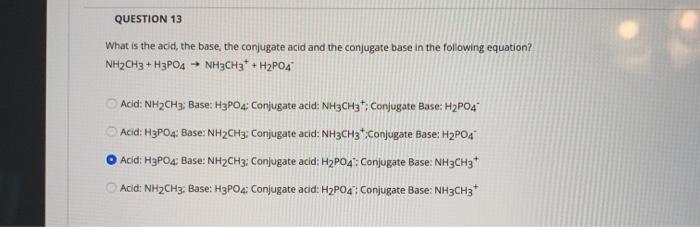
QUESTION 13 What is the acid, the base, the conjugate acid and the conjugate base in...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
QUESTION 13 What is the acid, the base, the conjugate acid and the conjugate base in the following equation? NH2CH3 + H3PO4 - NH3CH3 + H2PO4 Acid: NH₂CH3: Base: H3PO4; Conjugate acid: NH3CH3; Conjugate Base: H₂PO4 Acid: H3PO4: Base: NH₂CH3: Conjugate acid: NH3CH3*:Conjugate Base: H₂PO4 Acid: H3PO4; Base: NH2CH3; Conjugate acid: H2PO4: Conjugate Base: NH3CH3* Acid: NH₂CH3: Base: H3PO4: Conjugate acid: H₂PO4: Conjugate Base: NH3CH3* QUESTION 13 What is the acid, the base, the conjugate acid and the conjugate base in the following equation? NH2CH3 + H3PO4 - NH3CH3 + H2PO4 Acid: NH₂CH3: Base: H3PO4; Conjugate acid: NH3CH3; Conjugate Base: H₂PO4 Acid: H3PO4: Base: NH₂CH3: Conjugate acid: NH3CH3*:Conjugate Base: H₂PO4 Acid: H3PO4; Base: NH2CH3; Conjugate acid: H2PO4: Conjugate Base: NH3CH3* Acid: NH₂CH3: Base: H3PO4: Conjugate acid: H₂PO4: Conjugate Base: NH3CH3*
Expert Answer:
Answer rating: 100% (QA)
Answer Acid HPO4 Base NH CH3 Congigate add NHCH Con... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the acid test ratio?
-
In general, as base strength increases, conjugate acid strength decreases. Explain why the conjugate acid of the weak base NH3 is a weak acid.
-
Question 13 What is the minimum size of memory required to implement a HEX to gray-code converter? Not yet answered Select one: Marked out of a. None of the given options. 1.00 O b. 16 x 2 bits. P...
-
A physical pendulum of mass m = 3 . 3 6 kg is comprised of an odd shape that has a centre - of - mass a distance of d = 0 . 5 5 5 m from the pivot point. The pendulum is displaced from equilibrium to...
-
An analysis of the income statement and the balance sheet accounts of Headrick, Inc., at December 31, 2009, provides the following information. Income Statement Items: Gain on sale of Marketable...
-
In September 2017, Manson Paint Corporation began operations in a state that requires new employers of one or more individuals to pay a state unemployment tax of 3.5% of the first $7,000 of wages...
-
Snail Creek Kennel, Inc., earns service revenue by caring for the pets of customers. Snail Creeks main expense is the salary paid to an employee. Requirement 1. Write the accounting equation for the...
-
Tech Support Services has the following unadjusted trial balance as of January 31, 2014. The debit and credit totals are not equal as a result of the following errors: a. The cash entered on the...
-
1. Identify some confusion about inflation 2. Explain the source of that confusion and the misconceptions that come about from it. 3. Select a tool, and explain how it works and how it is meant to...
-
What are the major causes of information risk? How can information risk be reduced?
-
A real estate agency would like to use artificial intelligence to better predict whether a certain customer will buy a specific house, so they can focus their efforts on promising potential sales....
-
Question 18 A Big Mac meal costs $3.00 and gives you an additional 5 units of utility; a meal at the Four Seasons Hotel costs $27.00 and gives you an additional 45 units of utility. Based only on the...
-
10. What is the consequence of neutropenia? A) Adhesion of leukocytes to microbes B) Production of large lysozomal granules C) Increased susceptibility to bacterial infections D) Inability to kill...
-
Mary has several trees in her back yard. She enjoys them verymuch as they make her back yard very appealing and most importantlythey provide her home with shade which results in lower utilitybills....
-
1. Which immunoglobulin is the largest in terms of size? A) Immunoglobulin G (IgG) B) Immunoglobulin M (IgM) C) Immunoglobulin A (IgA) D) Immunoglobulin D (IgD) 2. What properties do immunoglobulins...
-
15. What is the main cause of immunodeficiency in AIDS patients? A) Malnutrition B) Human immunodeficiency virus (HIV) infection C) Deficiency of C1 inhibitor D) Failure of helper T cells to produce...
-
XYZ Corporation has entered into an operating lease agreement with LMN Realty for warehouse space, The fair value of the warchouse space is Rs . 3 5 0 , 0 0 0 . The lease term is 5 years, and the...
-
For the next several days, take notes on your listening performance during at least a half-dozen situations in class, during social activities, and at work, if applicable. Referring to the traits of...
-
Solving the following road-map problem depends on determining the structure of A, the key intermediate. Give structures for compounds A through K. hept-1-yne SOCI CH3CuLi (2) (CHj,S (2) Ho HCN (2)...
-
Using the information in Figure 16-2, calculate the values of Ho for the following reactions: In Figure 16.2 (a) (b) (c) (-359 predicted) (-240 predicted) resonance enerty encrgy energy 240 kJ/mol232...
-
The following names are all incorrect or incomplete, but they represent real structures. Draw each structure and name it correctly. (a) 2-ethylpentane (b) 3-isopropylhexane (c)...
-
Derive the equation of motion of the system shown in Fig. 2.106, using the following methods: (a) Newton's second law of motion, (b) D'Alembert's principle, and (c) principle of virtual work. Uniform...
-
A mass \(m_{1}\) is attached at one end of a uniform bar of mass \(m_{2}\) whose other end is pivoted at point \(O\) as shown in Fig. 2.107. Determine the natural frequency of vibration of the...
-
The angular motion of the forearm of a human hand carrying a mass \(m_{0}\) is shown in Fig. 2.108. During motion, the forearm can be considered to rotate about the joint (pivot point) \(O\) with...

Study smarter with the SolutionInn App


