The surface of the sun has a temperature of about 5800K and consists largely of hydrogen...
Fantastic news! We've Found the answer you've been seeking!
Question:
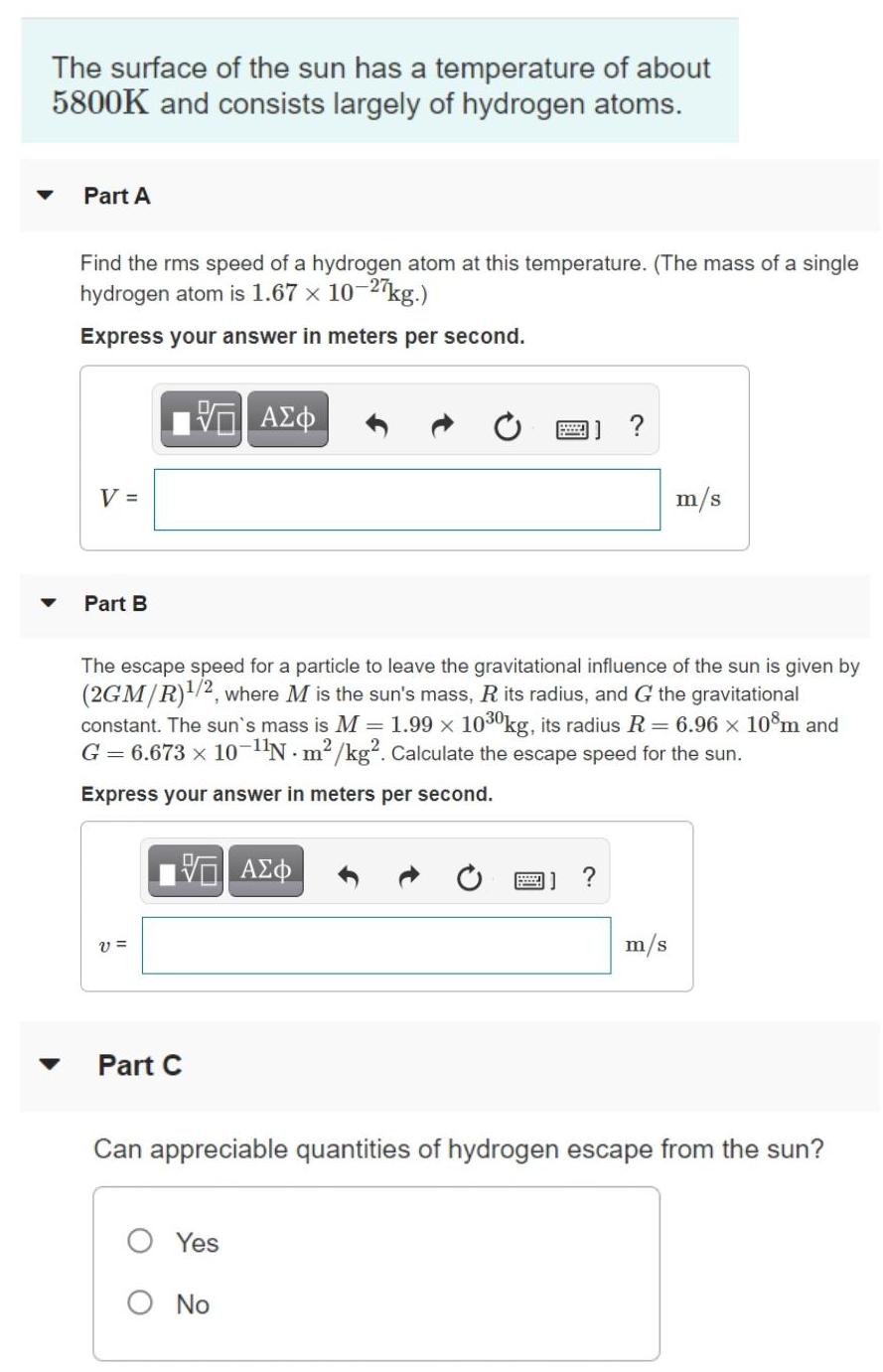
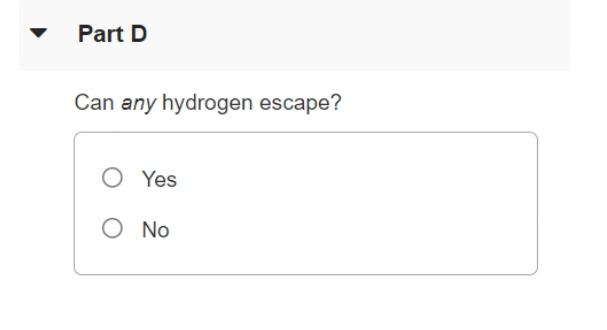
Transcribed Image Text:
The surface of the sun has a temperature of about 5800K and consists largely of hydrogen atoms. Part A Find the rms speed of a hydrogen atom at this temperature. (The mass of a single hydrogen atom is 1.67 x 10-2 kg.) Express your answer in meters per second. V = m/s Part B The escape speed for a particle to leave the gravitational influence of the sun is given by (2GM/R)/2, where M is the sun's mass, R its radius, and G the gravitational constant. The sun's mass is M = 1.99 x 1030kg, its radius R = 6.96 x 10m and G = 6.673 x 10-N m2 /kg². Calculate the escape speed for the sun. Express your answer in meters per second. A) ? v = m/s Part C Can appreciable quantities of hydrogen escape from the sun? O Yes O No Part D Can any hydrogen escape? O Yes O No The surface of the sun has a temperature of about 5800K and consists largely of hydrogen atoms. Part A Find the rms speed of a hydrogen atom at this temperature. (The mass of a single hydrogen atom is 1.67 x 10-2 kg.) Express your answer in meters per second. V = m/s Part B The escape speed for a particle to leave the gravitational influence of the sun is given by (2GM/R)/2, where M is the sun's mass, R its radius, and G the gravitational constant. The sun's mass is M = 1.99 x 1030kg, its radius R = 6.96 x 10m and G = 6.673 x 10-N m2 /kg². Calculate the escape speed for the sun. Express your answer in meters per second. A) ? v = m/s Part C Can appreciable quantities of hydrogen escape from the sun? O Yes O No Part D Can any hydrogen escape? O Yes O No
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
The surface of the Sun has a temperature of about 5 800 K. The radius of the Sun is 6.96 X 108 m. Calculate the total energy radiated by the Sun each second. Assume that the emissivity of the Sun is...
