Questions for Figure 13 What important conclusions can your make? List 2 important conclusions 1) A...
Fantastic news! We've Found the answer you've been seeking!
Question:

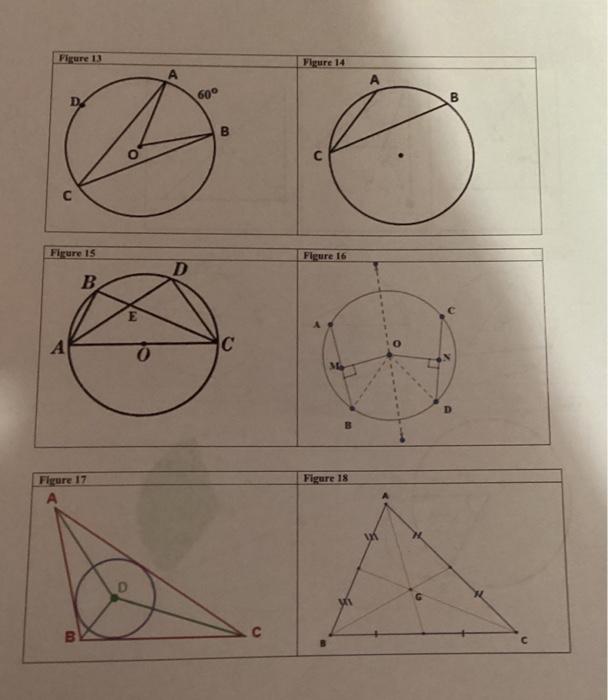
Transcribed Image Text:
Questions for Figure 13 What important conclusions can your make? List 2 important conclusions 1) A Questions for Figure 15 What are the measures of the angles below? Angle ABE Angle ADC What conclusion can you make about Triangle ABE and Triangle CDE? Questions for Figure 17 Point D is called the Triangle ABC of Point D was determined by obtaining the three Questions for Figure 14 What important conclusions can your make? List 2 important conclusions 1) A Questions for Figure 16 What important conclusions can your make? List 2 important conclusions 1) Questions for Figure 18 Point G is called the of Triangle ABC Point G was determined by obtaining the three Figure 13 D Figure 15 B A Figure 17 B D D 60° B C Figure 14 Figure 16 Figure 18 B Questions for Figure 13 What important conclusions can your make? List 2 important conclusions 1) A Questions for Figure 15 What are the measures of the angles below? Angle ABE Angle ADC What conclusion can you make about Triangle ABE and Triangle CDE? Questions for Figure 17 Point D is called the Triangle ABC of Point D was determined by obtaining the three Questions for Figure 14 What important conclusions can your make? List 2 important conclusions 1) A Questions for Figure 16 What important conclusions can your make? List 2 important conclusions 1) Questions for Figure 18 Point G is called the of Triangle ABC Point G was determined by obtaining the three Figure 13 D Figure 15 B A Figure 17 B D D 60° B C Figure 14 Figure 16 Figure 18 B
Expert Answer:

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these mathematics questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Carol Harris, Ph.D, CPA, is a single taxpayer and she lives at 674 Yankee Street, Durham, NC 27409. Her Social Security number is 793-52-4335. Carol is an Associate Professor of Accounting at a local...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
Discuss the methodology of logical positivism and falsification in questions of a pre history of science and technology studies
-
Determine the vertical force P which must be applied at G so that the linkage is in equilibrium for the position shown. 1.2 in. 3.6 in.- 20 lb 1,6 in. 15 lb-ft 1.6 in.
-
Washers used in a certain application are supposed to have a thickness of 2 millimeters. A quality control engineer measures the thicknesses for a sample of washers and tests H0: = 2 versus H1: 2....
-
How can a judgment creditor obtain funds in a judgment debtors bank account?
-
Marriott International, Inc., and Wyndham Worldwide Corporation are two major owners and managers of lodging and resort properties in the United States. Abstracted income statement information for...
-
A firm has 100 bonds outstanding with 10 years until maturity. The bonds pay an annual coupon, a coupon rate of 3%, a face value of $1,000, and a yield to maturity of 2.75%. Also, the firm has 50,000...
-
The Genola Company manufactures insulated windows. Costs for March were as follows. Direct labor $53,000 Indirect labor 18,000 Salary of corporate vice president for advertising Direct materials...
-
A lightly doped Si sample (ND = 1014 cm-3) is heated from 300 K to 400 K. Is its resistivity expected to increase or decrease? Explain your answer including calculation. Repeat for Si with ND 1018...
-
Three identical units of Item K113 are purchased during July, as shown below. Item K113 Units Cost July 9 Purchase 1 166 17 Purchase 1 168 26 Purchase 1 198 Assume that one unit is sold on July 31...
-
Why is the French legal system highly protective of employees' rights?
-
The units of an item available for sale during the year were as follows: Jan 1 Inventory 15 units at 121 April 15 Purchase 139 units at 114 September 9 Purchase 23 units at 121 There are 40 units of...
-
You are unexpectedly telephoned on December 28 with a request to perform your services/sell some of your products on the last day of December. In early January, your company will send an invoice for...
-
In 2022, Desi's Desserts had four employees. Two of the employees worked full time and earned salaries of $35,000 each. Two employees worked only part time, and each earned $5,000. The employer...
-
Answer the following questions: Why business writing and communication is critical to your success: 2. What writing skills are most important; 3. How non-verbal communication are demonstrated; and...
-
One study found that the elderly who do not have children dissave at about the same rate as the elderly who do have children. What might this finding imply about the reason the elderly do not dissave...
-
Olive Corporation was formed and began operations on January 1, 2012. The corporation's income statement for the year and the balance sheet at year-end are presented below. The corporation made...
-
Ken paid the following amounts for interest during 2012: Qualified interest on home mortgage...........................................$4,700 Auto loan...
-
Russell (age 50) and Linda (age 45) Long have brought you the following information regarding their income and expenses for the current year. Russell owns and operates a landscaping business called...
-
In the ammonia synthesis reaction \(\mathrm{N}_{2}+3 \mathrm{H}_{2}=2 \mathrm{NH}_{3}+22.4 \mathrm{kcal}\), the formation of \(\mathrm{NH}_{3}\) will be favoured by (a) High temperature (b) Low...
-
The internal energy of an ideal gas depends on (a) Temperature, specific heat and volume (b) Temperature and specific heat (c) Temperature, specific heat and pressure (d) Pressure, volume and...
-
For steady flow, the first law of thermodynamics (a) Is concerned with heat interaction (b) Is an energy balance for a specified mass of fluid (c) Accounts for all energy entering and leaving a...

Study smarter with the SolutionInn App


