For a binary system consists of the cyclopentane (species 1) and cyclohexane (species 2), this binary...
Fantastic news! We've Found the answer you've been seeking!
Question:
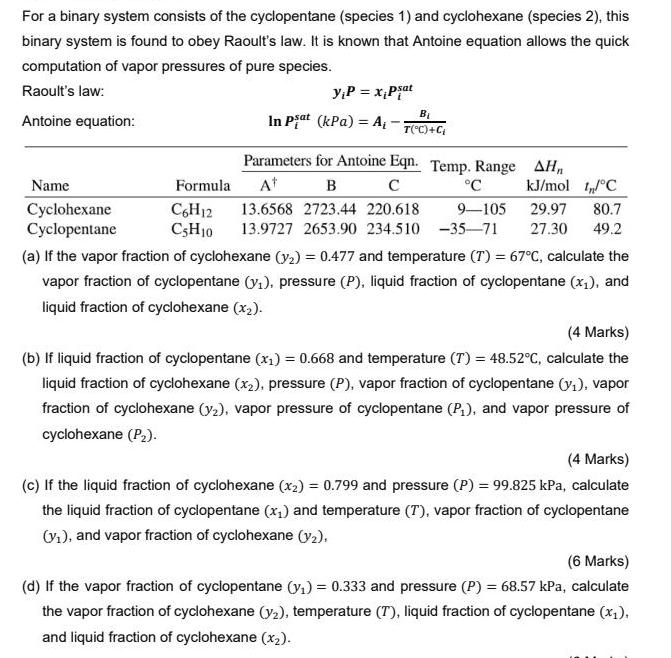
Transcribed Image Text:
For a binary system consists of the cyclopentane (species 1) and cyclohexane (species 2), this binary system is found to obey Raoult's law. It is known that Antoine equation allows the quick computation of vapor pressures of pure species. Raoult's law: yP = x,Pjat Antoine equation: In Piat (kPa) = A, - T("C)+C Parameters for Antoine Eqn. Temp. Range AHn Name Formula At в °C kJ/mol I°C Cyclohexane Cyclopentane C,H12 CSH10 13.6568 2723.44 220.618 9-105 29.97 80.7 13.9727 2653.90 234.510 -35-71 27.30 49.2 (a) If the vapor fraction of cyclohexane (y2) = 0.477 and temperature (T) = 67°C, calculate the vapor fraction of cyclopentane (y,), pressure (P), liquid fraction of cyclopentane (x1), and liquid fraction of cyclohexane (x2). (4 Marks) (b) If liquid fraction of cyclopentane (x,) = 0.668 and temperature (T) = 48.52°c, calculate the liquid fraction of cyclohexane (x2), pressure (P), vapor fraction of cyclopentane (y1), vapor fraction of cyclohexane (y2), vapor pressure of cyclopentane (P,), and vapor pressure of cyclohexane (P2). (4 Marks) (c) If the liquid fraction of cyclohexane (x2) = 0.799 and pressure (P) = 99.825 kPa, calculate the liquid fraction of cyclopentane (x,) and temperature (T), vapor fraction of cyclopentane Vi), and vapor fraction of cyclohexane (y2). (6 Marks) (d) If the vapor fraction of cyclopentane (y,) = 0.333 and pressure (P) = 68.57 kPa, calculate the vapor fraction of cyclohexane (y2), temperature (T), liquid fraction of cyclopentane (x;), and liquid fraction of cyclohexane (x2). For a binary system consists of the cyclopentane (species 1) and cyclohexane (species 2), this binary system is found to obey Raoult's law. It is known that Antoine equation allows the quick computation of vapor pressures of pure species. Raoult's law: yP = x,Pjat Antoine equation: In Piat (kPa) = A, - T("C)+C Parameters for Antoine Eqn. Temp. Range AHn Name Formula At в °C kJ/mol I°C Cyclohexane Cyclopentane C,H12 CSH10 13.6568 2723.44 220.618 9-105 29.97 80.7 13.9727 2653.90 234.510 -35-71 27.30 49.2 (a) If the vapor fraction of cyclohexane (y2) = 0.477 and temperature (T) = 67°C, calculate the vapor fraction of cyclopentane (y,), pressure (P), liquid fraction of cyclopentane (x1), and liquid fraction of cyclohexane (x2). (4 Marks) (b) If liquid fraction of cyclopentane (x,) = 0.668 and temperature (T) = 48.52°c, calculate the liquid fraction of cyclohexane (x2), pressure (P), vapor fraction of cyclopentane (y1), vapor fraction of cyclohexane (y2), vapor pressure of cyclopentane (P,), and vapor pressure of cyclohexane (P2). (4 Marks) (c) If the liquid fraction of cyclohexane (x2) = 0.799 and pressure (P) = 99.825 kPa, calculate the liquid fraction of cyclopentane (x,) and temperature (T), vapor fraction of cyclopentane Vi), and vapor fraction of cyclohexane (y2). (6 Marks) (d) If the vapor fraction of cyclopentane (y,) = 0.333 and pressure (P) = 68.57 kPa, calculate the vapor fraction of cyclohexane (y2), temperature (T), liquid fraction of cyclopentane (x;), and liquid fraction of cyclohexane (x2).
