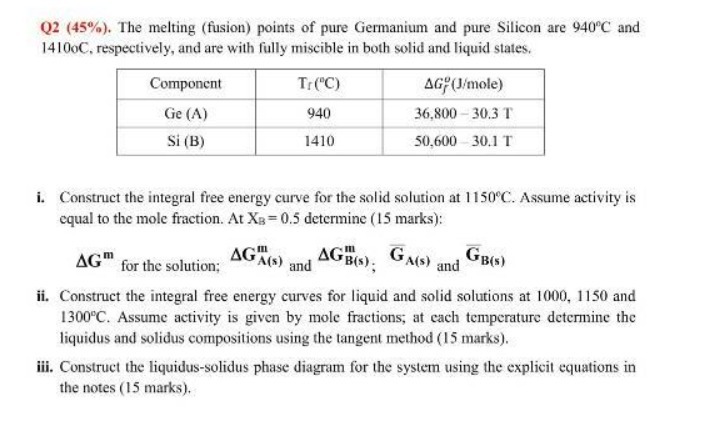
Q2 (45%). The melting (fusion) points of pure Germanium and pure Silicon are 940C and 14100C,...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Q2 (45%). The melting (fusion) points of pure Germanium and pure Silicon are 940C and 14100C, respectively, and are with fully miscible in both solid and liquid states. Component Ge (A) Si (B) Tr (C) 940 1410 AG (J/mole) 36,800 30.3 T 50,600 30.1 T i. Construct the integral free energy curve for the solid solution at 1150C. Assume activity is equal to the mole fraction. At XB=0.5 determine (15 marks): AGA(s) and AGB(s); GA(S) GB(s) and AG for the solution; ii. Construct the integral free energy curves for liquid and solid solutions at 1000, 1150 and 1300C. Assume activity is given by mole fractions; at each temperature determine the liquidus and solidus compositions using the tangent method (15 marks). iii. Construct the liquidus-solidus phase diagram for the system using the explicit equations in the notes (15 marks). Q2 (45%). The melting (fusion) points of pure Germanium and pure Silicon are 940C and 14100C, respectively, and are with fully miscible in both solid and liquid states. Component Ge (A) Si (B) Tr (C) 940 1410 AG (J/mole) 36,800 30.3 T 50,600 30.1 T i. Construct the integral free energy curve for the solid solution at 1150C. Assume activity is equal to the mole fraction. At XB=0.5 determine (15 marks): AGA(s) and AGB(s); GA(S) GB(s) and AG for the solution; ii. Construct the integral free energy curves for liquid and solid solutions at 1000, 1150 and 1300C. Assume activity is given by mole fractions; at each temperature determine the liquidus and solidus compositions using the tangent method (15 marks). iii. Construct the liquidus-solidus phase diagram for the system using the explicit equations in the notes (15 marks). Q2 (45%). The melting (fusion) points of pure Germanium and pure Silicon are 940C and 14100C, respectively, and are with fully miscible in both solid and liquid states. Component Ge (A) Si (B) Tr (C) 940 1410 AG (J/mole) 36,800 30.3 T 50,600 30.1 T i. Construct the integral free energy curve for the solid solution at 1150C. Assume activity is equal to the mole fraction. At XB=0.5 determine (15 marks): AGA(s) and AGB(s); GA(S) GB(s) and AG for the solution; ii. Construct the integral free energy curves for liquid and solid solutions at 1000, 1150 and 1300C. Assume activity is given by mole fractions; at each temperature determine the liquidus and solidus compositions using the tangent method (15 marks). iii. Construct the liquidus-solidus phase diagram for the system using the explicit equations in the notes (15 marks).
Expert Answer:
Answer rating: 100% (QA)
i To construct the integral free energy curve for the solid solution at 1150C we need to calculate the free energy values for both components Ge and Si at that temperature Lets start with component A ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these finance questions
-
Germanium forms a substitutional solid solution with silicon. Compute the number of germanium atoms per cubic centimeter for a germanium-silicon alloy that contains 15 wt% Ge and 85 wt% Si. The...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Fox Erasing has a system of internal control with the following procedures. Match the procedure to the corresponding internal control principle. Procedure Internal Control Principle A. Establish...
-
For the year ended December 31, 2017, Jet Set Airlines reported profit of $920,000 and a gain on an equity investment of $66,000, before income tax. This gain is other comprehensive income. Jet Set...
-
1. What features of the 7-Eleven Japan distribution system illustrate the concept of the bidirectional service supply relationship? 2. Does the 7-Eleven Japan distribution system exhibit scalability...
-
In the photoelectric effect experiment, as illustrated by Figure Q28.6, a current is measured while light is shining on the cathode. But this does not appear to be a complete circuit, so how can...
-
Labor data for making one gallon of finished product in Tang Company are as follows: (1) Pricehourly wage rate $13.00, payroll taxes $0.80, and fringe benefits $1.20. (2) Quantityactual production...
-
Darren applies a mark-up on cost of 40% to his purchases. (a) How much will his gross profit be if Darren's sales for the year are 98,000? (b) What would the cost of sales be if the mark-up is 25%?
-
a) Calculate the net asset value of a G share. b) Calculate the necessary compensation payment for a submitted D share if the intrinsic values serve as exchange rates and the exchange ratio is 3 : 1....
-
Epstein's creditors are threatening to garnish his bank account. Epstein, who is insolvent, makes a gift of $20,000 to the Jewish Community Federation of Richmond. Within two years, Epstein files for...
-
Describe the features of an asset and give two examples of assets typically recorded in a company's balance sheet.
-
Explore the principles of deadlock detection and avoidance in operating systems, discussing the various deadlock resolution strategies, such as resource preemption, process rollback, and deadlock...
-
Emerald City Stained Glass Company is creating their Selling and Administrative Expense Budget for next year. They know the following information: Budgeted sales are expected to be 10,000 units,...
-
In these scenarios, you are a community correctional administrator tasked with making policy changes, changes in the way things are done and, if need be, personnel adjustments. You also have control...
-
Find the commutators of the operator's Lx and Ly; Ly and Lz, also operators of Lz and Lx. In order to study the dynamical quantity angular momentum in quantum mech- anics, we construct the associated...
-
The electric field due to a line charge is given by where l is a constant. Show that E is solenoidal. Show that it is also conservative. E =
-
Mercury(I) oxide decomposes into elemental mercury and elemental oxygen: . (a)Write the equilibrium-constant expression for this reaction in terms of partial pressures. (b) Suppose you run this...
-
It has been suggested that strontium-90 (generated by nuclear testing) deposited in the hot desert will undergo radioactive decay more rapidly because it will be exposed to much higher average...
-
Decay of which nucleus will lead to the following products: (a) Bismuth-211 by beta decay (b) Chromium-50 by positron emission (c) Tantalum-179 by electron capture (d) Radium- 226 by alpha decay?
-
A change in chromosome structure that does not involve a change in the total amount of genetic material is a. a deletion. b. a duplication. c. an inversion. d. none of the above.
-
The term endopolyploidy refers to the phenomenon of having a. too many chromosomes. b. extra chromosomes inside the cell nucleus. c. extra sets of chromosomes in certain cells of the body. d. extra...
-
During transduction involving a P1 phage, a. any small fragment of the bacterial chromosome may be transferred to another bacterium by a new phage. b. only a specific fragment of DNA may be...

Study smarter with the SolutionInn App


