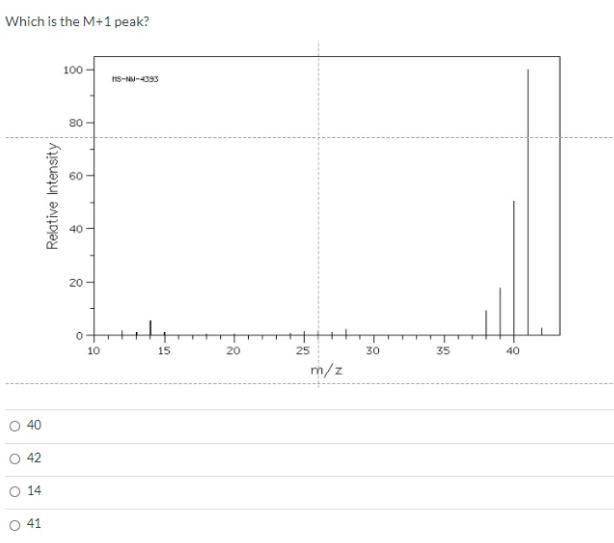
Which is the M+1 peak? O O 40 O 42 SO 14 41 100- 80 8...
Fantastic news! We've Found the answer you've been seeking!
Question:
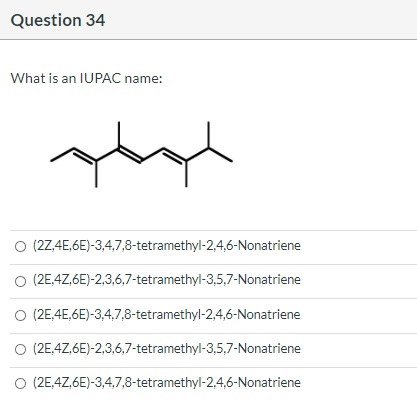
Transcribed Image Text:
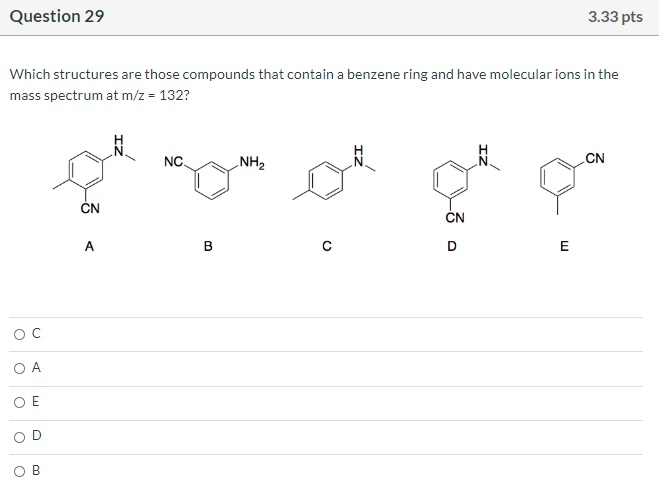
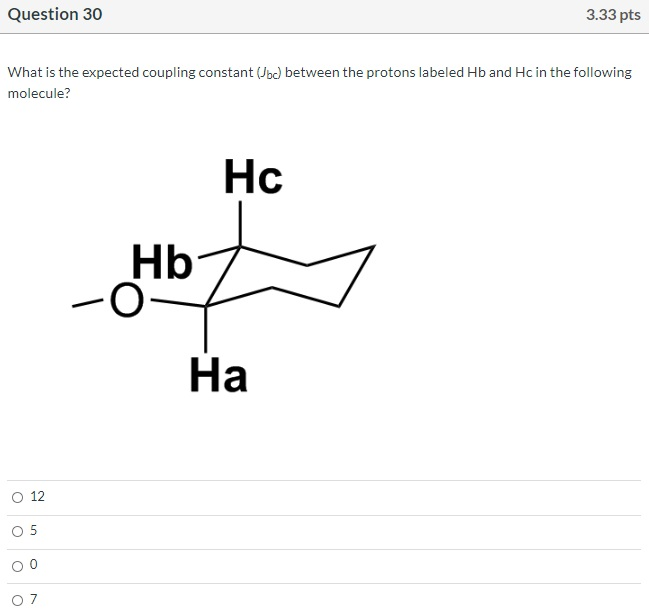
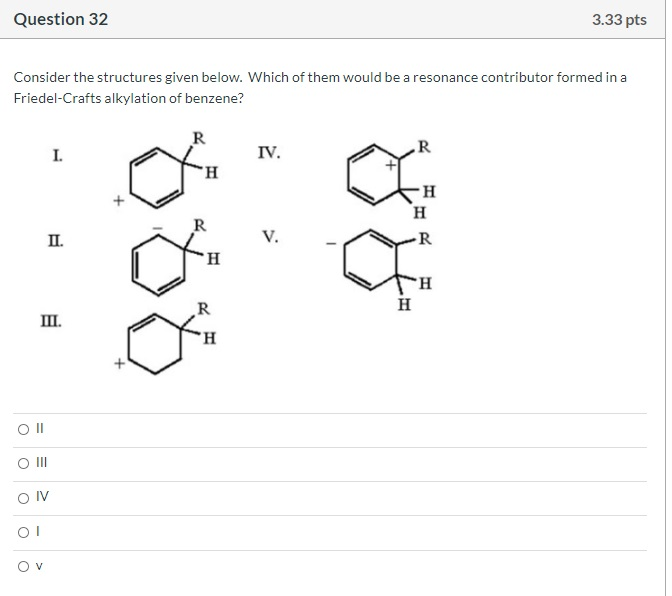
Which is the M+1 peak? O O 40 O 42 SO 14 41 100- 80 8 Relative Intensity 40 20- 10 MS-NM-4393 15 20 25 m/z 30 35 40 Question 29 Which structures are those compounds that contain a benzene ring and have molecular ions in the mass spectrum at m/z = 132? O C O A E CN < NC. 3.33 pts NH₂ CN gå quo CN E Question 30 What is the expected coupling constant (Jbc) between the protons labeled Hb and Hc in the following molecule? O 12 05 о о о Hb -O Hc 3.33 pts На Question 32 Consider the structures given below. Which of them would be a resonance contributor formed in a Friedel-Crafts alkylation of benzene? Oll O III OIV OI O I. III. V II. R R R IV. V. I R H H H R 3.33 pts H Question 34 What is an IUPAC name: (2Z,4E,6E)-3,4,7,8-tetramethyl-2,4,6-Nonatriene O (2E,4Z,6E)-2,3,6,7-tetramethyl-3,5,7-Nonatriene O (2E,4E,6E)-3,4,7,8-tetramethyl-2,4,6-Nonatriene (2E,4Z,6E)-2,3,6,7-tetramethyl-3,5,7-Nonatriene O (2E,4Z,6E)-3,4,7,8-tetramethyl-2,4,6-Nonatriene Which is the M+1 peak? O O 40 O 42 SO 14 41 100- 80 8 Relative