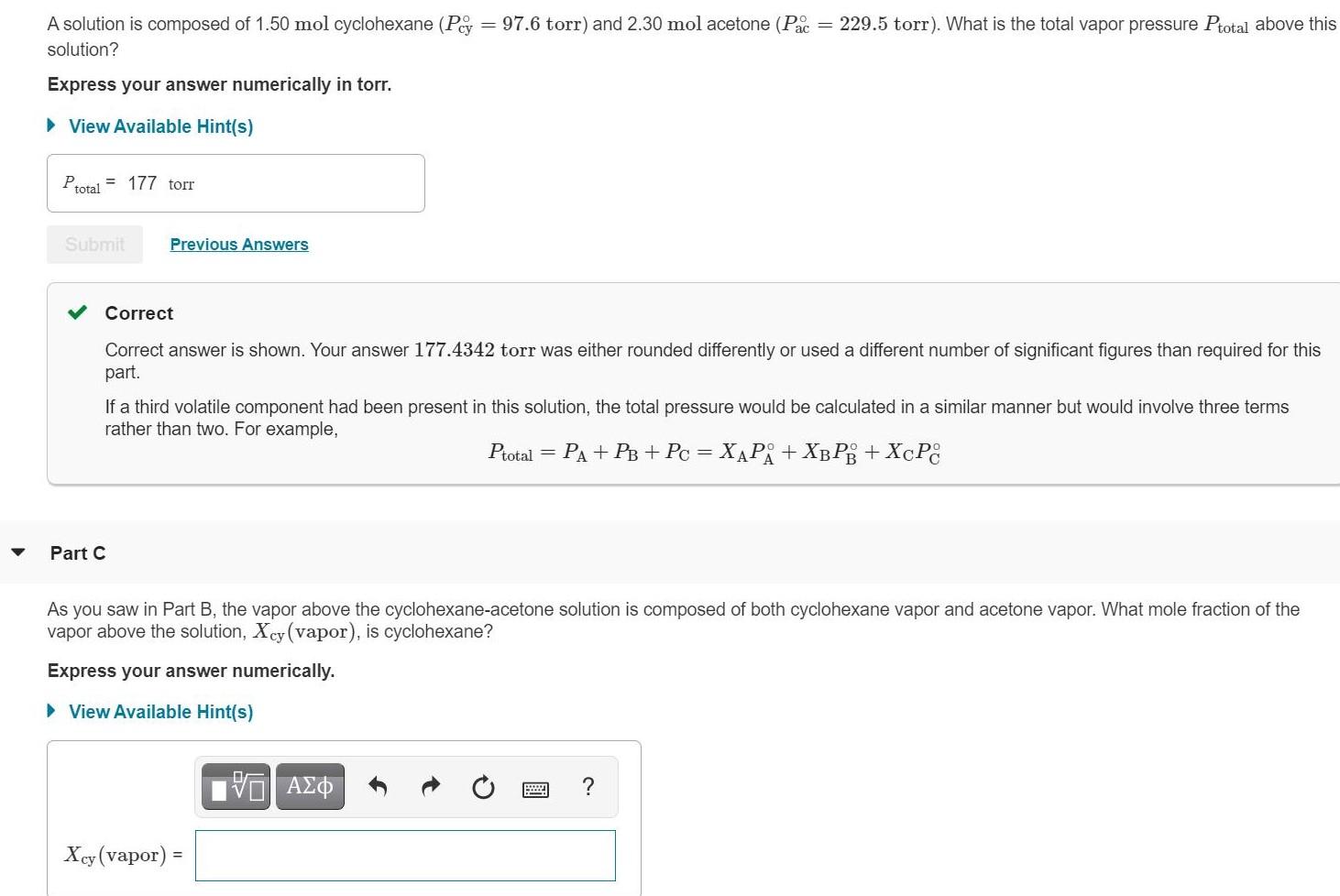
A solution is composed of 1.50 mol cyclohexane (Pey = 97.6 torr) and 2.30 mol acetone...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A solution is composed of 1.50 mol cyclohexane (Pey = 97.6 torr) and 2.30 mol acetone (Pac = 229.5 torr). What is the total vapor pressure Ptotal above this solution? Express your answer numerically in torr. • View Available Hint(s) Ptotal = 177 torr Submit Previous Answers Correct Correct answer is shown. Your answer 177.4342 torr was either rounded differently or used a different number of significant figures than required for this part. If a third volatile component had been present in this solution, the total pressure would be calculated in a similar manner but would involve three terms rather than two. For example, Ptotal = Ра + Рв + Рс 3D ХАРА + ХВРВ + ХcРC Part C As you saw in Part B, the vapor above the cyclohexane-acetone solution is composed of both cyclohexane vapor and acetone vapor. What mole fraction of the vapor above the solution, Xcy (vapor), is cyclohexane? Express your answer numerically. > View Available Hint(s) ? Xey (vapor): !! A solution is composed of 1.50 mol cyclohexane (Pey = 97.6 torr) and 2.30 mol acetone (Pac = 229.5 torr). What is the total vapor pressure Ptotal above this solution? Express your answer numerically in torr. • View Available Hint(s) Ptotal = 177 torr Submit Previous Answers Correct Correct answer is shown. Your answer 177.4342 torr was either rounded differently or used a different number of significant figures than required for this part. If a third volatile component had been present in this solution, the total pressure would be calculated in a similar manner but would involve three terms rather than two. For example, Ptotal = Ра + Рв + Рс 3D ХАРА + ХВРВ + ХcРC Part C As you saw in Part B, the vapor above the cyclohexane-acetone solution is composed of both cyclohexane vapor and acetone vapor. What mole fraction of the vapor above the solution, Xcy (vapor), is cyclohexane? Express your answer numerically. > View Available Hint(s) ? Xey (vapor): !!