Expert Answer:
Related Book For 

Introduction to Chemical Engineering Thermodynamics
ISBN: 978-0071247085
7th edition
Authors: J. M. Smith, H. C. Van Ness, M. M. Abbott
Posted Date:
Students also viewed these chemical engineering questions
-
For a binary system derive the expression for HE implied by the Wilson equation for GE/RT. Show that the implied excess heat capacity CEP is necessarily positive. Recall that the Wilson parameters...
-
Our number system consists of the digits 0, 1, 2, 3, 4, 5, 6, 7, 8, and 9. The first significant digit in any number must be 1, 2, 3, 4, 5, 6, 7, 8, or 9 because we do not write numbers such as 12 as...
-
P-V-T behavior of a simple fluid is found to obey the equation of state given in problem 8.14. (a) Derive a formula for the enthalpy departure for the fluid. (b) Determine the enthalpy departure at...
-
1) The following information relates to the ABCs depreciable assets. Class 1 - During 2020, a new office building was acquired at a total cost of $623,000. Of this total, it is estimated that the...
-
Why would perpetual inventory records be preferable to periodic inventory records in a manufacturing company?
-
The 2-kg rod AB and the 3-kg rod BC are connected as shown to a disk that is made to rotate in a vertical plane. Knowing that at the instant shown the disk has an angular acceleration of 18 rad/s2...
-
Assume that the following regression model was applied to historical quarterly data: Assume that the regression coefficients were estimated as follows: Also assume that the inflation differential in...
-
While auditing the financial statements of Petty Corporation, the certified public accounting firm of True blue and Smith discovered that its clients legal expense account was abnormally high....
-
pts) Find the quotient and remainder using polynomial long division: 4x -9x+1 x-2
-
The following table summarizes the operating results for Bene Petits first year of operations: Bene Petit First year operating data: Single (1 serving) Dual (2 servings) Family (4 servings) Total...
-
If you are borrowing $15,000 at 8% interest rate, compounded yearly. Amortize the loan if payments are made for 4 years.
-
You are the risk management officer for a large financial services company. One of the company's major clients, a multinational corporation, has just suffered acyber attackthat has compromised their...
-
3. Consider the following matrix 3 0 A = -1 1 0 (a) Calculate all of the eigenvalues of A. (b) For each eigenvalue of A found in (a), find a basis for the corresponding eigenspace. (c) Determine...
-
1. Find the truth values for the following compound statements, given that P, R and S are true, while Q is false. (PVR) (SAQ) (PQ) (SAR) 3. Show that the following is a valid argument form (PVQ) R R...
-
A group of people were asked to vote on their favorite pet. Use the results from the following poll to answer the questions. (If there is a tie for 1st place, select all winners.) Number of votes..>...
-
How do you respond to this post and ask a question to keep the discussion going? 1. Why does health care spending continue to grow in the U.S.? Consider specific, demonstrable reasons, as opposed to...
-
31, 5. Warranties. In the calendar year 2020, Golden Enterprises sold 4,000 units of its product for P10,000 each. The selling price included a one-year warranty on parts. It is expected that 3% of...
-
Show that gj concave AHUCQ Abadie For nonnegative variables, we have the following corollary.
-
Isobutane expands adiabatically in a turbine from 5,000 kPa and 250oC to 500 kPa at the rate of 0.7 kg mol s-1. If the turbine efficiency is 0.80, w hat is the power output of the turbine and what is...
-
One cubic meter of an ideal gas at 600 K and 1,000 kPa expands to five times its initial volume as follows; (a) By a mechanically reversible, isothermal process. (b) By a mechanically reversible,...
-
Calculate Z. HR and SR by the $oavc / Rcdlich / Kwong equation for the substance and conditions given by one of the parts of Pb. 6.14, and compare results with values found from suitable generalized...
-
Distinguish among the three types of responsibility
-
Using the information in E7-1, assume that in July 2002, Voss Company incurs the following manufacturing overhead costs. Instructions (a) Prepare a flexible budget performance report, assuming that...
-
Samano Company uses flexible budgets to control its selling expenses. Monthly sales are expected to range from \($170,000\) to \($200,000\). Variable costs and their percentage relationship to sales...

Study smarter with the SolutionInn App



