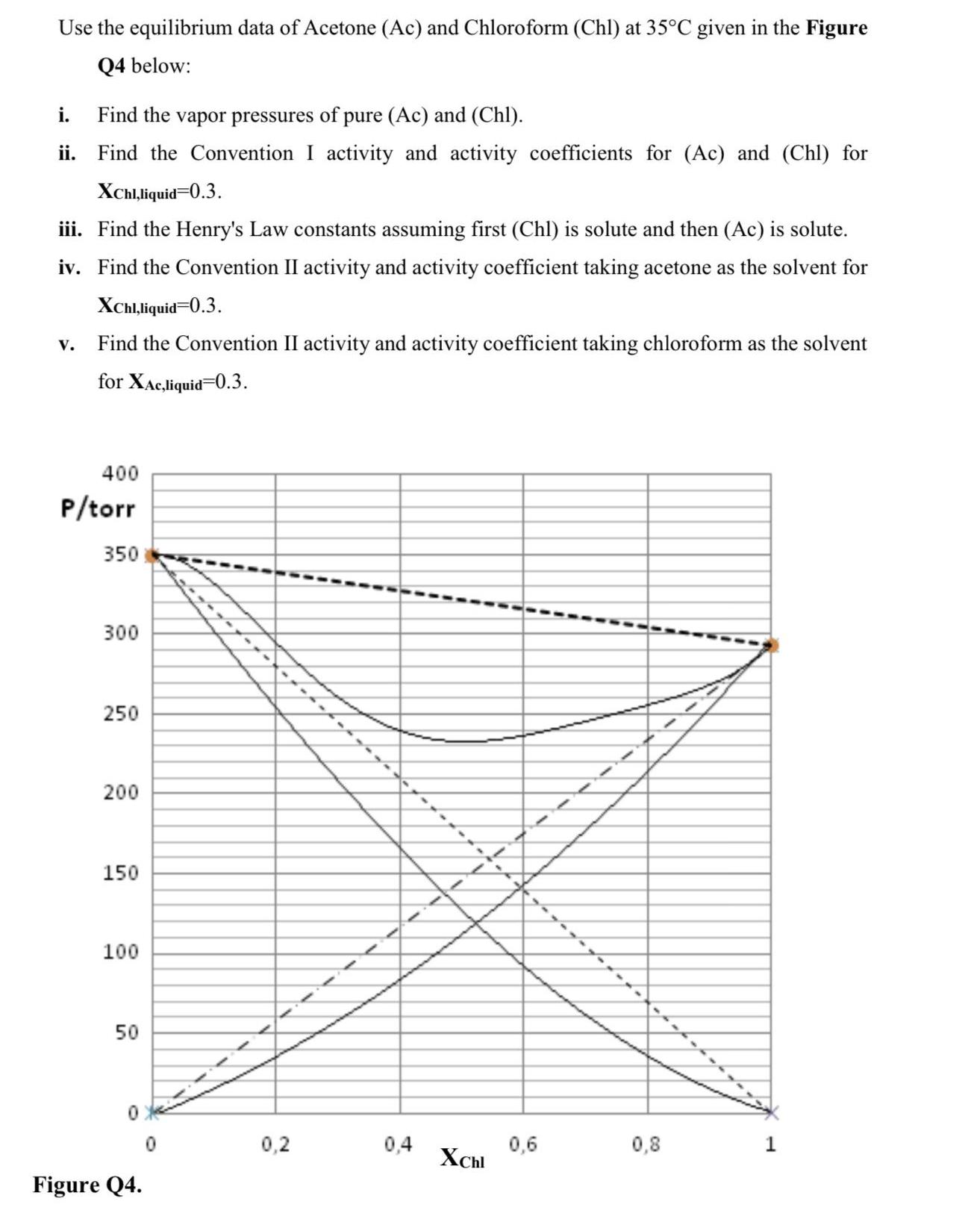
Use the equilibrium data of Acetone (Ac) and Chloroform (Chl) at 35C given in the Figure...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Use the equilibrium data of Acetone (Ac) and Chloroform (Chl) at 35°C given in the Figure Q4 below: i. Find the vapor pressures of pure (Ac) and (Chl). ii. Find the Convention I activity and activity coefficients for (Ac) and (Chl) for XChl,liquid=0.3. iii. Find the Henry's Law constants assuming first (Chl) is solute and then (Ac) is solute. iv. Find the Convention II activity and activity coefficient taking acetone as the solvent for XChl,liquid=0.3. V. Find the Convention II activity and activity coefficient taking chloroform as the solvent for XAc,liquid=0.3. 400 P/torr 350 300 250 200 150 100 50 Figure Q4. 0,2 0,4 XChl 0,6 0,8 Use the equilibrium data of Acetone (Ac) and Chloroform (Chl) at 35°C given in the Figure Q4 below: i. Find the vapor pressures of pure (Ac) and (Chl). ii. Find the Convention I activity and activity coefficients for (Ac) and (Chl) for XChl,liquid=0.3. iii. Find the Henry's Law constants assuming first (Chl) is solute and then (Ac) is solute. iv. Find the Convention II activity and activity coefficient taking acetone as the solvent for XChl,liquid=0.3. V. Find the Convention II activity and activity coefficient taking chloroform as the solvent for XAc,liquid=0.3. 400 P/torr 350 300 250 200 150 100 50 Figure Q4. 0,2 0,4 XChl 0,6 0,8
Expert Answer:
Answer rating: 100% (QA)
To find the various properties and coefficients as requested we can use the given equilibrium data for Acetone Ac and Chloroform Chl at 35C It appears ... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Posted Date:
Students also viewed these chemical engineering questions
-
Use the graph input tool to help you answer the following questions. You will not be graded on any changes you make to this graph. Note: Once you enter a value in a white field, the graph and any...
