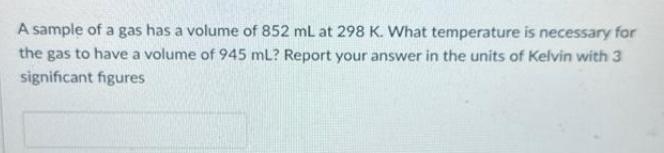
A sample of a gas has a volume of 852 mL at 298 K. What temperature...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A sample of a gas has a volume of 852 mL at 298 K. What temperature is necessary for the gas to have a volume of 945 mL? Report your answer in the units of Kelvin with 3 significant figures A sample of a gas has a volume of 852 mL at 298 K. What temperature is necessary for the gas to have a volume of 945 mL? Report your answer in the units of Kelvin with 3 significant figures
Expert Answer:
Answer rating: 100% (QA)
This question relates to Charless Law which states that at constant pressure and amount of gas ... View the full answer

Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Posted Date:
Students also viewed these chemical engineering questions
-
Your retirement goal is to have enough in savings to be able to pay yourself $150,000 per year for 20 years. If you expect to earn 4% on your savings during retirement, how much will you need in...
-
7. A sample of oxygen gas has a volume of 545 mL at 35 degree C. The gas is heated to 151 degree C at constant pressure in a container that can contract or expand. What is the final volume of the...
-
1. The following table contains closing monthly stock prices for Oracle Corporation (ORCL), Microsoft Corporation (MSFT), and NVidia (NVDA) for the first half of 2017. Ticker 6/30/2017 5/31/2017...
-
Indicate identities used n cot20-1 Prove: cot 20 2cot0
-
Gloria Varay, MD, opened a medical practice. The business completed the following transactions: After these transactions, how much cash does the business have to work with? Use a T account to show...
-
Give examples of private company costs that might be understated, and explain why.
-
3. Fifteen-year-old Terri Stubblefield was riding in the back seat of a Ford Mustang II when the car was hit from behind. The Mustang was engulfed in a ball of fire, and Terri was severely burned....
-
Outdoor Life manufactures snowboards. Its cost of making 2,000 bindings is as follows: Suppose Lancaster will sell bindings to Outdoor Life for $14 each. Outdoor Life would pay $3 per unit to...
-
Here is my assignment no 01 of Principal of Accounting i want to have a proper solution for my assignment. Thnkew. Q.1 xyz company provides transportation between major airports in various business...
-
You have been assigned to analyze the year-end inventory of Dynomil Company. This company sells three items of inventory and tracks of the average purchase cost and average selling price each month....
-
Newel1 Corp. sells household cleaners producing a revenue stream that has remained unchanged in the last few years. The firm does not expect any change in its sales or earnings in the next several...
-
SCR 1845-6357 A is a faint red dwarf star, 163 LY from us, with a wavelength of maximum emission at 1132 nm. What is the surface temperature of this star? Compare the surface temperature of this star...
-
How do educational institutions contribute to the reproduction of social hierarchies and stratification in contemporary societies ?
-
Answer the questions below in a 250-word review of the memo and paper below: Does the introduction clearly announce the topic and engage the readers interest? If not, why not? If yes, what is...
-
Beth Company sold assets to Karen Company with an alleged value of $ 2 , 4 0 0 , 0 0 0 . Beth Company paid $ 2 , 1 0 0 , 0 0 0 for the assets. The actual value of the assets was $ 1 , 7 0 0 , 0 0 0 ....
-
Given the program: importjava.util.Stack; public class SweetCandy { public static void main(String [ ] args) { Stack container = new Stack( ); container.push (new Candy("Purple")); container.push...
-
there are 2322 students in a school district. In a sample of 463 students from this school district, 170 have a mathematics scores below grade level. Based on the sample statistics, estimate the...
-
Annual dividends of ATTA Corp grew from $0.96 in 2005 to $1.76 in 2017. What was the annual growth rate?
-
The linear density of a string is l6 x 10-4 kg/m. A transverse wave on the string is described by the equation y = (0.021 m) sin [(2.0m-1)x + (30 s-1)t]. What are? (a) The wave speed and (b) The...
-
On a dry road, o car with good tires may be able to brake with a constant deceleration of 4.92 m/s 2 . (a) How long does such a car, initially traveling at 24.6 m/s, take to stop? (b) How far does it...
-
In Figure, a force F of magnitude 12 N is applied to a FedEx box of mass m2 = 1.0 kg. The force is directed up a plane tilted by = 37o. The box is connected by a cord to a UPS box of mass m1 = 3.0...
-
From the following selected balances of State plc as at 31 December 2008, prepare (j) a detailed trading and profit and loss account for the year ended 31 December 2008 for internal use and (ii) a...
-
The following information has been extracted from the books of account of Rufford plc for the year to 31 March 2006: Additional information: : 1. Administrative expenses include the following items:...
-
From the following balances in the books of Breaker plc you are to draw up (i) a detailed trading and profit and loss account for the year ending 31 March 2004 for internal use, and (ii) a profit and...

Study smarter with the SolutionInn App


