Q.15 A complex is comlposed of one chromium, three bromides and six water molecules. Upon addition...
Fantastic news! We've Found the answer you've been seeking!
Question:
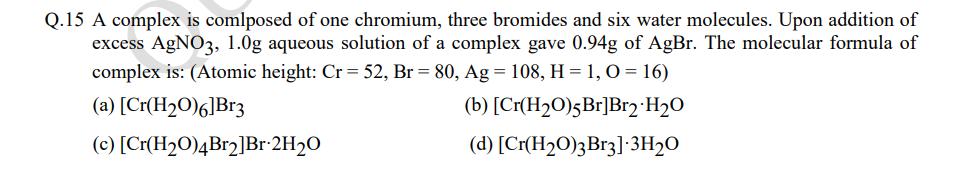
Transcribed Image Text:
Q.15 A complex is comlposed of one chromium, three bromides and six water molecules. Upon addition of excess AgNO3, 1.0g aqueous solution of a complex gave 0.94g of AgBr. The molecular formula of complex is: (Atomic height: Cr = 52, Br = 80, Ag = 108, H = 1, 0 = 16) (a) [Cr(H₂0)6]Br3 (b) [Cr(H₂O)5 Br]Br2 H₂O (c) (d) [Cr(H₂O)3 Br3].3H₂O [Cr(H₂O)4Br2]Br-2H₂O Q.15 A complex is comlposed of one chromium, three bromides and six water molecules. Upon addition of excess AgNO3, 1.0g aqueous solution of a complex gave 0.94g of AgBr. The molecular formula of complex is: (Atomic height: Cr = 52, Br = 80, Ag = 108, H = 1, 0 = 16) (a) [Cr(H₂0)6]Br3 (b) [Cr(H₂O)5 Br]Br2 H₂O (c) (d) [Cr(H₂O)3 Br3].3H₂O [Cr(H₂O)4Br2]Br-2H₂O
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A 50.00-mL solution containing NaBr was treated with excess AgNO3 to precipitate 0.214 6 g of AgBr (FM 187.772). What was the molarity of NaBr in the solution?
-
The following test tubes each contain a different chromium complex ion. For each compound, predict the predominant color of light absorbed. If the complex ions are Cr(NH3)63+, Cr(H2O)63+, and...
-
A hollow box beam with height h = 16 in., width b = 8 in., and constant wall thickness t = 0.75 in. is shown in the figure. The beam is constructed of steel with yield stress ÏY = 32 ksi....
-
Why are lipids and proteins free to move laterally in membranes? O a. Lipids and proteins repulse each other in the membrane O b. The interior of the membrane is filled with liquid water Oc. There...
-
The local supermarket buys lettuce each day to ensure really fresh produce. Each morning any lettuce that is left from the previous day is sold to a dealer that resells it to farmers who use it to...
-
Sublime Sandals Company was formed on January 1, 2014, and is preparing its annual financial statements dated December 31, 2014. Ending inventory information about the four major items stocked for...
-
A 20-cm-circumference loop of wire has a resistance of \(0.12 \Omega\). The loop is placed between the poles of an electromagnet, and a field of \(0.55 \mathrm{~T}\) is switched on in a time of \(15...
-
Job costing, accounting for manufacturing overhead, budgeted rates. The Solomon Company uses a job-costing system at its Dover, Delaware, plant. The plant has a Machining Department and a Finishing...
-
Using a failed project you are aware of, at County/ National level or in an organization you are familiar with, explain the importance of proper project planning. Discuss the importance of good time...
-
In this mini-case, you will complete the test of details on accounts receivable for the 2019 audit of EarthWear Clothiers, Inc. The principal test of detail involves sending "confirmations" or...
-
For a graph G, the chromatic polynomial P(G, k) counts the number of proper vertex colorings of G with k colors. In other words, P(G, k) is the number of ways to color the vertices of G using k...
-
What is the relationship/connection between Liberalism and Capitalism? How they are related ?
-
The effect of the new vouchers being offered by the state of Florida, will greatly impact the market of private schools. Considering the pre-voucher price was at equilibrium with both supply and...
-
An apartment building was purchased for $1,000,000 and was sold 5 years later at $1,200,000. During its holding period it produced 100,000 of profit each year. What is the HPR (before tax)?
-
1) Housing Prices in Melbourne discuss some of the welfare implications (think of consumer and producer surplus) of changes in housing prices.
-
3. Find a formula A containing three atoms p, q, and r, with the following property: for every assignment v: {p,q,r}{T, F} changing any of the values of v(p), v(q), v(r) will also change v(A).
-
You have two credit cards. Card Name (APR %) Credit Limit $1,500.00 Murk (5.6%) Mini (9.85%) $902.43 $1,000.00 What is your debt ratio? If you budget $375 to payoff your credit card debt and you...
-
Four GWU students have been selected to taste food sold by 3 different food trucks labeled as food truck A, B and C on H & 22nd Streets every Monday for 3-weeks. For each student, food trucks are...
-
a. Calculate K1 at 25C for phosphoric acid: b. Which thermodynamic factor is the most significant in accounting for the fact that phosphoric acid is a weak acid? Why? H3PO4(aq)--H"(aq) + H,PO4-(aq)...
-
The heat of vaporization of carbon disulfide, CS2, at 25oC is 27.2 kJ/mol. What is the entropy change when 1.00 mol of vapor in equilibrium with liquid condenses to liquid at 25oC? The entropy of...
-
Briefly explain how a mass spectrometer works. What kinds of information does one obtain from the instrument?
-
Sketch the \(P-V\) phase diagram for helium-4 using the sketch of the \(P-T\) phase diagram in Figure 4.3. Ps P S superfluid Pe T To T FIGURE 4.3 Sketch of the P-T phase diagram for helium-4. The...
-
(a) Assuming that the total number of microstates accessible to a given statistical system is \(\Omega\), show that the entropy of the system, as given by equation (3.3.13), is maximum when all...
-
What trends are occurring in outpatient and long-term care?

Study smarter with the SolutionInn App



