Reagent: A 1) Na 2) Met 3) 9-BBN 4) HO, NaOH 1) 9-6BN 2) HO, NaOH...
Fantastic news! We've Found the answer you've been seeking!
Question:
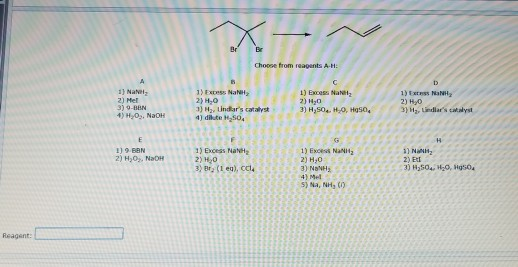
Transcribed Image Text:
Reagent: A 1) Na 2) Met 3) 9-BBN 4) H₂O₂, NaOH 1) 9-6BN 2) H₂O₂, NaOH X Br Br Choose from reagents A-H: B 1) Excess Na 2) H₂0 3) H₂, Lindlar's catalyst 4) dikute H₂50, 1) Exors NaN H 2) H₂0 3) Br, (1 eq), CCl4 C 1) Excess Na 2) H₂0 3) H₂50 H₂O, Hg50, 1) Exors NaN 2) H₂0 3) MẠNH, 4) Mel 5) Na, NH₂ (1) D 1) Extrus NINH 2) H₂0 3) H₂, Lindlar's catalyst H 1) Na 2) Et 3) H₂50₁, H₂O, Hg50₂ Reagent: A 1) Na 2) Met 3) 9-BBN 4) H₂O₂, NaOH 1) 9-6BN 2) H₂O₂, NaOH X Br Br Choose from reagents A-H: B 1) Excess Na 2) H₂0 3) H₂, Lindlar's catalyst 4) dikute H₂50, 1) Exors NaN H 2) H₂0 3) Br, (1 eq), CCl4 C 1) Excess Na 2) H₂0 3) H₂50 H₂O, Hg50, 1) Exors NaN 2) H₂0 3) MẠNH, 4) Mel 5) Na, NH₂ (1) D 1) Extrus NINH 2) H₂0 3) H₂, Lindlar's catalyst H 1) Na 2) Et 3) H₂50₁, H₂O, Hg50₂
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
x / 9 1 / 3 = 2 / 9
-
A steel ball of mass m = 50 g falls from the height h = 1.0 m on the horizontal surface of a massive slab. Find the cumulative momentum that the ball imparts to the slab after numerous bounces, if...
-
5 / 4 + 9 / 2x = 3 / 8 x 1 / 4
-
In the context of channel-of-distribution structures, discuss the distinguishing features of the Japanese distribution structure. Now, explain why the Japanese have not embraced the U.S distribution...
-
Ron Seiser, president of Seiser Corporation, is considering the issuance of bonds to finance an expansion of his business. He has asked you to (1) Discuss the advantages of bonds over common stock...
-
Starting from rest, a 12-cm-diameter compact disk takes 3.0 s to reach its operating angular velocity of 2000 rpm. Assume that the angular acceleration is constant. The disks moment of inertia is 2.5...
-
Locate an article that discusses an injunction that has been obtained by a party in California. What was the defendant enjoined from doing? Was this a temporary, preliminary, or permanent injunction?...
-
In the 1990s, Pfizer, Inc., developed a new antibiotic called Trovan (trovafloxacin mesylate). Tests showed that in animals Trovan had life-threatening side effects, including joint disease, abnormal...
-
Sanchez Corporation runs two convenience stores, one in Connecticut and one in Rhode Island. Operating income for each store in 2020 is as follows: (Click to view the operating income for the...
-
In this mini-case, you will complete the test of details on accounts receivable for the 2019 audit of EarthWear Clothiers, Inc. The principal test of detail involves sending "confirmations" or...
-
Complete the square to explain why a quadratic polynomial has either a global maxi- mum or a global minimum but not both.
-
The following was submitted under the original assignment of "write small program that uses dynamic binding".. now what is requested is to suggest another way to take advantage of the dynamic biding...
-
Productivity can also be described as the effectiveness with which the resources input in a plant are translated to customer satisfaction-oriented products. Explain
-
Alex worked for years for MegaCorp. During his employment with MegaCorp, he learned a great deal of confidential information and knew that if this information got into the hands of competitors or the...
-
Maxey & Sons manufactures two types of storage cabinets-Type A and Type Band applies manufacturing overhead to all units at the rate of $152 per machine hour. Production information follows....
-
Suppose S 0 SF / $ = SF 0 . 9 4 9 4 / $ and F 6 - monthSF / $ = SF 0 . 9 4 7 8 / $ . What is the percentage forward premium / discount on the U . S . dollar?
-
Sheffield Company showed the following balances at the end of its first year: Cash Prepaid insurance Accounts receivable Accounts payable Notes payable Owner's Capital Owner's Dividends Revenues...
-
One hundred pounds of water at atmospheric pressure are heated from 60F to 200F. What is the enthalpy change? The internal energy change? Why is the difference between the internal energy change and...
-
Propose a mechanism for the following reaction. Show the structure of the compound that results from hydrolysis and decarboxylation of the product. CHO + CH2(COOCH2CH3)2 hydrolysis, decarboxylation...
-
An unknown reducing disaccharide is found to be unaffected by invertase enzymes. Treatment with an -galactosidase cleaves the disaccharide to give one molecule of D-fructose and one molecule of...
-
Without looking back, propose a mechanism for the hydrolysis of this iminium salt to the alkylated ketone. The first step is attack by water, followed by loss of a proton to give a carbinolamine....
-
Applying the criterion for equilibrium, derive the Clausius-Clapeyron equation.
-
A binary liquid mixture consists of \(60 \mathrm{~mol}\) per cent ethylene and \(40 \mathrm{~mol}\) per cent propylene. At \(423 \mathrm{~K}\), the vapour pressure of ethylene and propylene are...
-
The pure component vapour pressure of two organic liquids \(\mathrm{X}\) and \(\mathrm{Y}\) by Antoine equations are given by \[ \ln P_{1}^{\text {Sat }}=14.35-\frac{2942}{T+220} \] and \[...

Study smarter with the SolutionInn App