-
The revenue R (in millions of dollars) for a beverage company is related to its advertising expense by the function R = 1/100,000 (-x3 + 600x2), 0 ¤ x ¤ 400 Where x is the amount spent...
-
The "Optimal Deadbeat" Problem: The World Bank is considering a stream of loans to the Puglian government to help it develop its nationalized oil fields and refineries. This is the only set of loans...
-
The director of cost management for Odessa Company uses a statistical control chart to help management determine when to investigate variances. The critical value is 1 standard deviation. The company...
-
Consider the same vortex filament as in Prob. 5.1. Consider also a straight line through the center of the loop, perpendicular to the plane of the loop. Let A be the distance along this line,...
-
What test does the auditor make to determine if property additions are complete?
-
Beccan Company is a discount tire dealer operating 25 retail stores in the metropolitan area. Beccan sells both private-brand and name-brand tires. The company operates a centralized purchasing and...
-
A partner in the national accounting firm mentioned in the previous questions does not believe that the time estimates for each activity can be specified with certainty. Instead, she prefers to...
-
BTR is a manufacturing company in B-land. In the year ended 31 December 20X5, BTR has a working capital cycle of 40 days, a receivables collection period of 30 days, and average inventory of B$240...
-
U.P.I. Industries Ltd., a Canadian corporation, has recently been designated a public corporation. Its shares are traded on the Winnipeg Stock Exchange. Over the past year, the company has pursued an...
-
Question Industrial Adhesives manufactures and markets commercial grade petroleum-based Adhesives, through wholesalers in most cases, but also directly to several large clients. It is currently...
-
One of the methods for deciding how much life insurance to buy is to consider how much future income would be lost in the event of a premature death and buy enough life insurance today to replace...
-
.The vertical distance h travelled by a freely falling body can be computed from the formula h = vot + gt2 where vo is the initial velocity, g is the gravitational acceleration, and t is the time of...
-
2. Use linear approximation to approximate 10 - 1.0012/(5.002).
-
The following items are taken from the financial statements of Pelle Ltd. For the year ended December 31, 2022: ...... Accounts payable.............................................................
-
Discuss the electronic discovery/electronic evidence issues with each of the scenarios. discussion should include the Federal Rules of Civil Procedure (FRCP), ABA Model Rules of Professional Conduct,...
-
Jim Daniels Health Products has eight stores. The firm wants to expand by two more stores and needs a bank loan to do this. Mr. Hewitt, the banker, will finance construction if the firm can present...
-
The Place-Plus real estate development firm in Problem 24 is dissatisfied with the economists estimate of the probabilities of future interest rate movement, so it is considering having a financial...
-
A frustrated professor once claimed that if all the reports she had graded in her career were stacked on top of one another, they would reach from the Earth to the moon. Assume that an average report...
-
In terms of Le Chateliers Principle, explain why steam reforming is done at low pressures while methanol synthesis is performed at moderate to high pressures. Explain why reforming is done at high...
-
The Cookenwythe Gas Company pumps propane gas to the nearby Noxious Chemicals, Inc., polypropylene production plant. The gas is metered at the noxious plant at 400m 3 /h at 4.7atm gauge and 30C. The...
-
Photovoltaics, Inc. is an Arizona-based manufacturer and distributor of photovoltaic solar energy units. The company was founded in 2015 by Arthur Manelas and Harry Linn. Manelas, formerly a research...
-
In January 2017, Susan and Clark Shipley, co-owners of Island Foods, Inc. began discussing the possibility of expanding their restaurant business from a single location in Glendale, Arizona, to two...
-
The 2015 annual report of The Procter & Gamble Company (P&G) is available at www.pginvestor.com. After reviewing P&Gs annual report, respond to the following questions: a. P&Gs net income and net...

Study smarter with the SolutionInn App