Intensity 40 20- 10 MS-NM-4393 15 20 25 m/z 30 35 40 Question 29 Which structures are those compounds that contain a benzene ring and have molecular ions in the mass spectrum at m/z = 132? O C O A E CN < NC. 3.33 pts NH₂ CN gå quo CN E Question 30 What is the expected coupling constant (Jbc) between the protons labeled Hb and Hc in the following molecule? O 12 05 о о о Hb -O Hc 3.33 pts На Question 32 Consider the structures given below. Which of them would be a resonance contributor formed in a Friedel-Crafts alkylation of benzene? Oll O III OIV OI O I. III. V II. R R R IV. V. I R H H H R 3.33 pts H Question 34 What is an IUPAC name: (2Z,4E,6E)-3,4,7,8-tetramethyl-2,4,6-Nonatriene O (2E,4Z,6E)-2,3,6,7-tetramethyl-3,5,7-Nonatriene O (2E,4E,6E)-3,4,7,8-tetramethyl-2,4,6-Nonatriene (2E,4Z,6E)-2,3,6,7-tetramethyl-3,5,7-Nonatriene O (2E,4Z,6E)-3,4,7,8-tetramethyl-2,4,6-Nonatriene
Expert Answer:
Answer rating: 100% (QA)
Question 29 Compounds that contain a benzene ring and have molecul... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
question 3 20 marks z plc is considering a project which will necessitate the acquisition of a new machine to neutralize th
-
The data in Question 28 are converted to z scores here, thus allowing you to compute the standardized beta coefficients for each predictor variable from the data given in the previous question. Using...
