Question 1 1. Which, if any, of the substances had resonance structures? How many resonance structures...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
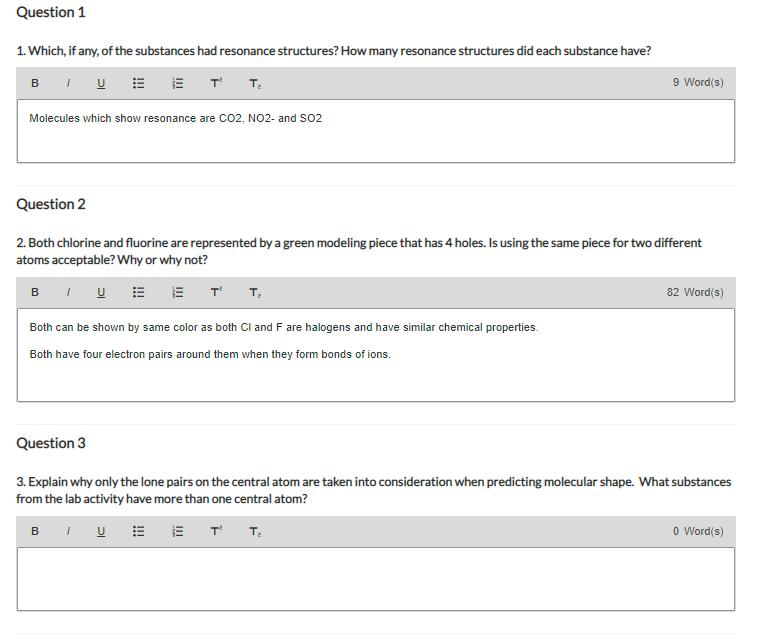
Question 1 1. Which, if any, of the substances had resonance structures? How many resonance structures did each substance have? BUE ETT, Molecules which show resonance are CO2, NO2- and SO2 Question 2 2. Both chlorine and fluorine are represented by a green modeling piece that has 4 holes. Is using the same piece for two different atoms acceptable? Why or why not? BIU T T₂ 9 Word(s) Both can be shown by same color as both Cl and F are halogens and have similar chemical properties. Both have four electron pairs around them when they form bonds of ions. 82 Word(s) Question 3 3. Explain why only the lone pairs on the central atom are taken into consideration when predicting molecular shape. What substances from the lab activity have more than one central atom? BUEET T₂ 0 Word(s) Question 1 1. Which, if any, of the substances had resonance structures? How many resonance structures did each substance have? BUE ETT, Molecules which show resonance are CO2, NO2- and SO2 Question 2 2. Both chlorine and fluorine are represented by a green modeling piece that has 4 holes. Is using the same piece for two different atoms acceptable? Why or why not? BIU T T₂ 9 Word(s) Both can be shown by same color as both Cl and F are halogens and have similar chemical properties. Both have four electron pairs around them when they form bonds of ions. 82 Word(s) Question 3 3. Explain why only the lone pairs on the central atom are taken into consideration when predicting molecular shape. What substances from the lab activity have more than one central atom? BUEET T₂ 0 Word(s)
Expert Answer:
Related Book For 

Understanding Basic Statistics
ISBN: 978-1111827021
6th edition
Authors: Charles Henry Brase, Corrinne Pellillo Brase
Posted Date:
Students also viewed these accounting questions
-
If any of the following octahedral complexes display geometric isomerism, draw the structures and label them cis or trans. a. [Co(NO2)4(NH3)2] b. [Co(NH3)5(NO2)]2+ c. [Co(NH3)6]3+ d. [Cr(NH3)5Cl]2+
-
Which if any of these statements are true? Stock prices appear to behave as though successive values. a. are random numbers. b. follow regular cycles. c. differ by a random number.
-
If any of the three assumptions in Modigliani and Miller Proposition 1 are relaxed, which has the most predictably quantifiable impact on the value of the firm?
-
When are objects on the periphery of your vision most noticeable?
-
Write a query to display the invoice number, line numbers, product SKUs, product descriptions, and brand ID for sales of sealer and top coat products of the same brand on the sameinvoice. nv Num...
-
MSI is considering eliminating a product from its ToddleTown Tours collection. This collection is aimed at children one to three years of age and includes "tours" of a hypothetical town. Two...
-
Must the economic ascent of nations such as China and India come at the price of decline in Europe and North America, or is it possible for economic conditions to simultaneously improve in all...
-
According to the Netflix Reference Guide on Our Freedom & Responsibility Culture, Freedom and Responsibility applies to our salaried employees. Our hourly employees are important but have more...
-
Consider the following cases: A child informs you that she has been abused physically and/or sexually. An adult female informs you that they are being abused physically and emotionally by their...
-
Consolidated Income Statement, Intercompany Transactions ments for Pon and its 80 percent-owned subsidiary, Star, appear below. Condensed income state- Condensed Income Statements Star Pon $9,000,000...
-
Ramort Company reports the following cost data for its single product The company regularly sells 17,000 units of its product at a price of $54.00 per unit. 9.4 per unit 11.40 per unit Direct...
-
Describe the major perspectives on the influence of peer groups on antisocial and delinquent behavior.
-
Elizabeth and Rick would like to purchase a new home. Their mortgage broker has pre-qualified them for two mortgages: a 30-year fixed at 4 percent or a 5-year ARM at 3 percent. Which of the following...
-
What are the three sources to which behavioral geneticists attribute individual differences in behavior?
-
A tenancy by the entirety may be terminated in which of the following ways? I. Creditor of a spouse. II. Death of either spouse. III. Divorce settlement. IV. Mutual agreement by both spouses. A. I,...
-
George and Alice Center are married. George has one brother, Monroe. George has become incompetent and unable to update his will. Alice and Georges residence is held as a joint tenancy with rights of...
-
make a Calendar table from (1980 jan1) to (2050 dec 31).
-
Write a paper about the Working relationship in the organization- collaboration within and outside the organization
-
What is a random sample from a population?
-
Consider a binomial experiment with n = 6 trials where the probability of success on a single trial is p = 0.20. (a) Find P(0 < r 22. (b) If you conducted the experiment and got 1 or 2 successes,...
-
(a) Draw a tree diagram to display all the possible headstails sequences that can occur when you flip a coin three times. (b) How many sequences contain exactly two heads? (c) Probability Extension...
-
One of the models used for predicting the severity of head injury is shown in Fig. 5.68 where \(f(t)\) denotes the force of blow to the head, \(m_{1}\) is the mass of that part of the head which lies...
-
Write the equations of motion of a multidegree-of-freedom system in matrix form using a. the flexibility matrix, and b. the stiffness matrix.
-
What is a generalized mass matrix?

Study smarter with the SolutionInn App



