Reaction Product Reactant requirements (kg) per kg product 0.6 A; 0.4 B 0.4 A; 0.5 B;...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
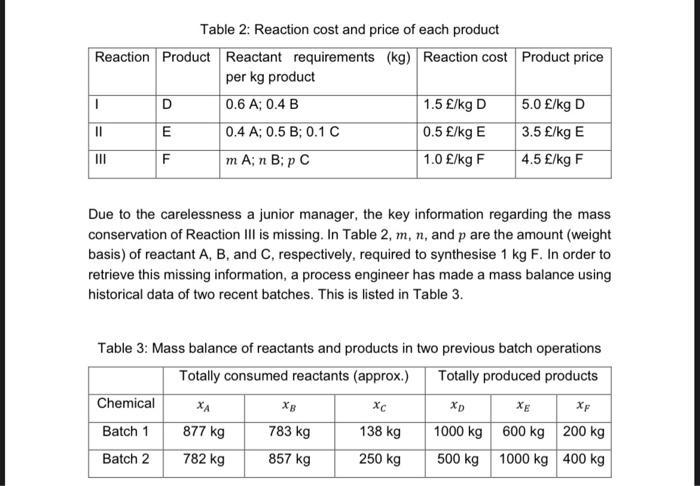
Reaction Product Reactant requirements (kg) per kg product 0.6 A; 0.4 B 0.4 A; 0.5 B; 0.1 C m A; n B; p C I || ||| Table 2: Reaction cost and price of each product Reaction cost Product price D E F Chemical Batch 1 Batch 2 Due to the carelessness a junior manager, the key information regarding the mass conservation of Reaction III is missing. In Table 2, m, n, and p are the amount (weight basis) of reactant A, B, and C, respectively, required to synthesise 1 kg F. In order to retrieve this missing information, a process engineer has made a mass balance using historical data of two recent batches. This is listed in Table 3. 1.5 £/kg D 0.5 £/kg E 1.0 £/kg F Table 3: Mass balance of reactants and products in two previous batch operations Totally consumed reactants (approx.) Totally produced products xc 138 kg 250 kg XA 877 kg 782 kg 783 kg 857 kg 5.0 £/kg D 3.5 £/kg E 4.5 £/kg F XD 1000 kg 500 kg 600 kg 1000 kg XF 200 kg 400 kg 2. Least-squares model construction (5 marks): 2.1. Use only Table 2 and Table 3 to construct a full least-squares model with an objective to calculate m, n, and p. You can only build the model using: x₁,1 (kg day-1 of A consumed in batch 1), XB,1 (kg day¹ of B consumed in batch 1), xc.1 (kg day-¹ of C consumed in batch 1), XA2 (kg day¹ of A consumed in batch 2), x8,2 (kg day-¹ of B consumed in batch 2), xcz (kg day¹ of C consumed in batch 2), m, n, and p. No other symbols are allowed to use. The full model should consist of both an objective function and all possible constraints - remember that all constraints, as well as the objective function, should be independent from each other. Note: Do not use the 'normalised least-squares objective function. 2.2. Restructure the full optimisation model in 2.1 into a reduced optimisation model by substituting the equality constraints where appropriate. Make sure the reduced model only consists of inequality constraints. The reduced model should consist of both an objective function and all possible inequality constraints. Note: Do not use the 'normalised' least-squares objective function. Reaction Product Reactant requirements (kg) per kg product 0.6 A; 0.4 B 0.4 A; 0.5 B; 0.1 C m A; n B; p C I || ||| Table 2: Reaction cost and price of each product Reaction cost Product price D E F Chemical Batch 1 Batch 2 Due to the carelessness a junior manager, the key information regarding the mass conservation of Reaction III is missing. In Table 2, m, n, and p are the amount (weight basis) of reactant A, B, and C, respectively, required to synthesise 1 kg F. In order to retrieve this missing information, a process engineer has made a mass balance using historical data of two recent batches. This is listed in Table 3. 1.5 £/kg D 0.5 £/kg E 1.0 £/kg F Table 3: Mass balance of reactants and products in two previous batch operations Totally consumed reactants (approx.) Totally produced products xc 138 kg 250 kg XA 877 kg 782 kg 783 kg 857 kg 5.0 £/kg D 3.5 £/kg E 4.5 £/kg F XD 1000 kg 500 kg 600 kg 1000 kg XF 200 kg 400 kg 2. Least-squares model construction (5 marks): 2.1. Use only Table 2 and Table 3 to construct a full least-squares model with an objective to calculate m, n, and p. You can only build the model using: x₁,1 (kg day-1 of A consumed in batch 1), XB,1 (kg day¹ of B consumed in batch 1), xc.1 (kg day-¹ of C consumed in batch 1), XA2 (kg day¹ of A consumed in batch 2), x8,2 (kg day-¹ of B consumed in batch 2), xcz (kg day¹ of C consumed in batch 2), m, n, and p. No other symbols are allowed to use. The full model should consist of both an objective function and all possible constraints - remember that all constraints, as well as the objective function, should be independent from each other. Note: Do not use the 'normalised least-squares objective function. 2.2. Restructure the full optimisation model in 2.1 into a reduced optimisation model by substituting the equality constraints where appropriate. Make sure the reduced model only consists of inequality constraints. The reduced model should consist of both an objective function and all possible inequality constraints. Note: Do not use the 'normalised' least-squares objective function.
