Moreover, managers began to screen the...
-
Question: Pippa became pregnant the week she started work as an administrator at Awesome University. Her supervisor was so annoyed at her that he would not consider her for promotion to another job...
-
Question: When Theodore Staats went to his company's "Council of Honor Con- vention," he was accompanied by a woman who was not his wife although he told everyone she was. The company fired him....

Study smarter with the SolutionInn App


