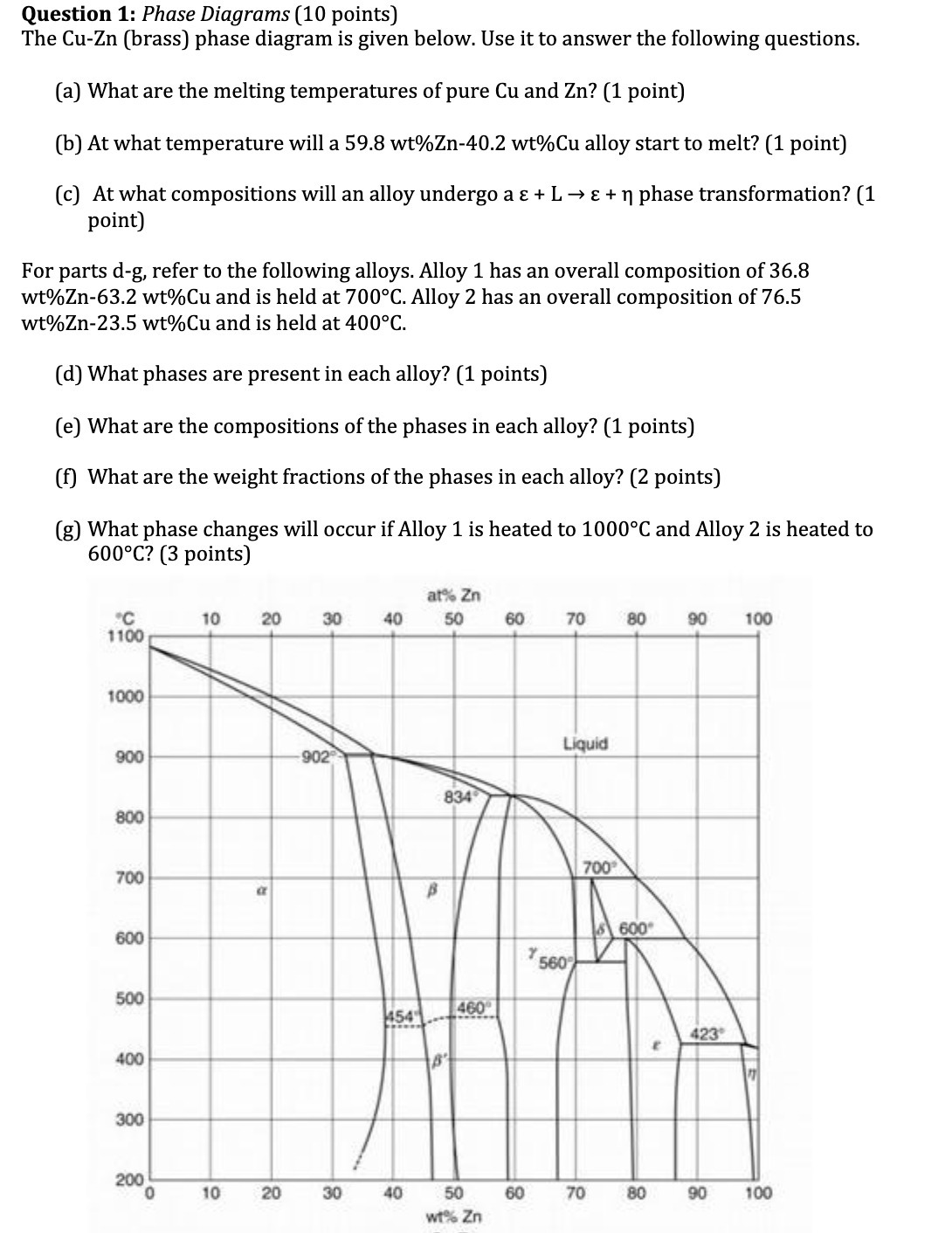
Question 1: Phase Diagrams (10 points) The Cu-Zn (brass) phase diagram is given below. Use it...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Question 1: Phase Diagrams (10 points) The Cu-Zn (brass) phase diagram is given below. Use it to answer the following questions. (a) What are the melting temperatures of pure Cu and Zn? (1 point) (b) At what temperature will a 59.8 wt% Zn-40.2 wt%Cu alloy start to melt? (1 point) (c) At what compositions will an alloy undergo a + L + phase transformation? (1 point) For parts d-g, refer to the following alloys. Alloy 1 has an overall composition of 36.8 wt%Zn-63.2 wt%Cu and is held at 700C. Alloy 2 has an overall composition of 76.5 wt%Zn-23.5 wt%Cu and is held at 400C. (d) What phases are present in each alloy? (1 points) (e) What are the compositions of the phases in each alloy? (1 points) (f) What are the weight fractions of the phases in each alloy? (2 points) (g) What phase changes will occur if Alloy 1 is heated to 1000C and Alloy 2 is heated to 600C? (3 points) C 1100 1000 900 800 700 600 500 400 300 200 0 10 10 20 a 20 30 -902 30 40 454 40 at% Zn 50 B 834 B 460 50 wt% Zn 60 60 Y 70 Liquid 560 700 70 80 600 80 E 90 100 423 90 100 Question 1: Phase Diagrams (10 points) The Cu-Zn (brass) phase diagram is given below. Use it to answer the following questions. (a) What are the melting temperatures of pure Cu and Zn? (1 point) (b) At what temperature will a 59.8 wt% Zn-40.2 wt%Cu alloy start to melt? (1 point) (c) At what compositions will an alloy undergo a + L + phase transformation? (1 point) For parts d-g, refer to the following alloys. Alloy 1 has an overall composition of 36.8 wt%Zn-63.2 wt%Cu and is held at 700C. Alloy 2 has an overall composition of 76.5 wt%Zn-23.5 wt%Cu and is held at 400C. (d) What phases are present in each alloy? (1 points) (e) What are the compositions of the phases in each alloy? (1 points) (f) What are the weight fractions of the phases in each alloy? (2 points) (g) What phase changes will occur if Alloy 1 is heated to 1000C and Alloy 2 is heated to 600C? (3 points) C 1100 1000 900 800 700 600 500 400 300 200 0 10 10 20 a 20 30 -902 30 40 454 40 at% Zn 50 B 834 B 460 50 wt% Zn 60 60 Y 70 Liquid 560 700 70 80 600 80 E 90 100 423 90 100
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these mechanical engineering questions
-
Q1. You have identified a market opportunity for home media players that would cater for older members of the population. Many older people have difficulty in understanding the operating principles...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Suppose we have an automatic module on the module path named lizard-^-cricket-^-1.0.0-SNAPSHOT.jar and no Automatic-Module-Name specified. What module name should named modules use to reference it?...
-
Gill Company, organized in 2019, has the following transactions related to intangible assets . 1/2/19...................Purchased patent (7-year life)..............$595,000 4/1/19............Goodwill...
-
Given a Q-point of IDQ = 3 mA and VGS = -3 V, determine IDSS if VP = -6 V.
-
A \(30 \mathrm{ft} \times 20 \mathrm{ft}\) office space has a photosensor dimmer control working with installed lighting of \(2 \mathrm{~W} / \mathrm{ft}^{2}\). The required workplace illuminance is...