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the total vapor pressure at 20C of a liquid solution containing 0.30 mole fraction benzene, C6H6, and 0.70 mole fraction toluene, C6H5CH3? Assume that Raoult's law holds for each component of...
-
A solution is composed of 1.10 mol cyclohexane (Pe = 97.6 torr) and 2.70 mol acetone (Pa = 229.5 torr). What is the total vapor pressure Ptotal above this solution? Express your answer with the...
-
A 1200 kg car is accelerated from 30 to 50 km/h in 5 s. How much work is that? If you continue from 50 to 70 km/h in 5 s is that the same?
-
Assume that on January 1, 2016 an investor company paid $11,600 to an investee company in exchange for the following assets and liabilities transferred from the investee company: Investee's Estimated...
-
Find the points on the lemniscates in Exercise 31 where the tangent is horizontal.
-
Figure P20.55 shows four charges at \(-10 \mathrm{nC}\) the corners of a square of side \(L\). What magnitude and sign of charge \(Q\) will make the force on charge \(q\) zero? 10 nC L 2 -10 nC...
-
The salaries (in thousand dollars) of a sample of 10 employees Organize the data using the indicated type of graph. Describe any patterns. 225 410 368 310 228 298 361 159 486 296
-
Baldwin Company produces treadmills. One of its plants produces two versions: a standard model and a deluxe model. The deluxe model has a wider and sturdier base and a variety of electronic gadgets...
-
A rigid container contains an ideal gas 40C which is stirred by a blade. The blade produces 200kJ of work. The temperature remains constant thanks to a heat exchange with the surroundings which are...
-
Pam Corporation purchased 75 percent of the outstanding voting stock of Sun Corporation for $4,800,000 on January 1, 2016. Sun's stockholders' equity on this date consisted of the following (in...
-
Use a subcontractor who is a carpenter that is registered with the Victorian Building Association (VBA) , responsible for framing 8 apartment building. 1. Inform the successful and unsuccessful...
-
On June 10, 20X6, Dar Al Fatina purchased material supplies on account for $35,000. Which of the journal entries for this transaction in the table below is correct. A Date June 10, 20X6 Cash Accounts...
-
__________theory assumes that language acquisition is based on principles of conditioning and shaping.
-
Developmental psychologists are interested in the effects of both __________and __________on development.
-
Winning the lottery is likely to a. Produce an immediate and long-term increase in the level of well-being. b. Produce an immediate but not lingering increase in the level of well-being. c. Produce a...
-
Which of the following statements is true? A. An object storage bucket can only exist in one compartment. B. An object storage bucket can exist in multiple compartments. C. An object storage bucket...
-
The teacher-made test is better than the standardized tests for evaluating students performances. Discuss the merits of this statement
-
A business had revenues of $280,000 and operating expenses of $315,000. Did the business (a) Incur a net loss (b) Realize net income?
-
Acetic acid, CH3COOH, is contained in vinegar. Suppose acetic acid was formed from its elements, according to the following equation: 2C(graphite) + 2H2(g) + O2(g) CH3COOH(l) Find the enthalpy...
-
The following is a possible mechanism for a reaction involving hydrogen peroxide in aqueous solution; only a small amount of sodium bromide was added to the reaction mixture. H2O2 + Br BrO + H2O...
-
Phosphorus pentachloride is normally a white solid. It exists in this state as the ionic compound [PCl4+][PCl6]. Describe the electron structure of the PCl6 ion in terms of valence bond theory.
-
In testing the claim that the mean IQ score of statistics students is greater than 100, the alternative hypothesis is expressed as > 100. Decide whether the statement makes sense (or is clearly...
-
In testing a claim about a population mean, a larger z test statistic always results in a larger P-value. Decide whether the statement makes sense (or is clearly true) or does not make sense (or is...
-
A handy mnemonic for interpreting the P-value in a hypothesis test is this: If the P (value) is low, then the null must go.

Study smarter with the SolutionInn App