Calculate the average atomic mass of a sample of oxygen with the masses and relative abundances...
Fantastic news! We've Found the answer you've been seeking!
Question:
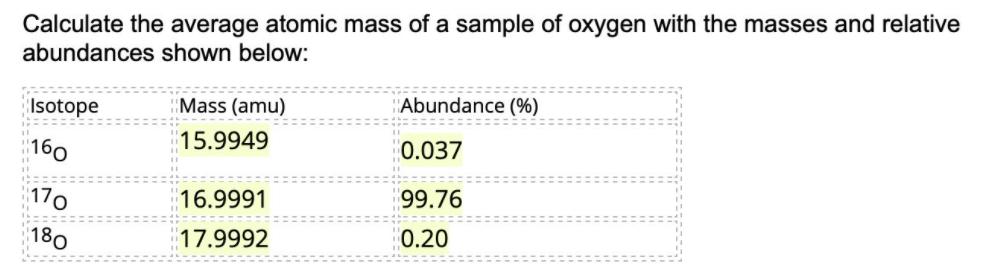
Transcribed Image Text:
Calculate the average atomic mass of a sample of oxygen with the masses and relative abundances shown below: Isotope Mass (amu) Abundance (%) 15.9949 160 0.037 170 16.9991 99.76 180 17.9992 0.20 Calculate the average atomic mass of a sample of oxygen with the masses and relative abundances shown below: Isotope Mass (amu) Abundance (%) 15.9949 160 0.037 170 16.9991 99.76 180 17.9992 0.20
Expert Answer:
Answer rating: 100% (QA)
The average atomic mass of an element is the sum the masses of its isotopes each multi... View the full answer

Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Posted Date:
Students also viewed these chemistry questions
-
A periodic table might list the average atomic mass of magnesium as being 24.312 u. That average value is the result of weighting the atomic masses of the magnesium isotopes according to their...
-
Calculate the average atomic mass of a sample of silicon has that has the following isotopes with the masses and relative abundances shown below: Isotope Mass (amu) Abundance (%) 28Si 27.9769 92.70...
-
Calculate the average atomic mass of a sample of argon has that has the following isotopes with the masses and relative abundances shown below: Isotope Mass (amu) Abundance (%) Ar 35.9675 0.3402 Ar...
-
As mentioned in the case, the joint venture between Renault and Mahindra & Mahindra (India) dissolved/failed. Speculate what are the possible socio-cultural issues that triggered this dissolution.
-
For n ¥ l, let X n , Y n and X, Y be r.v.s defined on the probability space (Ω, A, P), let d 1 , d 2 and c n be constants with 0 c n as n , and suppose that Let g: R 2 R be...
-
You are provided with the following transactions that took place during a recent fiscal year: Instructions Assuming the company is reporting under IFRS, complete the above table for each of the...
-
Describe an experiment, and give the necessary equations for measurement of the complex flexural modulus, \(E_{\mathrm{fx}}^{*}\), of a symmetric laminated beam.
-
Assume that the Turner, Roth, and Lowe partnership of Exercise 12-10 is a limited partnership. Turner and Roth are general partners and Lowe is a limited partner. How much of the remaining $28,000...
-
Find an example of a physical force from within the New Testament and discuss the example. Be sure to provide the scripture reference. Physical force means an event that occurred during biblical...
-
A camera is listed for $599 less 30%, 20%, and 5%. (a) What is the net price? (b) What is the total amount of discount allowed? (c) What is the exact single rate of discount that was allowed?
-
The US had an annual inflation rate of 11%, whereas Malaysia had an annual inflation rate of 9%. Currently, a Malaysian Ringgit can be exchanged for 3.2885 US dollars. Other things being equal, how...
-
A scissors jack for an automobile is shown below. The screw threads exert a force F on the blocks at joints A and B. If F = 700 N, determine the shearing stress on a cross section of the 10-mm-...
-
The Organization of the Petroleum Exporting Countries held a meeting due to rising oil prices caused by new sanctions on the Russian invasion of Ukraine. During the meeting, a proposal was introduced...
-
Explain whether option C is correct or not? Minimum Variance Hedge Ratio t = time at which hedge is put in place t = time at which hedge is closed out Sti spot price of asset being hedged at time t;...
-
In relation to its payables control account, at its year end of 30 April 20X1 Company A has discovered that: A contra of 85 with the receivables control account is required. Discount allowed to...
-
Pipe Supply Company offers its customers trade credit terms of 2/10 net 30. What does this mean?
-
Charla is at the end of the third year of a five-year investment. Charla initially invested $3,000. Over the past three years, her investment rates of return were -4.25%, -4% and 2.75% respectively....
-
One Way Cellular accountants have assembled the following data for the year ended September 30, 2014: Prepare the operating activities section using the indirect method for One Way Cellulars...
-
A 0.15 kg ball hits a wall with a velocity of (5.00 m/s)i + (6.50 m/s)j + (4.00 m/s)k. It rebounds from the wall with a velocity of (2.00 m/s)i + (3.50 m/s)j + (3.20 m/s)k. What are? (a) The change...
-
An object is moved along the central axis of a spherical mirror while the lateral magnification m of it is measured. Figure gives m versus object distance p for the range pa = 2.0 cm to pb = 8.0 cm....
-
A sinusoidal transverse wave of amplitude ym and wavelength travels on a stretched cord. (a) Find the ratio of the maximum particle speed (the speed with which a single particle in the cord moves...
-
(a) Complete a steady-state simulation of the vinyl-chloride process in Figure 2.6. First, create a simulation flowsheet. Assume that: Cooling water is heated from 30 to \(50^{\circ} \mathrm{C}\)...
-
For the monochlorobenzene separation process in Figure 7.14, the results of an ASPEN PLUS simulation are provided in the multimedia modules under ASPEN \( ightarrow\) Principles of Flowsheet...
-
Complete a simulation of the entire process for the hydrodealkylation of toluene in Figure 6.14. Initially, let the purge/recycle ratio be 0.25 ; then, vary this ratio and determine its effect on the...

Study smarter with the SolutionInn App


