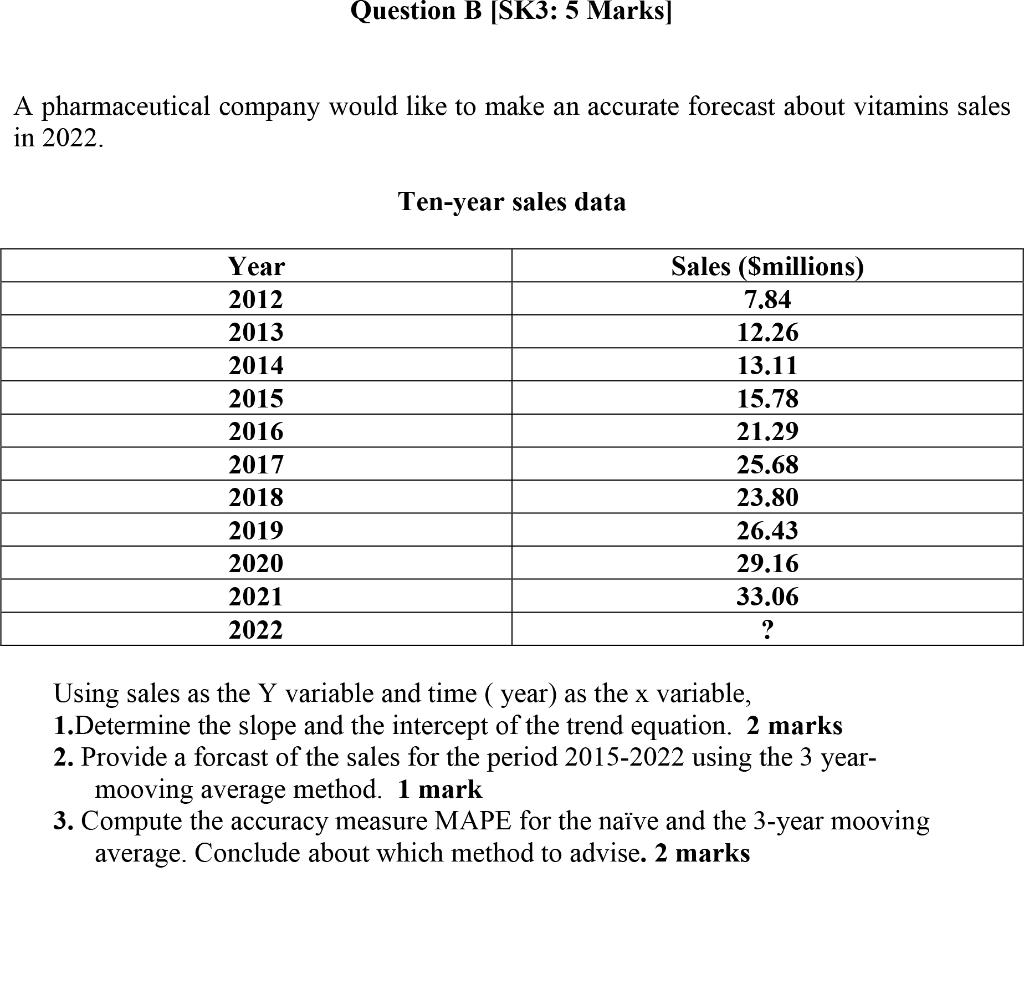
Question B [SK3: 5 Marks] A pharmaceutical company would like to make an accurate forecast about...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Question B [SK3: 5 Marks] A pharmaceutical company would like to make an accurate forecast about vitamins sales in 2022. Ten-year sales data Year Sales (Smillions) 2012 7,84 2013 12.26 2014 13.11 2015 15.78 2016 21.29 2017 25.68 2018 23.80 2019 26.43 2020 29.16 2021 33.06 2022 Using sales as the Y variable and time ( year) as the x variable, 1.Determine the slope and the intercept of the trend equation. 2 marks 2. Provide a forcast of the sales for the period 2015-2022 using the 3 mooving average method. 1 mark 3. Compute the accuracy measure MAPE for the naïve and the 3-year mooving average. Conclude about which method to advise. 2 marks уear- Question B [SK3: 5 Marks] A pharmaceutical company would like to make an accurate forecast about vitamins sales in 2022. Ten-year sales data Year Sales (Smillions) 2012 7,84 2013 12.26 2014 13.11 2015 15.78 2016 21.29 2017 25.68 2018 23.80 2019 26.43 2020 29.16 2021 33.06 2022 Using sales as the Y variable and time ( year) as the x variable, 1.Determine the slope and the intercept of the trend equation. 2 marks 2. Provide a forcast of the sales for the period 2015-2022 using the 3 mooving average method. 1 mark 3. Compute the accuracy measure MAPE for the naïve and the 3-year mooving average. Conclude about which method to advise. 2 marks уear-
Expert Answer:
Answer rating: 100% (QA)
1 LINEAR FORCASTING X Y YEAR Period Sales million 2012 1 784 2013 2 1226 2014 3 1311 2... View the full answer

