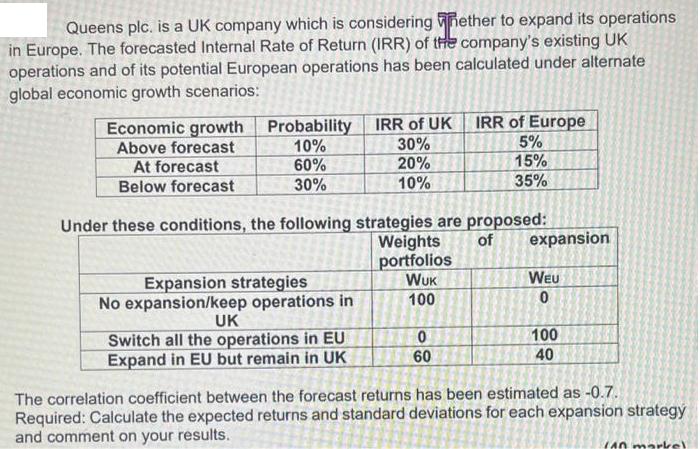
Queens plc. is a UK company which is considering whether to expand its operations in Europe....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Queens plc. is a UK company which is considering whether to expand its operations in Europe. The forecasted Internal Rate of Return (IRR) of the company's existing UK operations and of its potential European operations has been calculated under alternate global economic growth scenarios: Economic growth Above forecast At forecast Below forecast Probability IRR of UK 10% 30% 60% 20% 30% 10% Under these conditions, the following strategies are proposed: Weights of portfolios WUK 100 Expansion strategies No expansion/keep operations in UK Switch all the operations in EU Expand in EU but remain in UK IRR of Europe 5% 15% 35% 0 60 expansion WEU 0 100 40 The correlation coefficient between the forecast returns has been estimated as -0.7. Required: Calculate the expected returns and standard deviations for each expansion strategy and comment on your results. (40 Queens plc. is a UK company which is considering whether to expand its operations in Europe. The forecasted Internal Rate of Return (IRR) of the company's existing UK operations and of its potential European operations has been calculated under alternate global economic growth scenarios: Economic growth Above forecast At forecast Below forecast Probability IRR of UK 10% 30% 60% 20% 30% 10% Under these conditions, the following strategies are proposed: Weights of portfolios WUK 100 Expansion strategies No expansion/keep operations in UK Switch all the operations in EU Expand in EU but remain in UK IRR of Europe 5% 15% 35% 0 60 expansion WEU 0 100 40 The correlation coefficient between the forecast returns has been estimated as -0.7. Required: Calculate the expected returns and standard deviations for each expansion strategy and comment on your results. (40
Expert Answer:

Related Book For 

Advanced Accounting
ISBN: 978-0077431808
10th edition
Authors: Joe Hoyle, Thomas Schaefer, Timothy Doupnik
Posted Date:
Students also viewed these accounting questions
-
Trendsetter plc is a UK company which produces specialist footwear and imports leather from a company in India, Amulta Limited. The company exports the footwear worldwide, but the majority of the...
-
Your company is planning to expand its operations from direct delivery to the markets to a distribution network. The company needs to decide the location of its plants and warehouses. It is important...
-
Under the internal rate of return method, how would you decide which projects to accept and which projects to reject? Explain.
-
(a) Find the first order fraction transformation where z 1 =,z 2 =0, z 3 =1 is thought to be w 1 =1, w 2 =i, w 3 =-1 each. (b) Find the anchor points of w=(z-1)/(z+1)
-
On January 5, Brian drafts a check for $3,000 drawn on Southern Marine Bank and payable to his assistant, Shanta. Brian puts last years date on the check by mistake. On January 7, before Shanta has...
-
Use the Creator Corporation data in E10-46B to prepare the stockholders equity section of the companys balance sheet at December 31, 2019. Data from E10-46B At December 31, 2018, Creator Corporation...
-
When to use the official form complaints?
-
Five months before the new 2002 Lexus ES hit showroom floors, the company's U.S. engineers sent a test report to Toyota City in Japan: The luxury sedan shifted gears so roughly that it was "not...
-
1) State the MVT 2) Suppose that t (1)=6 and '(x) <2. , use the MVT to give an upper bound for rf (10). (In other words say how big it can be)
-
You have just been hired as a brand manager at Kelsey-White, an American multinational consumer goods company. Recently the firm invested in the development of K-W Vision, a series of systems and...
-
Assume that you manage a $10.00 million mutual fund that has a beta of 1.05 and a 9.50% required return. The risk-free rate is 2.20%. You now receive another $14.50 million, which you invest in...
-
Write a program that takes a start string and a stop string as commandline arguments and prints all substrings of a given string that start with the first, end with the second, and otherwise contain...
-
Develop a data type for the naming scheme for vehicles known as the Vehicle Identification Number (VIN). A VIN describes the make, model, year, and other attributes of cars, buses, and trucks in the...
-
Add a method isReachable(v) to PathFinder that returns true if there exists some path from the source to \(v\), and false otherwise.
-
Implement the method size() for BST.
-
Apply the scientific method to develop and validate a hypothesis about the order of growth of the running time of each of the following two code fragments as a function of \(n\). String s = for (int...
-
A state university has a system where most of the decisions are made by the top level of management and the subordinates are not supposed to question them. There are no complex decision-making...
-
In Problem use absolute value on a graphing calculator to find the area between the curve and the x axis over the given interval. Find answers to two decimal places. y = x 3 ln x; 0.1 x 3.1
-
Domer Corporation is preparing to issue a relatively small amount of securities and does not want to go to the trouble and expense of filing a registration statement with the SEC. Company officials...
-
A company is being created and the owners are trying to decide whether to form a general partnership, a limited liability partnership, or a limited liability company. What are the advantages and...
-
A city operates a solid waste landfill. This facility is 11 percent full after the first year of operation and 24 percent after the second year. How much expense should be recognized on the...
-
In a laboratory investigation, acetic acid is esterified in the liquid phase with ethanol at \(373.15 \mathrm{~K}\) and \(1 \mathrm{~atm}\) pressure to produce ethyl acetate and water according to...
-
The reaction for oxidation of sulphur is given by \[ \mathrm{SO}_{2}(\mathrm{~g})+\frac{1}{2} \mathrm{O}_{2}(\mathrm{~g}) ightarrow \mathrm{SO}_{3}(\mathrm{~g}) \] A mixture of sulphur dioxide and...
-
The following thermal decomposition occurs at \(400 \mathrm{~K}\) : \[ A(\mathrm{~s}) ightarrow B(\mathrm{~s})+C(\mathrm{~g}) \] The standard Gibbs free energy of the reaction, \(\Delta...

Study smarter with the SolutionInn App


