For 14 points, match each of these reactions with their types. (SR) Single Replacement; (DR) Double...
Fantastic news! We've Found the answer you've been seeking!
Question:

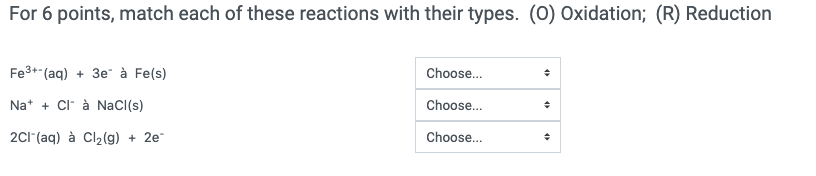
Transcribed Image Text:
For 14 points, match each of these reactions with their types. (SR) Single Replacement; (DR) Double Replacement; (DC) Decomposition; (CM) Combination; (X) Combustion. 2Nl3(s) à N₂(g) + 312(g) MgCl₂(aq) + Pb(NO3)2(aq) à PbCl₂(s) + Mg(NO3)2(g) 2V(s) + 3Cl₂(g) à 2VCI3(s) NaBr(s) + Li(s) à Na(s) + LiBr(s) C5H12 (s) 80₂(g) à CO₂(g + 6H₂O(g) KCI(s) + Nal(s) à NaCl(s) + Kl(s) 4Al(s) + 30₂ à 2Al₂O3(s) Choose... Choose... Choose... Choose... Choose... Choose... Choose... For 6 points, match each of these reactions with their types. (O) Oxidation; (R) Reduction Fe³+ (aq) + 3e à Fe(s) Na+ + Cl à NaCl(s) 2Cl(aq) à Cl₂(g) + 2e- Choose... Choose... Choose... + + + For 14 points, match each of these reactions with their types. (SR) Single Replacement; (DR) Double Replacement; (DC) Decomposition; (CM) Combination; (X) Combustion. 2Nl3(s) à N₂(g) + 312(g) MgCl₂(aq) + Pb(NO3)2(aq) à PbCl₂(s) + Mg(NO3)2(g) 2V(s) + 3Cl₂(g) à 2VCI3(s) NaBr(s) + Li(s) à Na(s) + LiBr(s) C5H12 (s) 80₂(g) à CO₂(g + 6H₂O(g) KCI(s) + Nal(s) à NaCl(s) + Kl(s) 4Al(s) + 30₂ à 2Al₂O3(s) Choose... Choose... Choose... Choose... Choose... Choose... Choose... For 6 points, match each of these reactions with their types. (O) Oxidation; (R) Reduction Fe³+ (aq) + 3e à Fe(s) Na+ + Cl à NaCl(s) 2Cl(aq) à Cl₂(g) + 2e- Choose... Choose... Choose... + + +
Expert Answer:
Answer rating: 100% (QA)
2NI3 N 3I2 D C Deumposhon MyCh Pb Ngl2 Pbc Mgng BR double h... View the full answer

Related Book For 

Holes Human Anatomy and Physiology
ISBN: 978-0078024290
14th edition
Authors: David Shier, Jackie Butler, Ricki Lewis
Posted Date:
Students also viewed these chemistry questions
-
Match each of these numbered descriptions with the term it best describes. Indicate your answer by writing the letter for the correct principle in the blank space next to each description. a. Cost...
-
Match the following terms with their definitions: ___ A. S Corp ___ B. Dissociation ___ C. Close corp ___ D. Dissolution ___ E. Limited partnership 1. The first step in the process of terminating a...
-
Match these three types of cash balances with the functions that they serve: a. Vault cash b. Deposits at the Federal Reserve c. Deposits at other FIs (1) Used to meet legal reserve requirements (2)...
-
Graph the function y = (x + |x|). At what values of x does the derivative exist and what is the value of the derivative when it does exist?
-
Discuss the legal implications involved in writing credit refusals.
-
Refer to the map in Figure 8.8 on page 346. Sketch possible graphs of the predicted high temperature on a north-south line and an east-west line through Boise. 60s Boise 70s 80s 90s 60s 100s 80s 90s...
-
4. Explain the difference between judicial restraint activism and judicial activism in contract law.
-
Each autumn, as a hobby, Anne Magnuson weaves cotton place mats to sell through a local craft shop. The mats sell for $20 per set of four. The shop charges a 10% commission and remits the net...
-
Bula Investments acquired $260,400 of Effenstein Corp., 10% bonds at their face amount on October 1, 20Y1. The bonds pay interest on October 1 and April 1. On April 1, 20Y2, Bula sold $67,600 of...
-
Table 1 shows Apple's online orders for the last week. When shoppers place an online order, several "recommended products" (upsells) are shown as at checkout an attempt to upsell See table 2 in cell...
-
Avicorp has a $11.7 million debt issue outstanding, with a 5.9% coupon rate. The debt has semi-annual coupons, the next coupon is due in six months, and the debt matures in five years. It is...
-
A thin rigid wire of mass M and length L = 1 m rotates about a vertical axis passing through its centre, as illustrated in the diagram below. You may assume its moment of inertia is = ML/12. The wire...
-
Bheksizwe (Pty) Ltd is a company that specializes in the production of custom made mountain bikes, Bhekisizwe (Pty) Luis a micro bossiness and qualifies for all of the advantages of turnover tux. The...
-
1 If 83 cubic inches of O 2 gas at 56 psig is allowed to expand to 354 cubic inches w/o changing its temperature, what will be the final pressure of the O 2 gas expressed as a gauge pressure? 2. In a...
-
Tom spent his entire life in Australia until he ceased to be an Australian resident and became a resident of the United Kingdom in June 2 0 2 3 . When he left Australia, Tom s only asset was a...
-
11. As a hot hands pilot you get to fly an aircraft with air refueling capability. You take on fuel from a tanker and your weight before refueling was 30,000 lbs and you take on 5000 lbs of fuel. If...
-
In the context of the methods used to set an advertising budget, identify a true statement about the percentage of sales or profits method. Multiple choice question. Marketing managers decide how...
-
Currently, there are five concepts of food stands, including: hot dogs, soft pretzels, turkey legs, sandwich wraps, and funnel cakes. This approach will double the existing number of food stands...
-
List five factors that control when and if a cell divides.
-
Discuss the mechanism of swallowing.
-
Explain why DNA replication is essential.
-
For the following, determine if the given point is a removable singularity, an essential singularity, or a pole (indicate its order). a. \(\frac{1-\cos z}{z^{2}}, \quad z=0\). b. \(\frac{\sin...
-
Find the Laurent series expansion for \(f(z)=\frac{\sinh z}{z^{3}}\) about \(z=0\). [You need to first do a MacLaurin series expansion for the hyperbolic sine.]
-
Find the residues at the given points: a. \(\frac{2 z^{2}+3 z}{z-1}\) at \(z=1\). b. \(\frac{\ln (1+2 z)}{z}\) at \(z=0\). c. \(\frac{\cos z}{(2 z-\pi)^{3}}\) at \(z=\frac{\pi}{2}\).

Study smarter with the SolutionInn App


