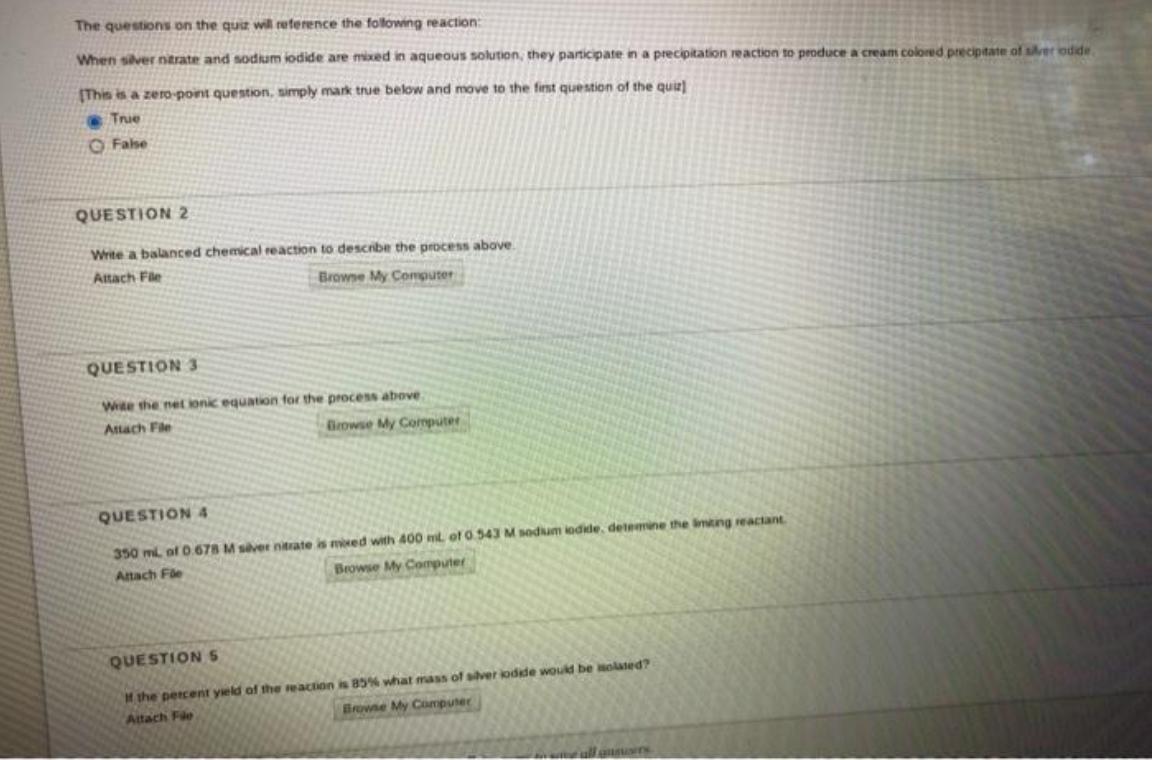
The questions on the quiz will reference the following reaction: When siver ntrate and sodium iodide...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The questions on the quiz will reference the following reaction: When siver ntrate and sodium iodide are mixed in aqueous solution, they participate in a precipitation reaction to produce a cream colored precipitate of srer odde (This is a zero-point question, simply mark true below and move to the fint question of the quie) True O False QUESTION 2 Write a balanced chemical reaction to describe the pocess above. Attach Fle Browse My Computer QUESTION 3 Wite the net ionic equation for the process above Attach Fle Browse My Coputer QUESTION 4 350 ml of .678 M svet nitrate is moed with 400 mt of 0.543 M sodium iodide, detemine the imting reactant Attach Fle Browse My Computer QUESTION 5 Mthe percent yeld of the reaction is 85% what mass of silver kodde would be Holated? Browne My Computer Attach Fle The questions on the quiz will reference the following reaction: When siver ntrate and sodium iodide are mixed in aqueous solution, they participate in a precipitation reaction to produce a cream colored precipitate of srer odde (This is a zero-point question, simply mark true below and move to the fint question of the quie) True O False QUESTION 2 Write a balanced chemical reaction to describe the pocess above. Attach Fle Browse My Computer QUESTION 3 Wite the net ionic equation for the process above Attach Fle Browse My Coputer QUESTION 4 350 ml of .678 M svet nitrate is moed with 400 mt of 0.543 M sodium iodide, detemine the imting reactant Attach Fle Browse My Computer QUESTION 5 Mthe percent yeld of the reaction is 85% what mass of silver kodde would be Holated? Browne My Computer Attach Fle
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Write a balanced chemical equation to describe how magnesium ions are removed in water treatment by the addition of slaked lime, Ca(OH)2.
