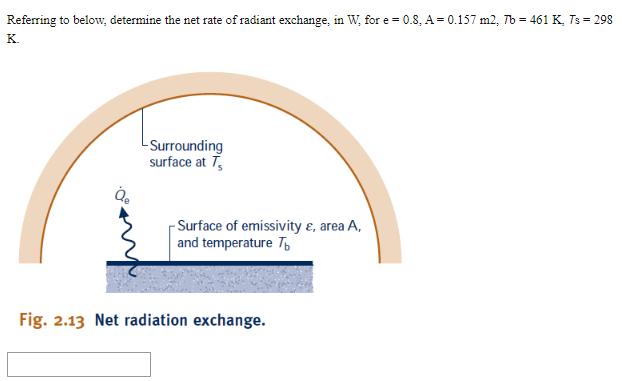
Referring to below, determine the net rate of radiant exchange, in W, for e=0.8, A =...
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
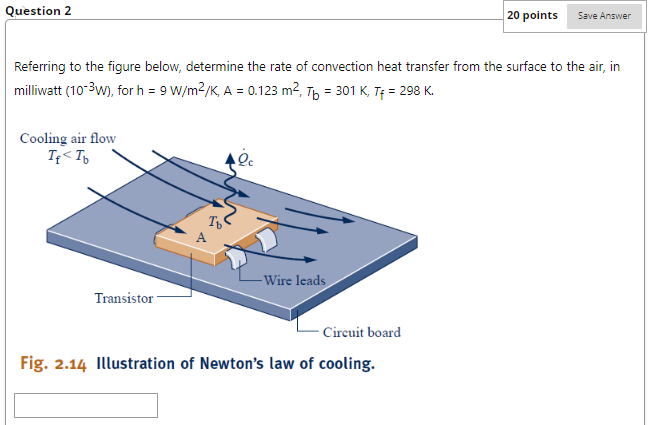
Referring to below, determine the net rate of radiant exchange, in W, for e=0.8, A = 0.157 m2, Tb = 461 K, Ts = 298 K. -Surrounding surface at T -Surface of emissivity &, area A, and temperature T Fig. 2.13 Net radiation exchange. Question 2 Cooling air flow If Question 3 20 points Save Answer A gas is compressed in a piston-cylinder assembly from p = 2.2 bar to p2 = 7.0 bar, V = 0.02 m in a process during which the relation between pressure and volume is pv1.4 = constant. The mass of the gas is 0.4 kg. If the specific internal energy of the gas increases by 40 kJ/kg during the process, determine the heat transfer, in Joules. Kinetic and potential energy changes are negligible. Question 4 20 points Save Answer A mass of 10 kg undergoes a process during which there is heat transfer from the mass at a rate of 5 kJ per kg, an elevation decrease of 47 m, and an increase in velocity from 15 m/s to 27 m/s. The specific internal energy decreases by 5 kJ/kg and the acceleration of gravity is constant at 9.7 m/s2. Determine the work for the process, in Joules. Question 5 20 points Evaluate the overall work, in kJ. Save Answer Carbon monoxide gas (CO) contained within a piston- cylinder assembly undergoes three processes in series: Process 1-2: Expansion from p = 3.7 bar, V = 0.9 m to V = 1.2 m, during which the pressure-volume relationship is pV = constant. Process 2-3: Constant-volume heating from state 2 to state 3, where p3 = 3.7 bar. Process 3-1: Constant-pressure compression to the initial state. Referring to below, determine the net rate of radiant exchange, in W, for e=0.8, A = 0.157 m2, Tb = 461 K, Ts = 298 K. -Surrounding surface at T -Surface of emissivity &, area A, and temperature T Fig. 2.13 Net radiation exchange. Question 2 Cooling air flow If Question 3 20 points Save Answer A gas is compressed in a piston-cylinder assembly from p = 2.2 bar to p2 = 7.0 bar, V = 0.02 m in a process during which the relation between pressure and volume is pv1.4 = constant. The mass of the gas is 0.4 kg. If the specific internal energy of the gas increases by 40 kJ/kg during the process, determine the heat transfer, in Joules. Kinetic and potential energy changes are negligible. Question 4 20 points Save Answer A mass of 10 kg undergoes a process during which there is heat transfer from the mass at a rate of 5 kJ per kg, an elevation decrease of 47 m, and an increase in velocity from 15 m/s to 27 m/s. The specific internal energy decreases by 5 kJ/kg and the acceleration of gravity is constant at 9.7 m/s2. Determine the work for the process, in Joules. Question 5 20 points Evaluate the overall work, in kJ. Save Answer Carbon monoxide gas (CO) contained within a piston- cylinder assembly undergoes three processes in series: Process 1-2: Expansion from p = 3.7 bar, V = 0.9 m to V = 1.2 m, during which the pressure-volume relationship is pV = constant. Process 2-3: Constant-volume heating from state 2 to state 3, where p3 = 3.7 bar. Process 3-1: Constant-pressure compression to the initial state.
Expert Answer:
Answer rating: 100% (QA)
a Critically damped system The steadystate error for a critically damped system can be calculated us... View the full answer

