Q : Calculate the standard cell potentials of galvanic cells in which the following reactions take place:
Fantastic news! We've Found the answer you've been seeking!
Question:
Q :
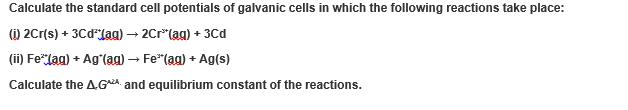
Transcribed Image Text:
Calculate the standard cell potentials of galvanic cells in which the following reactions take place: (i) 2Cr(s) + 3CdJag) → 2Cr(ag) + 3Cd (ii) Fe* (ag) + Ag (ag) → Fe*(ag) + Ag(s) Calculate the AGA and equilibrium constant of the reactions. Calculate the standard cell potentials of galvanic cells in which the following reactions take place: (i) 2Cr(s) + 3CdJag) → 2Cr(ag) + 3Cd (ii) Fe* (ag) + Ag (ag) → Fe*(ag) + Ag(s) Calculate the AGA and equilibrium constant of the reactions.
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
P Ltd has two divisions, Q and R, that operate as profit centres. Division Q has recently been set up to provide a component (Comp1) which division R uses to produce its product (ProdX). Prior to...
-
1. The following relations describe demand and supply. Q = 700 - 100P (1) Q = -100 + 100P (2) Where P is price in dollar and Q is quantity in unit. a. Which equation (1) or (2), is demand equation...
-
Let Q be a set of n points in the plane. We say that point (x, y) dominates point (x?, y?) if x ? x? and y ? y?. A point in Q that is dominated by no other points in Q is said to be maximal. That Q...
-
An undamped mass spring system is released from rest from an initial displacement of x = 0.24 m and starts to oscillate. You see that the mass reaches its largest positive displacement for the first...
-
New lithographic equipment, acquired at a cost of $100,000 at the beginning of a fiscal year, has an estimated useful life of 5 years and an estimated residual value of $8,000. The manager requested...
-
Find the area A of a triangle with height 9 centimeters and base 4 centimeters.
-
If there are 20 pairs (x, y) in a data set, then the number of degrees of freedom for the critical value is _______________ . In Exercises 7 and 8, fill in each blank with the appropriate word or...
-
The Ace Steel Mill estimates the demand for steel in millions of tons per year as follows: Millions of Tons Probability 10 ..................10 12 ..................25 14 ..................30 16...
-
Topper Sports, Incorporated, produces high-quality sports equipment. The company's Racket Division manufactures three tennis rackets-Standard, Deluxe, and Pro-widely used in amateur play. Selected...
-
Continuing Payroll Project: Prevosti Farms and Sugarhouse - EERF (Static) Prevosti Farms and Sugarhouse pays its employees according to their job classification. The following employees make up...
-
A room is 15 m long and 12 m broad. Find the cost of covering the room with a carpet, which is a square of length 2m,at the rate of rs 50 per square metre.
-
A 0.2 kg tennis ball is traveling toward a racket with a velocity of 59 m/sec. It hits the racket and travels in the opposite direction at 65 m/sec. a. What is the change in momentum of the tennis...
-
On December 31, 2020, Headland Company has $7,024,000 of short-term debt in the form of notes payable to Gotham State Bank due in 2021. On December 28, 2020, Headland enters into a refinancing...
-
Ferry Chemical uses a standard cost system to account for the costs of its production of Chemical X . Standards are 2 . 9 gallons of materials at $ 1 2 0 per gallon and 2 5 hours of labor at a...
-
what ways can leaders cultivate their own resilience and well-being to effectively lead through adversity and uncertainty, serving as role models for their teams and promoting a culture of holistic...
-
Tony is a business owner that is under the impression that people will buy more goods if aggressive promotional techniques are used with potential buyers. Which of the four marketing management...
-
5 ints eBook Print References The following financial statement information is from five separate companies. Beginning of year Assets Liabilities End of year Assets Liabilities Changes during the...
-
At the beginning of the year, Lam Ltd. had total assets of $800,000 and total liabilities of $500,000. Use this information to answer each of the following independent questions. (a) If Lam's total...
-
(a) Define the terms limiting reactant and excess reactant. (b) Why are the amounts of products formed in a reaction determined only by the amount of the limiting reactant? (c) Why should you base...
-
The iodine bromide molecule, IBr, is an inter-halogen compound. Assume that the molecular orbitals of IBr are analogous to the homo nuclear diatomic molecule F2. (a) Which valence atomic orbitals of...
-
The figure shows the three lowest regions of Earth's atmosphere. (a) Name each and indicate the approximate elevations at which the boundaries occur. (b) In which region is ozone a pollutant? In...
-
The major reason that the difference and ratio estimation methods would be expected to produce audit efficiency compared to mean estimation is that the a. Number of members of the populations of...
-
Probability proportional to size sampling (PPS) is normally used when it is thought that the population contains a a. Few understatements. b. Large number of understatements. c. Few overstatements....
-
Vale has decided to use PPS sampling in the audit of a client's accounts receivable balances. Vale plans to use the following PPS sampling table: What sample size should Vale use? a. 120 b. 108 c. 60...

Study smarter with the SolutionInn App