-
Consider the experimental data: 15 20 25 30 35 40 33 37 37 40 45 50 41 5 10 y 17 24 31 40 42 Note: Report your numeric answers with at least 3 decimals. It is required to fit the data to a straight...
-
What will be the sum of third proportional to 18 and 54 and fourth proportional to 12, 18 and LCM of 12 and 18? 1. 162 2. 216 3. 612 4. 256 526 5.
-
Much of the public and private industry in Hawaii depends on tourism. The following time series plot shows the number of domestic visitors to Hawaii by air from the rest of the United States per...
-
a. Use the results of Example 7.5 to obtain a 95% lower confidence bound for the parameter l of an exponential distribution, and calculate the bound based on the data given in the example. b. If...
-
Scientists at NASA collected data to study which forces, including both natural and human factors, are responsible for the increase in observed temperature in the last two centuries. Go to http://www...
-
1. A patent increases the incentive to develop new products because it ________ the price of the product and thus generates profit to cover a firms costs of _______. 2. In some cases, a patent is...
-
Net income for June\ Withdrawals by Billie Soares in June\ Additional investment by Billie Soares in June\ 115,000\ 25,000\ 50,000 \ a. Prepare a statement of owner's equity for the month ended June...
-
A youth soccer club has contracted with Holiday Helpers, a local travel agency, to broker hotel rooms for out-of-town teams that have entered the club's Labor Day weekend soccer tournament. The...
-
21 Fitz Company reports the following information. Selected Annual Income Statement Data Net income Depreciation expense Amortization expense Gain on sale of plant assets Selected Year-End Balance...
-
2x The 22 inch driver's side windshield wiper moves through an angle of radians. The passenger's side windshield wiper moves through the same angle, but is one inch shorter. What is the difference of...
-
Competitor data Customer or supplier feedback Existing commitments Financial information from suppliers and key stakeholders Income, expenditure and performance of previous budgets Departmental,...
-
What is daily payroll if annual payroll is 244,458?
-
Can you provide different management decisions that could be made after performing further analysis of the financial ratios and trend analysis within this assignment?
-
1. Describe the elements that shape the permanent establishment, according to the model of the Organization for Economic Cooperation and Growth. 2. Describes the criteria to follow for the resolution...
-
Time Value of Money Exercises 1. Martin wants to invest $5,000. If the interest rate is 8% what will the value be after three years, a. (2 points) if simple interest is used to calculate the value?...
-
Show, if u(x, y) and v(x, y) are harmonic functions, that u + v must be a harmonic function but that uv need not be a harmonic function. Is e"e" a harmonic function?
-
Let the temperature T in a body be independent of z so that it is given by a scalar function T = T(x, t). Identify the isotherms T(x, y) = const. Sketch some of them. Sketch or graph isotherms of the...
-
Explain and compare the different roles of the two shifting theorems, using your own formulations and simple examples. Give no proofs.
-
Integrate by the first method or state why it does not apply and use the second method. Show the details. Re z dz, C the parabola y = 1+ (x 1)2 from 1+ i to 3 + 3i
-
The suitability concern discussed by Kroll and Park most likely deals with: A. governance. B. transparency. C. investment horizon. Kevin Kroll is the chair of the investment committee responsible for...
-
Which of Parks statements regarding the asset allocation approaches is correct? A. Only Statement 3 B. Only Statement 4 C. Both Statement 3 and Statement 4 Kevin Kroll is the chair of the investment...
-
Based on the forecasted environment, liquidity planning should take into account that general partners may: A. call capital at a slower pace. B. make distributions at a faster pace. C. exercise an...

Study smarter with the SolutionInn App