-
The surface of the Sun has a temperature of 5500 C and the temperature of deep space is 3.0 K. (a) Find the entropy increase produced by the Sun in one day, given that it radiates heat at the rate of...
-
The surface of the Sun has a temperature of about 6000 K. What is the speed of a typical hydrogen molecule in the Suns atmosphere?
-
Assume that Jimmy Cash has $2000 in his checking account at Folsom Bank and uses his checking account card to withdraw $200 of cash from the banks ATM machine. By what dollar amount did the M1 money...
-
In one-way ANOVA, identify the statistic used a. As a measure of variation among the sample means. b. As a measure of variation within the samples. c. To compare the variation among the sample means...
-
Companies often try to keep accounting earnings growing at a relatively steady pace, thereby avoiding large swings in. earnings from period to period. They also try to me earnings targets. To do so...
-
Accountants for Compass Software have developed the following data from the companv's accounting records for the vear ended April 30, 2005: a. Purchase of plant assets, \($59,400 b. \) Cash receipt...
-
The adjusted trial balances of Rosa Corporation at August 31, 2014, and August 31, 2013, include these amounts (in millions): Rosa completed these transactions (in millions) during the year ended...
-
! 13 Required information [The following information applies to the questions displayed below.] Mack Precision Tool and Die has two production departments, Fabricating and Finishing, and two service...
-
HTLM5 AND CSS View as TextDownload / 1 Exam 4: Video and Animation 1. Add video a1.mp4 to the page, size is 300px by 300px, letthe video play automatically, rewind to play automatically (15 points)...
-
Let L be the smallest digit appearing in your UID and let R be the largest digit appearing in your UID. The function f(x) = (x-L)(R-x) opens down and crosses the x-axis at x = L and x = R. Use int to...
-
Instructions Collegiate Publishing Inc. began printing operations on March 1. Jobs 301 and 302 were completed during the month, and all costs applicable to them were recorded on the related cost...
-
Thesis Statement: People have praised Facebook for being innovative and having a positive effect on society. However, the company's internal corporate culture has been called into question, and...
-
The bookkeeper at Martin Company has asked you to prepare a bank reconciliation as of May 31. The May 31 bank statement and the May T-account for cash (summarized) are below. Martin Company's bank...
-
Stackit Ltd. manufactures three sizes of wooden bookcases. The information relating to the production of the three bookcases for next year is as follows: Selling Price Cost of wood Direct labour...
-
3. Consider the following production function (Isoquant) IC= Q2 LK and values w=P =$15, r=Px =$8 LABOUR L e) Find the slope of the Isocost curve below. f) Set up a Cost Minimization problem and find...
-
Instructions Before making any last decisions about the new campaign, Costa's Customs would like you to look over their last attempt at digital marketing. It was a complete failure, and Costa's...
-
In Problems 718, write the augmented matrix of the given system of equations. f0.01x0.03y = 0.06 [0.13x + 0.10y = 0.20
-
Suppose a neuron in the brain carries a current of 5.0 10-8 A. Treating the neuron as a straight wire, what is the magnetic field it produces at a distance of 7.5 cm? A. 1.3 10-13 T B. 4.2 10-12 T...
-
Suppose you pour water into a container until it reaches a depth of 14 cm. Next, you carefully pour in a 7.5-cm thickness of olive oil so that it floats on top of the water. What is the pressure at...
-
Figure 28-49 shows a single-slit diffraction pattern formed by light passing through a slit of width W = 11.2 mm and illuminating a screen 0.855 m behind the slit. (a) What is the wavelength of the...
-
List the modifications of the standard audit report that normally do not result in a qualification, a disclaimer, or an adverse opinion.
-
When more than one auditor is involved in an audit of a company's financial statements, what two decisions about reporting must the principal auditor make?
-
What disclosure is made in the principal auditors' report if they decide to assume responsibility for other auditors' work? If they decide not to assume responsibility for other auditors' work?

Study smarter with the SolutionInn App