-
Barrymore Costume Company, located in New York City, sews costumes for plays and musicals. Barrymore considers itself primarily a service firm, as it never produces costumes without a pre-existing...
-
1. A 45 kg sled is pulled at a constant velocity across a horizontal slippery surface. If a force of 320.0 N is being applied to the sled rope at an angle of 28.0 degrees to the ground, what is the...
-
. Two long, parallel wires carry currents of I = 3.00 A and I = 5.00 A in the directions indicated in Figure P30.20. (a) Find the magnitude and direction of the magnetic field at a point midway...
-
1. It is found that the equilibrium concentration of the undissociated form (HA) of a weak monoprotic acid, in an aqueous solution of pH-3, is 9.398x104 mol/L. It is also known that an equal amount...
-
C. Home Insert Draw Page Layout X n AutoSave Paste C21 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 A Hours Worked Pay Rate...
-
0 Required information [The following information applies to the questions displayed below) Henrich is a single taxpayer. In 2023, his taxable income is $537,000. What are his income tax and net...
-
Parker Corporation reported the following related to property and equipment (all in millions): MOI-LAL. Requirement 1. T-accounts for Property and Equipment and Accumulated Depreciation have been...
-
The financial statements of Evans News, Inc., include the following items: (Click the icon to view the financial statements.) Requirements Requirement 1. Compute the following ratios for 2020 and...
-
The Berry Phoenix BrickWorks, in Saskatoon, Saskatchewan, manufactures high-quality bricks used in residential and commercial construction. The firm is small but highly automated and typically...
-
Catalytic hydrogenation of naphthalene over PdC results in rapid addition of 2 moles of H 2 . Propose a structure for this product.
-
Detonation of nitroglycerin proceeds as follows: (a) If a sample containing 2.00 mL of nitroglycerin (density 1.592 g/mL is detonated, how many total moles of gas are produced? (b) If each mole of...
-
The solubility of Cr(NO3)3 ( 9 H2O in water is 208 g per 100 g of water at 15oC. A solution of Cr(NO3)3 ( 9 H2O in water at is formed by dissolving 324 g in 100 g water. When this solution is slowly...
-
(a) What is the mass percentage of iodine (I2) in a solution containing 0.035 mol I2 in 125 g of CCl4? (b) Seawater contains per 0.0079 g Sr2+ kilogram of water. What is the concentration of Sr2+...
-
S1 Ltd and S2 Ltd belong to the same capital gains group. In May 2020, S1 Ltd transferred a chargeable asset to S2 Ltd. The original cost of this asset to S1 Ltd was 10,000 and its market value in...
-
The ordinary share capital of W Ltd (which is a trading company) is owned 30% by X Ltd, 25% by Y Ltd and 45% by Z Ltd. All of these companies are UK resident and they prepare accounts to 31 March....
-
E Ltd has taxable total profits of 800,000 for the year to 31 March 2024 and receives no dividends. For many years, the company has owned 65% of the ordinary shares of F Ltd and 30% of the ordinary...

Study smarter with the SolutionInn App


