If 15.0 ml of 0.1 normal KMNO, solution is used to titrate 0.12 g of an...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
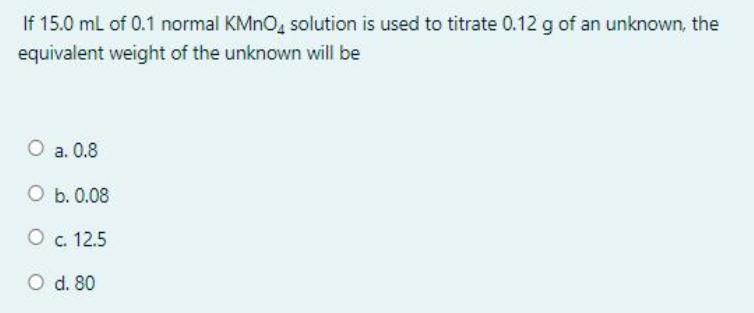
If 15.0 ml of 0.1 normal KMNO, solution is used to titrate 0.12 g of an unknown, the equivalent weight of the unknown will be O a. 0.8 O b. 0.08 O c. 12.5 O d. 80 If 15.0 ml of 0.1 normal KMNO, solution is used to titrate 0.12 g of an unknown, the equivalent weight of the unknown will be O a. 0.8 O b. 0.08 O c. 12.5 O d. 80
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Consider the diode bridge rectifier shown below with the voltage regulating capacitor in parallel with the load resistor. 60Hz 120V 10:1 C-470F IL R=1kQ a) For the input signal, sketch waveforms of...
-
If 45.1 mL of a solution containing 8.30 g of silver nitrate is added to 30.6 mL of 0.511 M sodium carbonate solution, calculate the molarity of silver ion in the resulting solution. (Assume volumes...
-
A solution was prepared by mixing 10.00 mL of unknown (X) with 5.00 mL of standard (S) containing 8.24 g S/mL and diluting the mixture to 50.0 mL. The measured signal quotient was (signal due to...
-
Calculate the standard entropy change for the following reactions at 25C. Comment on the sign of r S. (a) 2 Al(s) + 3 Cl 2 (g) 2 AlCl 3 (s) (b) 2 CH 3 OH() + 3 O 2 (g) 2 CO 2 (g) + 4 H 2 O(g)
-
How many runs are there in each of the following series? a. 1 1 0 0 0 1 0 0 1 0 1 1 0 0 0 0 1 1 0 1 1 0 1 0 1 1 1 0 0 1 b. 0 1 0 1 1 0 1 1 1 0 0 0 1 0 1 1 0 0 1 0 1 0 0 1 0 1 1 0 0 1 c. 1 1 1 0 0 0 1...
-
Watch the Video Highlight in this chapter. Find at least two other references related to this topic and discuss how it relates to project schedule management. Summarize your findings and opinions in...
-
The accounting records for ADR, Inc., reflected the following amounts at the end of August 2010: Prepare ADRs multi-step income statement for the fiscal year ended August 31,2010. Cash...... $3,500...
-
After its first month of operation, the following amounts were taken from the accounting records of Polaris Realty Inc. as of November 30, 2011.Prepare an income statement for the month ending...
-
Question 3 Provide short but precise answers to the following questions a.Considering the threat money laundering poses for international finance, what are bankers required to do to prevent this...
-
Go back and quickly review the rules that the Sarbanes-Oxley Act of 2002 placed on the auditing profession as described in this chapter. What do you think are the two or three main purposes of these...
-
Svetlana and Yoric are each thinking of starting the same type of business. Svetlana is young and into technology and mechanization. She has a large, fixed cost of $15,000, but her advanced...
-
Ruby Company manages the daily operations for the human resources department of Jade Company. In terms of the contract, Ruby receives a fee of \($100\) per year for each employee on Jades payroll,...
-
Rhodes Co. manages a school for the Old England School District. In year 1, Rhodes Co.s first year of operating the school, the school earned per pupil fees of \($100,000.\) Rhodes is responsible for...
-
Greasy Patties Corporation, a hamburger chain, has 100 stores. In its tenth year of operating, its total sales for year 10 amounted to \($100\) million. During year 11, Greasy Patties Corporation...
-
In the ____________case, the issue was that it did not accurately and completely describe the realities of its operations in its MD & A.
-
Explain the need for financial managers to balance liquidity and solvency.
-
Answer each part by using well-labelled graphs for the IS-LM, WS-PS and AS-AD models: a) Assume that Australian government has made it very difficult for refugees (who are prepared to work at lower...
-
Linda Lopez opened a beauty studio, Lindas Salon, on January 2, 2011. The salon also sells beauty supplies. In January 2012, Lopez realized she had never filed any tax reports for her business and...
-
A molecular compound is composed of 58.8% Xe, 7.2% O, and 34.0% F, by mass. If the molecular weight is 223 amu, what is the molecular formula? What is the Lewis formula? Predict the molecular...
-
Write Lewis symbols for the following: a. Br b. Br c. Sr d. Sr2+
-
Tertiary butyl chloride reacts in basic solution according to the equation The accepted mechanism for this reaction is What should be the rate law for this reaction? (CH3);CCI + OH (CH3),COH + CI
-
Distinguish among the following tests and indicate the assertions to which each test pertains: a. Vouch recorded payables to supporting documentation. b. Examine subsequent payments. c. Determine...
-
a. Discuss materiality from the perspective of the production cycle. b. Discuss inherent risk from the perspective of the production cycle. c. Why might the auditor use different audit strategies for...
-
What circumstances pertaining to property, plant, and equipment may warrant the following assessments of inherent risk (IR)? a. Low for the existence or occurrence assertion. b. Moderate or high for...

Study smarter with the SolutionInn App