Related Book For 

Principles of heat transfer
ISBN: 978-0495667704
7th Edition
Authors: Frank Kreith, Raj M. Manglik, Mark S. Bohn
Posted Date:
Students also viewed these mechanical engineering questions
-
You have an expected activity duration of 26 days, optimistic value of 18 days, and pessimistic value of 67 days. Calculate the PERT.This type of question contains radio buttons and checkboxes for...
-
Find the error in the derivative of f(x)=cos(x) by Computing directly and using the central divided difference x=0.8 (rad) x=0.6 (rad) ? Determine h so that absolute error is a) less than 10?3 b)...
-
A 0.61 m radius hemisphere (811 K surface temperature) is filled with a gas mixture at 533 K and 2-atm pressure containing 6.67 percent CO2 and water vapor at 0.5 percent relative humidity. Determine...
-
During 2018, Susan incurred and paid the following expenses for Beth (her daughter), Ed (her father), and herself: Surgery for...
-
Paulsen Corporation makes two products, W and P, in a joint process. At the split-off point, 50,000 units of W and 60,000 units of P are available each month. Monthly joint production costs are...
-
Propane gas at 1 bar and 35oC is compressed to a final state of 135 bar and 195oC. Estimate the molar volume of the propane in the final state and the enthalpy and entropy changes for the process. In...
-
Advertisers use several types of publicity mechanisms. Look through some recent newspapers and magazines or use an Internet search engine and identify a news release, a feature article, or a...
-
Go to Extended Case 29.1, Citizens National Bank of Jessamine County v. Washington Mutual Bank, 309 S.W.3d 792 (2010), on pages 567568. Read the excerpt and answer the following questions. (a) Issue:...
-
2. Personal Ethics: Discuss and explain three values that you believe Tony Hsieh has. Next, with consideration to the various ethical theories and lives of ethical philosophers, identify and explain...
-
Mrs. Sam Taffer was a brilliant engineer at IBM and made a lot of money from stock options. Since then, she has worked as a musician and raised her two sons. She wishes to have most of her property...
-
In its income statement for the year ended December 31, 2025, Sandhill Company reported the following condensed data. Salaries and wages expenses $455,700 Loss on disposal of plant assets $63,700...
-
Here is a FDT for a small data set: data freq 22 23 23 2 3 24 3 25 4 8 26 Find the following measures of central tendency. mean median = mode = Enter an integer or decimal number [more..]
-
What is the present value of $1 received eight years from now if the discount rate is 12%?
-
The following information is available for Keller Co.'s new product line: Selling price per unit P15 Variable manufacturing cost per unit of production 8 Total annual fixed manufacturing costs 25,000...
-
1(a). Two colleagues, Leila and Junshang, share an office which needs some cleaning. Both would like a clean office. However, each of them prefers not to be the one that does the cleaning. Here's the...
-
Draw 2 simple graphs of t on the x axis . Car A is moving at a constant 20 m/s Car B is moving at a constant 60 m/s a. The first graph puts velocity on the y axis. Sketch the 2 velocities over time...
-
Determine bad debt expense, and prepare the adjusting entry E8.8 (LO 2), AP Godfreid Company has accounts receivable of $95,400 at March 31, 2025. Credit terms are 2/10, n/30. At March 31, 2025,...
-
By referring to Figure 13.18, determine the mass of each of the following salts required to form a saturated solution in 250 g of water at 30 oC: (a) KClO3, (b) Pb(NO3)2, (c) Ce2(SO4)3.
-
A Trombe wall is a masonry wall often used in passive solar homes to store solar energy. Suppose such a wall, fabricated from 20 cm thick solid concrete blocks (k = 0.13 W/(mK), = 0.05 \ 10 5 m 2 /s...
-
Wearing layers of clothing in cold weather is often recommended because dead-air spaces between the layers keep the body warm. The explanation for this is that the heat loss from the body is less....
-
By substituting Equation 9.1 for Eb (T) in Equation 9.4 and performing the integration over the entire spectrum, derive a relationship between s and the constants C1 and C2 in Equation...
-
Can any financial product normally make it possible to obtain resources at below market cost?
-
Redo the exercise above, assuming in the first case that the Faurecia share rises to 40 or falls to 25. What is the impact on the value of the option? What basic feature of the option have you...
-
You wish to value a call option on the Faurecia share (which does not pay dividends) after 6 months with a strike price of 35 and a 6-month duration. You do not know what volatility to factor in....

Study smarter with the SolutionInn App