Related Book For 

Cambridge International AS & A Level Physics Coursebook
ISBN: 9781108859035
3rd Edition
Authors: David Sang, Graham Jones, Gurinder Chadha, Richard Woodside
Posted Date:
Students also viewed these accounting questions
-
A global investment company would like to make an investment on several Oil and Gas Companies in the United States. Total annual expected return (in thousands) and cost for block of shares...
-
The Chewy Candy Company would like to determine an aggregate production plan for the next six months. The company makes many different types of candy but feels it can plan its total production in...
-
Y = x + 1 Determine the slope and the y-intercept. Then use the slope and the y-intercept to graph the equation by hand.
-
The following data represent the concentration of dissolved organic carbon (mg/L) collected from 20 samples of organic soil. Assume that the population is normally distributed. Complete parts (a)...
-
On December 31, 20X1, a company purchased $1 million of 10-year, 10% debentures for $885,300. The market interest rate was 12%. 1. Using the balance sheet equation format, prepare an analysis of bond...
-
For each of the following lists of premises, derive the indicated conclusion and complete the justification. In problems 4 and 8 you can add any statement you choose.
-
A smooth, flat plate of length \(\ell=6 \mathrm{~m}\) and width \(b=4 \mathrm{~m}\) is placed in water with an upstream velocity of \(U=0.5 \mathrm{~m} / \mathrm{s}\). Determine the boundary layer...
-
Stylz Company, a recent start-up fashion retailer based in the United States, is deciding between opening its first sales presence in either Italy's Tuscany Region or Spain's Matarrana Region....
-
K Let f(x)=2x+12x - 30x-9. a. Find all points on the graph of f(x) at which the tangent line is horizontal. b. Find all points on the graph of f(x) at which the tangent line has slope - 48.
-
Marias Food Service provides meals that nonprofit organizations distribute to handicapped and elderly people. Here is her forecasted income statement for April, when she expects to produce and sell...
-
The following data is given for the Bahia Company: Budgeted production Actual production Materials: Standard price per pound Standard pounds per completed unit Actual pounds purchased and used in...
-
Which of the following statements is false? a. GAAP specifically requires use of a multiple-step income statement. b. Under GAAP, companies can use either a perpetual or periodic system. c. IFRS does...
-
Which of the following is the correct accounting under GAAP for cash? a. Cash cannot be combined with cash equivalents. b. Demand deposits are not included as cash. c. Cash equivalents are...
-
In a perpetual inventory system: a. specific identification is always used. b. average costs are computed as a simple average of unit costs incurred. c. a new average is computed under the...
-
As a result of a thorough physical inventory, Railway Ltd. determined that it had inventory worth 180,000 at December 31, 2025. This count did not take into consideration the following facts. Rogers...
-
Lee Athletics overstated its inventory by NT$500,000 at December 31, 2024. It did not correct the error in 2024 or 2025. As a result, Lees equity was: a. overstated at December 31, 2024, and...
-
Schimmel Company provides the following information about its single product. Targeted operating $35,000 income Selling price per unit $7.85 Variable cost per unit $6.10 $96,250 Total fixed cost What...
-
The Thomas Corporation was organized on Jan. 1, 2020. On Dec. 31, 2021, the corporation lost most of its inventory in a warehouse fire before the year-end count of inventory was to take place. just...
-
Explain why an aircraft will tend to lose height when banking, unless the pilot increases its speed to provide more lift.
-
A 1.0 mW laser produces red light of wavelength 6.48 10 7 m. Calculate how many photons the laser produces per second. To answer questions you will need these values: Speed of light in a vacuum c =...
-
Use the graphs shown in Figure 18.15 to determine the values of the following quantities: a. Amplitude b. Time period c. Maximum velocity d. Maximum acceleration. 0.02 - period, T 0.01 - t/s -0.01 -...
-
The first three energy levels \(E(\mathrm{eV})\) of the fictitious element \(\mathrm{X}\) are \(\ldots . . \ldots\) shown in Figure P29.56. a. What wavelengths are observed in the absorption spectrum...
-
A sodium atom in the \(3 p\) state emits a photon, decreasing the energy of the atom by \(2.10 \mathrm{eV}\). By how much does the orbital angular momentum of the atom change? Does it increase or...
-
A 1.5 V D-cell battery is rated at \(15,000 \mathrm{~mA} \cdot \mathrm{h}\). For how long could such a battery power a flashlight bulb rated at \(1.5 \mathrm{~W}\) ?

Study smarter with the SolutionInn App


