(a) Explain why the reaction of SF4 with BF3 yields [SF3], whereas the reaction with CsF...
Fantastic news! We've Found the answer you've been seeking!
Question:
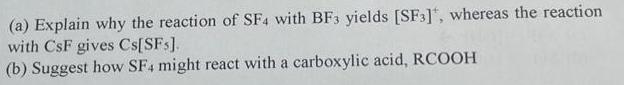
Transcribed Image Text:
(a) Explain why the reaction of SF4 with BF3 yields [SF3], whereas the reaction with CsF gives Cs[SFs]. (b) Suggest how SF4 might react with a carboxylic acid, RCOOH (a) Explain why the reaction of SF4 with BF3 yields [SF3], whereas the reaction with CsF gives Cs[SFs]. (b) Suggest how SF4 might react with a carboxylic acid, RCOOH
Expert Answer:
Answer rating: 100% (QA)
1 SFs Bf3 SFY CSF to its lay This is because Brs is dedtron deficient in nature act as Lewis Ac... View the full answer

Related Book For 

Managerial Economics and Strategy
ISBN: 978-0321566447
1st edition
Authors: Jeffrey M. Perloff, James A. Brander
Posted Date:
Students also viewed these chemistry questions
-
The North American Free Trade Agreement provides for two- way, long- haul trucking across the U. S.-Mexican border. U. S. truckers have objected, arguing that the Mexican trucks dont have to meet the...
-
Write a report describing the North American Free Trade Agreement (NAFTA), including its history, what it is, what it does, and its effectiveness in promoting trade between its member companies.
-
After Mexico signed the North American Free Trade Agreement (NAFTA) in 1994, corn imports from the United States doubled within a year, and today U.S. imports make up nearly one-third of the corn...
-
Simplify each expression in Problems 722. Classify each answer by number of terms and degree. (x + 2y3z) - (x - 5y + 4z)
-
Consultex, Inc., was founded in 2012 as a small financial consulting business. The company had done reasonably well in 2012 2014, but started noticing its cash dwindle early in 2015. In January 2015,...
-
The jet engine shown scoops in air at A at a rate of 90 kg/s and discharges it at B with a velocity of 600 m/s relative to the airplane. Determine the magnitude and line of action of the propulsive...
-
How are the mean value and variance of a random variable defined?
-
Wendells Donut Shoppe is investigating the purchase of a new $18,600 donut-making machine. The new machine would permit the company to reduce the amount of part-time help needed, at a cost savings of...
-
Write a program that will do the following: Consolidate all the information from the csv files they have provided into a single csv file called "survey_database.csv" write a summary of the collected...
-
The 2013 financial statements for Royale and Cavalier companies are summarized here: These two companies are in the same business and state but different cities. One-half of Royales sales and...
-
The accounting equation for a special revenue fund is : Assets Liabilities = Net Position. Financial Assets + Deferred Outflows Related Liabilities Deferred Inflows = Fund Balance. General Capital...
-
A (simplistic) model of the hydrogen atom treats the electron as a particle carrying a charge \(-e\) orbiting a proton (a particle carrying a charge \(+e\) ) in a circle of radius...
-
A parallel-plate capacitor consists of two conducting plates with a surface area of \(1.0 \mathrm{~m}^{2}\) and a plate separation distance of \(50 \mu \mathrm{m}\). (a) Determine the capacitance and...
-
Amherst Metal Works produces two types of metal lamps. Amherst manufactures 20,000 basic lamps and 5,000 designer lamps. Its simple costing system uses a single indirect-cost pool and allocates costs...
-
A motor of \(5 \mathrm{~kW}\) running at \(950 \mathrm{rpm}\) is used in a riveting machine. A flywheel is attached to the machine has a mass of \(100 \mathrm{~kg}\) and radius of gyration of \(0.4...
-
A porter governor has an equal arm's length of \(220 \mathrm{~mm}\) long and pivoted at the axis of rotation. Each ball has a mass of \(5 \mathrm{~kg}\) and the mass of central load on the sleeve is...
-
M&Ms and Skittles both have green-colored candy. You performed Paper chromatography to confirm the dye(s) in these green candy samples. You chose Fast green and Club House Green Dye as standard dyes....
-
The following table shows the rates of total return in successive years from 2004 to 2008 for the Sprott Canadian Equity Fund and for the benchmark Toronto Stock Exchange S&P/TSX Composite Index. By...
-
Under monopolistic competition with identical firms, is it possible for a firm to produce at the minimum of its average cost curve?
-
A refiner produces heating fuel and gasoline from crude oil in virtually fixed proportions. What can you say about economies of scope for such a firm? What is the sign of its measure of economies of...
-
Firm 1 and Firm 2 use the same type of production function, but Firm 1 is only 90% as productive as Firm 2. That is, the production function of Firm 2 is q2 = f(L, K), and the production function of...
-
The spring attachment point \(B\) is given a horizontal motion \(x_{B}=\) \(b \cos \omega t\). Determine the critical driving frequency \(\omega_{c}\) for which the oscillations of the mass \(m\)...
-
A simplified version of a pendulum used in impact tests is shown in the figure. Derive the equation of motion and determine the period for small oscillations about the pivot. The mass center \(G\) is...
-
The 100-lb piston is supported by a spring of modulus \(k=200 \mathrm{lb} / \mathrm{in}\). A dashpot of damping coefficient \(c=85 \mathrm{lb}\)-sec/ft acts in parallel with the spring. A fluctuating...

Study smarter with the SolutionInn App