-
Write a total ionic and net ionic equation for the following reaction between Pb2+ and HCl.
-
Silver nitrate reacts with strontium chloride in an aqueous precipitation reaction. What are the formulas of silver nitrate and strontium chloride? Write the molecular equation and net ionic equation...
-
The Archaic period also gave rise to some economic, social and political cooperation, best represented by the emergence of Kingdoms Republics Tyrannies Poleis
-
Discuss why corporations frequently offer both qualified and nonqualified retirement plans to their employees.
-
Water falls down a vertical pipe by gravity alone. The flow between vertical locations z 1 and z 2 is fully developed, and velocity profiles at these two locations are sketched in Fig. P10112. Since...
-
The 50-mm-diameter cylinder is made from Am 1004-T61 magnesium and is placed in the clamp when the temperature is T 1 = 20C. If the 304-stainless-steel carriage bolts of the clamp each have a...
-
DataPoint Engineering is considering the purchase of a new piece of equipment for $240,000. It has an eight-year midpoint of its asset depreciation range (ADR). It will require an additional initial...
-
A rigid container contains an ideal gas 40C which is stirred by a blade. The blade produces 200kJ of work. The temperature remains constant thanks to a heat exchange with the surroundings which are...
-
[The following information applies to the questions displayed below.) Assume Down, Incorporated, was organized on May 1 to compete with Despair, Incorporated-a company that sells de- motivational...
-
Companies that use digitized data collection systems generate information that can provide many knowledge nuggets dealing with customer buying preferences One issue that is contentious with employers...
-
When must Entry A be split into two entries, A1 and A2, so that goodwill can be allocated between the parent and the NCI on a different basis than the identifiable assets? When excess fair value is...
-
Let (an) be the sequence defined by a = 5 and an = 2an-1 - 3 when n > 2 - What is a4?
-
Multiple Select Question Select all that apply What are some recommendations concerning the use of support material for a business presentation? Multiple select question. Have additional support...
-
Kim and John separated 3 years after John's mother, Kathy passed away. When Kathy died, she left him $300,000 through her will. In which scenario would Kim be entitled to the inheritance? None of the...
-
Can you explain if it is possible for production and accounting to manage the generic product pilot project? What type of costing system would this be called? Explain what this costing system means....
-
teacher webpage. Webpages are another way the teacher can communicate with parents/guardians and students. You will need to create a webpage that looks like it has been actively maintained for at...
-
What are the four types of poultry production systems? Explain each type.
-
The compound shown is a somewhat stronger base than ammonia. Which nitrogen do you think is protonated when it is treated with an acid? Write a structural formula for the species that results. Refer...
-
Suggest a reasonable mechanism for each of the following reactions: (a) (b) (c) (d) I. excess NaNH, NH C,H,Br CH(COOCH,CH)OoCH,CH,) l excess NH 2.,0 CH2CH2CH2CH2COCH2CH3 NaNH Cl COOCH CH CH3 CH3...
-
Write equations showing how 1-phenylethanol could be prepared from each of the following starting materials: (a) Bromobenzene (b) Benzaldehyde (c) Benzyl alcohol (d) Acetophenone (e) Benzene...
-
You are setting up a chatbot agency to service marketing, sales and customer services teams. Discuss the advantages and disadvantages of setting up the business as a sole trader or company and the...
-
Principles for Responsible Management Education (PRME) is a not-for-profit entity. It engages business schools to ensure they provide future leaders with the skills needed to balance economic and...
-
Entities are expected to perform in environmental, governance and social areas as well as profit (unless a not-for-profit entity). List some key performance indicators applicable to each sphere.

Study smarter with the SolutionInn App