Expert Answer:
Answer rating: 100% (QA)
Anwer ex Given that E col 200 lit bioreactor alm is measured Jas ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these accounting questions
-
A bar of mass m = 5 kg is held as shown between four disks each of mass m?? = 2 kg and radius r = 75 mm. Knowing that the forces exerted on the disks are sufficient to prevent slipping and that the...
-
A 4 m x 5 m x 6 m room contains 120 kg of air. Determine (a) Density. (b) Specific volume. (c) Mole (d) Specific molar volume of air. Assume molar mass of air to be 29 kg/kmol.
-
The reaction between reactant A (blue spheres) and reactant B (red spheres) is shown in the following diagram: Based on this diagram, which equation best describes the reaction? (a) A2 + B A2B (b) A2...
-
Here are the comparative income statements of Eudaley Corporation. Instructions (a) Prepare a horizontal analysis of the income statement data for Eudaley Corporation, using 2013 as a base. (Show the...
-
For the demand data in Problem 11, develop a seasonally adjusted forecast for year 4. (Use a linear trend line model to develop a forecast estimate for year 4.) Which forecast model do you perceive...
-
On January 1, 2014, Nami Inc.s ledger shows its capital asset of $79,000 (at cost) and accumulated depreciation of $28,125. When the capital asset was purchased, Nami estimated that its useful life...
-
After factory overhead has been applied to work in process, what does a credit balance in Factory Overhead indicate?
-
Premium Garden Products (PGP) supplies gardening enthusiasts and commercial garden centers throughout the northeast with a wide range of gardening equipment and products. Their wide range of...
-
In a(n) ___________ employees have a hpothetical account into which is deposited what is typically a percentage of annual compenstion. 1) employee stock ownership plan 2) cash balance plan 3) profit...
-
An exercise advocate wants to determine the effect that walking rigorously has on weight loss. The researcher recruits participants to engage in a weeklong study. The researcher instructs...
-
Total cost per unit 138 The desired ROl per 41.4 Target selling price is 179.4 Assume the new product requires an investment of $20,000,000 to be manufactured, and the company wants to achieve ROl of...
-
Determine the symmetrical components of the following line currents: (a) \(I_{a}=5 / 90^{\circ}\), \(I_{b}=5 / 320^{\circ}, I_{c}=5 / 220^{\circ} \mathrm{A}\); (b) \(I_{a}=j 50, I_{b}=50, I_{c}=0...
-
Make a list of professionals, professors, alumni, family members, friends and others you know who might become part of your network.
-
What is third-party credibility? Why is it important to CEs? How is it achieved?
-
Write a short essay on what you would like your personal brand to be.
-
Revisit the definition of strategic communication and then review the vignette at the beginning of the chapter. Match the different terms in the definition to the different entities in the vignette.
-
Recursive Member Test Write a recursive boolean function named is Member. The function should accept three parameters: an array of integers, an integer indicating the number of elements in the array,...
-
A woman at a point A on the shore of a circular lake with radius 2 mi wants to arrive at the point C diametrically opposite on the other side of the lake in the shortest possible A time. She can walk...
-
A voltaic cell is constructed that uses the following half-cell reactions: The cell is operated at 298 K with [Cu+] = 0.20 M and [I-] = 3.5 M. (a) Determine E for the cell at these concentrations....
-
(a) What is meant by the term molecularity? (b) Why are termolecular elementary reactions so rare? (c) What is an intermediate in a mechanism?
-
(a) A serving of a particular ready-to-serve chicken noodle soup contains 2.5 g fat, 14 g carbohydrate, and 7 g protein. Estimate the number of Calories in a serving. (b) According to its nutrition...
-
The supply of a good _________ if the price of one of its substitutes in production rises.
-
The supply of a good _________ if the price of one of its substitutes in production falls.
-
A situation where quantity demanded is greater than quantity supplied is called a(n) _________.

Study smarter with the SolutionInn App


