Which of the following is NOT true about school leavers in Canada? they are more likely...
Fantastic news! We've Found the answer you've been seeking!
Question:

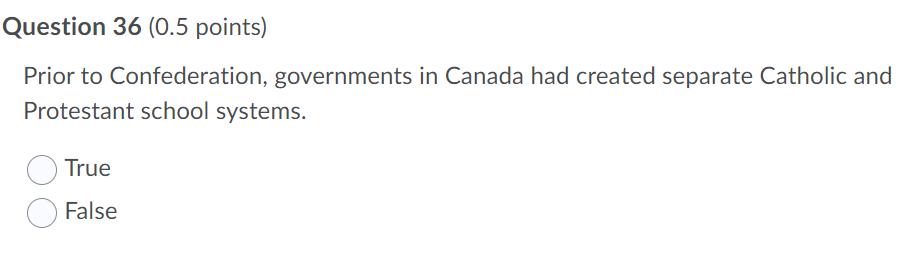

Transcribed Image Text:
Which of the following is NOT true about school leavers in Canada? they are more likely to be from single-parent families they are more likely to have failed a grade in elementary school they are more likely to be working more than 20 hours a week during schod they are more likely to have drug or alcohol addictions Question 36 (0.5 points) Prior to Confederation, governments in Canada had created separate Catholic and Protestant school systems. True False Question 35 (0.5 points) The social institution by which society provides people with important knowledge, including basic facts and information, job skills, and cultural norms and values is the family. the church. the peer group. education. Which of the following is NOT true about school leavers in Canada? they are more likely to be from single-parent families they are more likely to have failed a grade in elementary school they are more likely to be working more than 20 hours a week during schod they are more likely to have drug or alcohol addictions Question 36 (0.5 points) Prior to Confederation, governments in Canada had created separate Catholic and Protestant school systems. True False Question 35 (0.5 points) The social institution by which society provides people with important knowledge, including basic facts and information, job skills, and cultural norms and values is the family. the church. the peer group. education.
Expert Answer:
Answer rating: 100% (QA)
Which of the following is not true about school leavers in Canada They are more likely to be from si... View the full answer

Related Book For 

Income Tax Fundamentals 2019
ISBN: 9781337703062
37th edition
Authors: Gerald E. Whittenburg, Steven Gill
Posted Date:
Students also viewed these psychology questions
-
Which of the following is not true about an ideal gas? (a) The average kinetic energy of the gas molecules increases as the temperature increases. (b) The volume of an ideal gas increases with...
-
Access to mobile phone has re-shaped the financial services and commerce, especially during COVID-19 pandemic. The society are adapting to conduct transaction through mobile phone because there are...
-
Which of the following is not true of white blood cells? a. Formed in red bone marrow b. Carry oxygen and carbon dioxide c. Can leave the bloodstream and enter tissues d. Can fight disease and...
-
Answer the Multple Choice Questions and the code for problem 6in the end PROBLEM 1: General UNIX 1. What is UNIX? a) an operating system b) a text editor c) programming language d) software program...
-
Preparing the statement of retained earnings Prepare the statement of retained earnings of Decorating Arrangements for the year ended December 31, 2016. Insurance Expense....... $ 2,000 Service...
-
Obtain the Thevenin equivalent circuit for the circuit in Fig. 13.83 at terminals a-b. j2 52 3 . 10290 V (2 2 4/0 A
-
One year ago, Bioette, a biotech incubator, entered into a forward contract to sell one of its patents to Pharm, a major drug company, in 2 years for \(\$ 10\) million. Currently, with only 1 year...
-
Simon Products uses both special journals and a general journal as described in this chapter. Simon also posts customers? accounts in the accounts receivable subsidiary ledger. The postings for the...
-
Rubber and Steel Company is planning to manufacture a new product. The variable manufacturing costs will be $57 per unit and the fixed costs are estimated to be $6819 The selling price of the product...
-
Give a correct name for a. (CH3)2CHCH2OCH2CH3 b. c. O- CH2CH2CH,
-
Which of the following statements are true of converging lenses? Select all that apply A. Converging lenses are thicker at the center than they are at the edges B.Converging lenses have a negative...
-
OH&S programs can be implemented by following two very different approaches. One approach is to "force" compliance by establishing standards, conducting rigorous inspections on a regular basis and...
-
Temple City California Comprehensive Pan How does it Work? What purpose does it serve? What are the effects and second-order consequences? Describe the necessary primary stakeholders to have a...
-
Discuss the staffing planning process as well as the workforce planning process and how it impacts future business activities. Explain steps taken for an organization to forecast its workforce supply...
-
Mention some Communications issues in a workplace. Mention the impact of generational differences and evolving technologies on organizational communications. What strategies could one employ in...
-
develop these point with int-text citations and reference list. The risks of transferring non-physician personnel from the clinics to be closed to those that will remain open, and an assessment of...
-
What is included in a Business Case? O business requirements stakeholder requirements solution requirements functional and nonfunctional requirements transition requirements O feasibility study...
-
Which of the following statements is false? a. Capital leases are not commonly reported in a Capital Projects Fund. b. A governmental entity may report a Capital Project Fund in one year but not the...
-
Bob sells a stock investment for $35,000 cash, and the purchaser assumes Bobs $32,500 debt on the investment. The basis of Bobs stock investment is $55,000. What is the gain or loss realized on the...
-
If a taxpayer has beginning inventory of $25,000, purchases of $175,000, and ending inventory of $45,000, what is the amount of the cost of goods sold for the current year? a. $155,000. b. $180,000...
-
Helen, a single taxpayer, has modified adjusted gross income (before passive losses) of $126,000. During the tax year, Helens rental house generated a loss of $15,000. Assuming Helen is actively...
-
How many oxygen atoms are bonded to the carbon of the carbonyl of an ester functional group? (a) none (b) one (c) two (d) three
-
Explain why caprylic acid, CH 3 (CH 2 ) 6 COOH, dissolves in a 5, aqueous solution of sodium hydroxide but caprylaldehyde, CH 3 (CH 2 ) 6 CHO, does not dissolve. (a) With two oxygens, the caprylic...
-
Which would you expect to be more viscousa polymer made of long molecular strands or a polymer made of short molecular stands? Why? (a) long strands, because they tend to tangle among themselves (b)...

Study smarter with the SolutionInn App


