Q3: (20 points) The first three diffraction angles of the XRD pattem are observed using Cu...
Fantastic news! We've Found the answer you've been seeking!
Question:
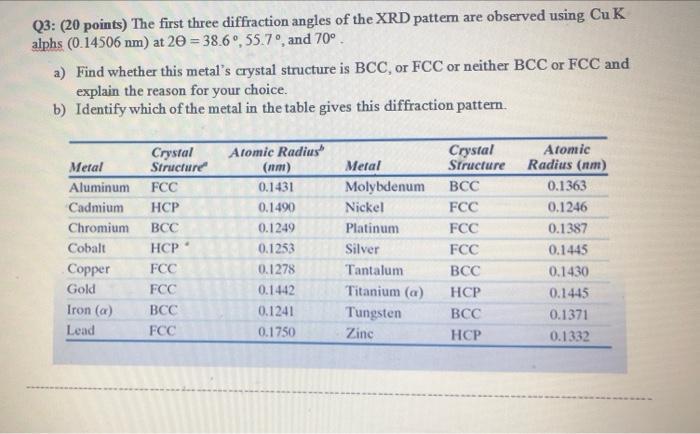
Transcribed Image Text:
Q3: (20 points) The first three diffraction angles of the XRD pattem are observed using Cu K alphs (0.14506 nm) at 20 = 38.6°, 55.7°, and 70°. a) Find whether this metal's crystal structure is BCC, or FCC or neither BCC or FCC and explain the reason for your choice. b) Identify which of the metal in the table gives this diffraction pattern. Crystal Structure Metal Aluminum FCC Cadmium HCP Chromium BCC HCP FCC FCC Cobalt Copper Gold Iron (a) Lead BCC FCC Atomic Radius (nm) 0.1431 0.1490 0.1249 0.1253 0.1278 0.1442 0.1241 0.1750 Metal Molybdenum Nickel Platinum Silver Tantalum Titanium (a) Tungsten Zinc Crystal Structure BCC FCC FCC FCC BCC HCP BCC HCP Atomic Radius (nm) 0.1363 0.1246 0.1387 0.1445 0.1430 0.1445 0.1371 0.1332 Q3: (20 points) The first three diffraction angles of the XRD pattem are observed using Cu K alphs (0.14506 nm) at 20 = 38.6°, 55.7°, and 70°. a) Find whether this metal's crystal structure is BCC, or FCC or neither BCC or FCC and explain the reason for your choice. b) Identify which of the metal in the table gives this diffraction pattern. Crystal Structure Metal Aluminum FCC Cadmium HCP Chromium BCC HCP FCC FCC Cobalt Copper Gold Iron (a) Lead BCC FCC Atomic Radius (nm) 0.1431 0.1490 0.1249 0.1253 0.1278 0.1442 0.1241 0.1750 Metal Molybdenum Nickel Platinum Silver Tantalum Titanium (a) Tungsten Zinc Crystal Structure BCC FCC FCC FCC BCC HCP BCC HCP Atomic Radius (nm) 0.1363 0.1246 0.1387 0.1445 0.1430 0.1445 0.1371 0.1332
Expert Answer:
Related Book For 

Materials Science and Engineering An Introduction
ISBN: 978-1118324578
9th edition
Authors: William D. Callister Jr., David G. Rethwisch
Posted Date:
Students also viewed these accounting questions
-
Iron has a BCC crystal structure, an atomic radius of 0.124 nm, and an atomic weight of 55.85 g/mol. Compute and compare its theoretical density with the experimental value found inside the front...
-
For both FCC and BCC crystal structures, there are two different types of interstitial sites. In each case, one site is larger than the other, and is normally occupied by impurity atoms. For FCC,...
-
Iron and vanadium both have the BCC crystal structure and V forms a substitutional solid solution in Fe for concentrations up to approximately 20 wt% V at room temperature. Determine the...
-
Did you ever purchase a bag of M&??s candies and wonder about the distribution of colors? Did you know in the beginning they were all brown? Now, peanut M&Ms are 12% brown, 15% yellow, 12% red, 23%...
-
Luxury Furniture designs and builds factory-made, premium, wood armoires for homes. All are of white oak. Its budgeted manufacturing overhead costs for the year 2012 are as follows. Overhead Cost...
-
A torque T is applied to a thin-walled tube having a cross section in the shape of a regular hexagon with constant wall thickness t and side length b (see figure). Obtain formulas for the shear...
-
(NPV and IRR with alternative costs) Mr. Fox has just bought an old apartment for $1,000,000. Mr. Fox plans to rent out the apartment. The apartment in its current condition can be rented out for...
-
Each of the Big 4 auditors along with Grant Thornton and BDO International provide illustrative financial statements on their web pages. www.pwc.com www.grantthornton.com www.kpmg.com...
-
the present value of the guaranteed residual value is deducted to determine the cost of goods sold. Indigo Equipment Leasing Company leased equipment to Cullumber Healthcare System on January 1 , 2 0...
-
Suppose that TechnoTCL is considering a new project. They are trying to determine the required rate of return for their debt and equity holders. See the information below: A 6 . 5 % ?percent annual...
-
Question no 11 Roberto invested $10,000 in an account that pays 2% per quarter. After 4 years, how much is the account worth? (Use the mathematical formula and round to the nearest penny A$10,201.51...
-
Why is it important to have a solid understanding of more than one technique in project management? Support your answer with an example.
-
Do Fijian organizations really need activity based costing? Critically support your view with the challenges in implementing ABC and make some suggestions to overcome with challenges? Presentation :...
-
A 2019 cash budget is being prepared for the purchase of Primer, a merchandise item. Budgeted data are: Cost of goods sold for 2019 P300,000;Accounts payable 1/1/19 20,000; Inventory, 1/1/19 30,000;...
-
The Stirling Cycle, or the cycle that the engine runs, is comprised of the following four steps: State 1-2: Isothermal expansion State 2-3: Isochoric heat removal State 3-4: Isothermal compression...
-
Board independence Are a majority of the board members independent? Is there evidence that the board ever meets without the CEO present? F. Board over commitment How many times does the entire board...
-
Find the nth term of a sequence whose first several terms are given 3 9 27 81 an
-
The relationship described in question 7 does not always appear to hold. What factors, besides the number of firms in the market, might affect margins?
-
For a continuous and oriented fiber-reinforced composite, the moduli of elasticity in the longitudinal and transverse directions are 33.1 and 3.66 GPa (4.8 106 and 5.3 105 psi), respectively. If...
-
A cylindrical piece of steel 38 mm (1*1/2 in.) in diameter is to be quenched in moderately agitated oil. Surface and center hard-nesses must be at least 50 and 40 HRC, respectively. Which of the...
-
Is it possible to produce an iron-carbon alloy that has a minimum tensile strength of 620 MPa (90,000 psi) and a minimum ductility of 50% RA? If so, what will be its composition and microstructure...
-
Ratio analysis over two years (Learning Objective 4) Comparative financial statement data of Weinstein, Inc., follow. 1. Market price of Weinsteins common stock: $49.00 at December 31, 2009, and...
-
Investment recommendation (Learning Objective 4) Take the role of an investment analyst at Prudential Bache. It is your job to recommend investments for your clients. The only information you have...
-
Make an investment decision (Learning Objective 4) Assume that you are purchasing an investment and have decided to invest in a company in the digital phone business. You have narrowed the choice to...

Study smarter with the SolutionInn App



