Pure caffeine has a melting point of around 235 C. Suppose after you isolated your caffeine...
Fantastic news! We've Found the answer you've been seeking!
Question:
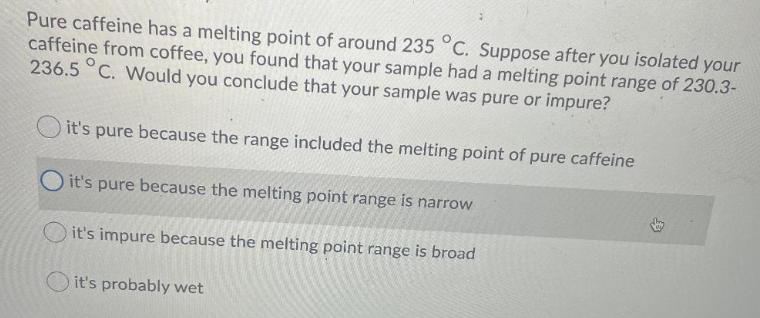
Transcribed Image Text:
Pure caffeine has a melting point of around 235 °C. Suppose after you isolated your caffeine from coffee, you found that your sample had a melting point range of 230.3- 236.5 °C. Would you conclude that your sample was pure or impure? it's pure because the range included the melting point of pure caffeine Oit's pure because the melting point range is narrow it's impure because the melting point range is broad it's probably wet Pure caffeine has a melting point of around 235 °C. Suppose after you isolated your caffeine from coffee, you found that your sample had a melting point range of 230.3- 236.5 °C. Would you conclude that your sample was pure or impure? it's pure because the range included the melting point of pure caffeine Oit's pure because the melting point range is narrow it's impure because the melting point range is broad it's probably wet
Expert Answer:
Answer rating: 100% (QA)
Question 391 has a sharp melt A Compound material shapp melting point if the mange of m... View the full answer

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
Tristearin has a melting point of 72 C. Based on this information, would you expect triarachadin to be classified as a fat or as an oil?
-
If copper (which has a melting point of 1085C) homogeneously nucleates at 849C, calculate the critical radius given values of 1.77 109 J/m3 and 0.200 J/m2, respectively, for the latent heat of...
-
Benzene (C6H6) has a melting point of 5.5oC and an enthalpy of fusion of 10.04 kJ/mol at 25.0oC. The molar heat capacities at constant pressure for solid and liquid benzene are 100.4 JK-1mol-1 and...
-
A major electronics manufacturer expects to generate additional revenue from its recently won government contract. The company forecasts that the revenue will be $190 million in the first year, but...
-
Consider a country that imports a good from abroad. For each of following statements, say whether it is true or false. Explain your answer. a. The greater the elasticity of demand, the greater the...
-
If the VF Corporation deposits $20 million of its retained earnings in an aggressive stock ownership fund for only 1 year, what will be the difference in the total amount accumulated (i.e., F) at 18%...
-
Use the summary statistics in Exercise 12 to test the null hypothesis H0: 1 = 0 versus H1: 1 > 0. Use the = 0.05 level of significance.
-
Soft phonon mode consider a line of ions of equal mass but alternating in charge, with e p = e(?1) p as the charge on the pth ion the inter atomic potential is the sum of two contributions (1) a...
-
Each year, Worrix Corporation manufactures and sells 3,700 premium-quality multimedia projectors at $12,700 per unit. At the current production level, the firm's manufacturing costs include variable...
-
Photo Tonight, a film-developing and camera-repair franchise, began business on January 1, 20X1. In the process of beginning operations, it incurred the following capital expenditures: Developing...
-
Dillingham, Inc., has a 60-day collection period. The projected sales for each quarter next year are shown below. What are collections for the third quarter? Q1 Q2 Q3 Q4 Sales $ 3720 $ 4320 $ 4725 $...
-
b. How much interest is paid in the third year? (Do not round intermediate calculations and round your answer to 2 decimal places, e.g., 32.16.) c. How much total interest is paid over the life of...
-
Since 2000, two different towns had populations that changed exponentially. Town A's population was 42,680 at the beginning of 2000 and changed by 2.8% per year. Town B's population was 57,920 at the...
-
The following will provide you with verification of Shell's law. Indeed, there must be some relation between the indexes of refraction and the amount of light bending. Use setup A from Part B. 1....
-
Ball Bearing Process Capability: A ball bearing manufacturing process has a target value of 9 for the diameter, and the design specifications specify a tolerance of +/- 0.22. If the process...
-
Using the definition of the logarithm, what expression should go in the blank below? 93 = 729 3
-
Distributed computing provides a means for complex problems to be solved efficiently and quickly by dividing the work among the processors in the system. This concept is interesting because of its...
-
What can you do to reduce hunger where you live? To reduce hunger globally?
-
Which alkyl halide would you expect to react more rapidly by an SN2 mechanism? Explain your answer. (a) (b) (c) (d) (e) Br r or or CI
-
Write the structures of the three products involved in this reaction sequence: Spectral data for B: MS (m/z): 314, 312, 310 (relative abundance 1:2:1) 1H NMR (δ): only 6.80 (s) after...
-
Triethylamine, (C2H5)3N, like all amines, has a nitrogen atom with an unshared pair of electrons. Dichlorocarbene also has an unshared pair of electrons. Both can be represented as shown below. Draw...
-
Why is the thermodynamic process also called cyclic process? Explain with suitable example.
-
A mercury manometer reads \(40 \mathrm{~cm}\) at \(30^{\circ} \mathrm{C}\) and \(1 \mathrm{~atm}\). Determine the absolute pressure, given that \(ho_{\mathrm{Hg}}=14.02 \mathrm{~g} /...
-
Define isothermal, isochoric and isobaric processes with suitable examples.

Study smarter with the SolutionInn App


