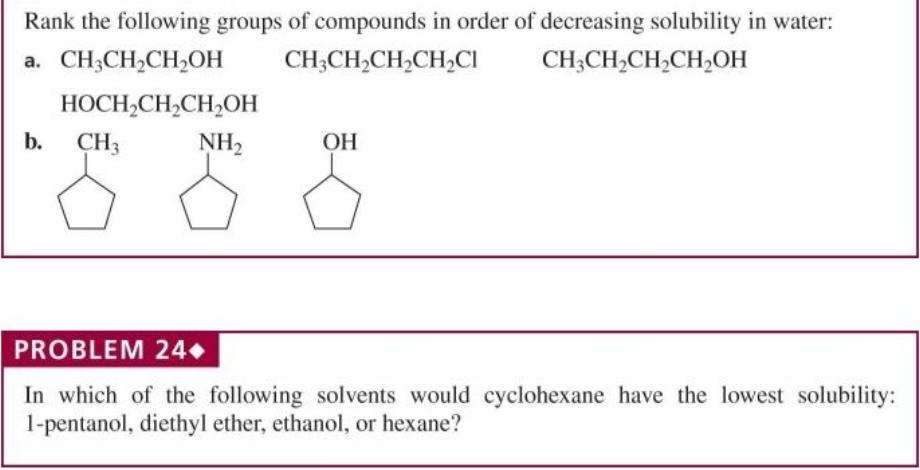
Rank the following groups of compounds in order of decreasing solubility in water: a. CH;CH2CH,OH CH;CH,CH,CH,CI...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
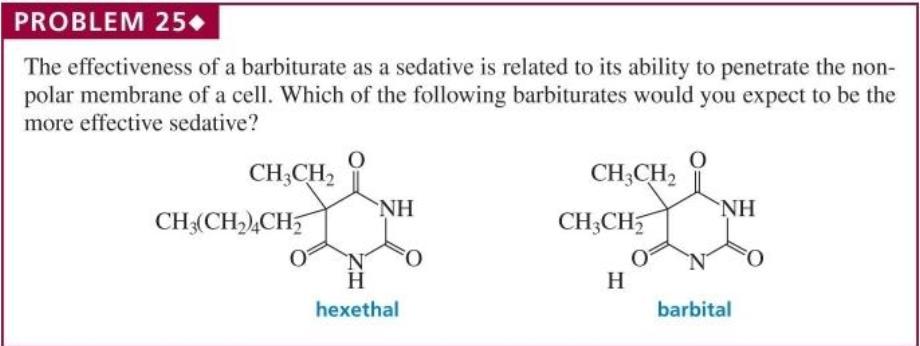
Rank the following groups of compounds in order of decreasing solubility in water: a. CH;CH2CH,OH CH;CH,CH,CH,CI CH;CH,CH,CH,OH HOCH,CH,CH,OH b. CH3 NH2 ОН PROBLEM 24 In which of the following solvents would cyclohexane have the lowest solubility: 1-pentanol, diethyl ether, ethanol, or hexane? PROBLEM 25+ The effectiveness of a barbiturate as a sedative is related to its ability to penetrate the non- polar membrane of a cell. Which of the following barbiturates would you expect to be the more effective sedative? CH;CH2 CH;CH, NH NH CH3(CH2),CH5 CH3CH5 O. H H hexethal barbital Rank the following groups of compounds in order of decreasing solubility in water: a. CH;CH2CH,OH CH;CH,CH,CH,CI CH;CH,CH,CH,OH HOCH,CH,CH,OH b. CH3 NH2 ОН PROBLEM 24 In which of the following solvents would cyclohexane have the lowest solubility: 1-pentanol, diethyl ether, ethanol, or hexane? PROBLEM 25+ The effectiveness of a barbiturate as a sedative is related to its ability to penetrate the non- polar membrane of a cell. Which of the following barbiturates would you expect to be the more effective sedative? CH;CH2 CH;CH, NH NH CH3(CH2),CH5 CH3CH5 O. H H hexethal barbital
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Rank the following groups of compounds in order of decreasing solubility in water: a. CH3CH3CH3OH CH3CH3CH3CH3Cl CH3CH3CH3CH3OH HOCH3CH3CH3OH b. CH NH2 OH
-
The effectiveness of a barbiturate as a sedative is related to its ability to penetrate the nonpolar membrane of a cell. Which of the following barbiturates would you expect to be the more effective...
-
Arrange the following groups of compounds in order of decreasing reactivity toward electrophilic aromatic substitution: a. Benzene, ethylbenzene, chlorobenzene, nitrobenzene, anisole b....
-
You make a one-time investment of $400 and leave it for 5 years, earning an annual interest rate of 7%. How much interest will you have earned after 5 years? If you leave the entire amount in your...
-
Use an elasticity concept to explain each of the following observations. a. During economic booms, the number of new personal care businesses, such as gyms and tanning salons, is proportionately...
-
Calculate financial leverage measures. The following information was available for the year ended December 31, 2013: Earnings before interest and taxes (operating income) ....... $60,000 Interest...
-
Consider the calculation of an external rate of return (ERR). The positive cash flows in the cash flow profile are moved forward to \(t=n\) using what value of \(i\) in the \((F \mid P i, n-t)\)...
-
Health Care Systems of Florida (HCSF) is planning to build a number of new emergency-care clinics in central Florida. HCSF management has divided a map of the area into seven regions. They want to...
-
The force exerted by a 2.4-m massless string on a 0.84-kg object being swung in a horizontal circle is 4.2 N. What is the tangential velocity of the object?
-
Bijou, a member, is preparing a personal tax return for Paloma.There are a number of positions being taken on the return thatwould be considered controversial matters. Bijou has been preparingreturns...
-
1. Project Title / Company Name Anti-Forced Sex Nail Polish / InvisiNail Australia 2. Project background Reducing sexual violence in our community is our main concern and we must put a stop to it....
-
Subway now requires all of its franchisees to submit their weekly sales and inventory reports electronically using new point-of-sale (POS) touch-screen cash registers. With the new POS registers,...
-
Describe an employee self-service system.
-
Go to the 2010 annual report for Kelloggs Company at http://investor.kelloggs.com/annuals.cfm and find the Consolidated Balance Sheet. Calculate for Kelloggs the current ratio for 2009 and 2010.
-
What are electronic bill presentment and payment (EBPP) systems? How do they reduce costs and accelerate cash flows?
-
What is the bullwhip effect?
-
What register does the "jal" instruction automatically update? $a0 $sp Sra Sat $s0 Sto SVO
-
Express these numbers in standard notation. a. 2.87 10-8 b. 1.78 1011 c. 1.381 10-23
-
Any base whose conjugate acid has a greater than _________ can remove a proton from a terminal alkyne to form an acetylide ion (in a reaction that favors products)
-
Arginine and lysine side chains fit into trypsin's binding pocket. One of these side chains forms a direct hydrogen bond with serine and an indirect hydrogen bond (mediated through a water molecule)...
-
Propose a mechanism for each of the following reactions: (a) (b) CH3 CH2CH2CHCH CH2 CH2CH3 H3C CH3 CH CH2 Ht
-
Answer the following questions: 1. Name at least five industries that might be affected by weather. 2. Is it appropriate for the forensic accountant/fraud examiner to examine the effect of weather?...
-
Discuss why collusive frauds, especially when perpetrated among related parties, is particularly difficult to detect?
-
In what year was the original company formed?

Study smarter with the SolutionInn App



