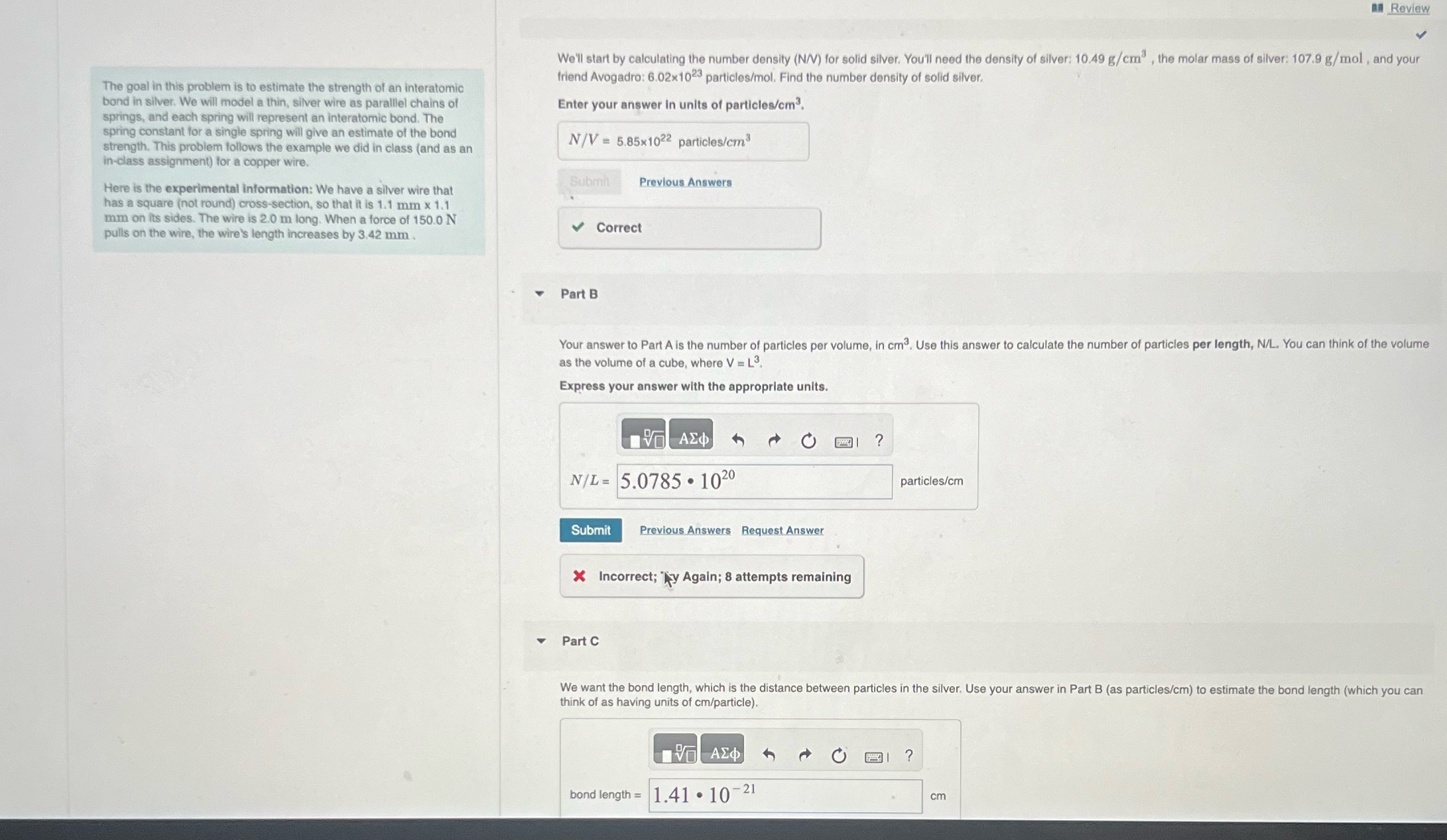
Review The goal in this problem is to estimate the strength of an interatomic bond in...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Review The goal in this problem is to estimate the strength of an interatomic bond in silver. We will model a thin, silver wire as parallel chains of springs, and each spring will represent an interatomic bond. The spring constant for a single spring will give an estimate of the bond strength. This problem follows the example we did in class (and as an in-class assignment) for a copper wire. Here is the experimental information: We have a silver wire that has a square (not round) cross-section, so that it is 1.1 mm x 1.1 mm on its sides. The wire is 2.0 m long. When a force of 150.0 N pulls on the wire, the wire's length increases by 3.42 mm. We'll start by calculating the number density (N/V) for solid silver. You'll need the density of silver: 10.49 g/cm, the molar mass of silver: 107.9 g/mol, and your friend Avogadro: 6.02x1023 particles/mol. Find the number density of solid silver. Enter your answer in units of particles/cm. N/V=5.85x1022 particles/cm Submit Previous Answers Correct 4 Part B Your answer to Part A is the number of particles per volume, in cm. Use this answer to calculate the number of particles per length, N/L. You can think of the volume as the volume of a cube, where V = L. Express your answer with the appropriate units. N/L=5.0785 1020 Submit Previous Answers Request Answer Incorrect; y Again; 8 attempts remaining ? particles/cm Part C We want the bond length, which is the distance between particles in the silver. Use your answer in Part B (as particles/cm) to estimate the bond length (which you can think of as having units of cm/particle). bond length = 1.41 107 -21 ? cm Review The goal in this problem is to estimate the strength of an interatomic bond in silver. We will model a thin, silver wire as parallel chains of springs, and each spring will represent an interatomic bond. The spring constant for a single spring will give an estimate of the bond strength. This problem follows the example we did in class (and as an in-class assignment) for a copper wire. Here is the experimental information: We have a silver wire that has a square (not round) cross-section, so that it is 1.1 mm x 1.1 mm on its sides. The wire is 2.0 m long. When a force of 150.0 N pulls on the wire, the wire's length increases by 3.42 mm. We'll start by calculating the number density (N/V) for solid silver. You'll need the density of silver: 10.49 g/cm, the molar mass of silver: 107.9 g/mol, and your friend Avogadro: 6.02x1023 particles/mol. Find the number density of solid silver. Enter your answer in units of particles/cm. N/V=5.85x1022 particles/cm Submit Previous Answers Correct 4 Part B Your answer to Part A is the number of particles per volume, in cm. Use this answer to calculate the number of particles per length, N/L. You can think of the volume as the volume of a cube, where V = L. Express your answer with the appropriate units. N/L=5.0785 1020 Submit Previous Answers Request Answer Incorrect; y Again; 8 attempts remaining ? particles/cm Part C We want the bond length, which is the distance between particles in the silver. Use your answer in Part B (as particles/cm) to estimate the bond length (which you can think of as having units of cm/particle). bond length = 1.41 107 -21 ? cm
Expert Answer:

Related Book For 

Shigleys Mechanical Engineering Design
ISBN: 978-1121345317
9th edition
Authors: Richard G. Budynas, J. Keith Nisbett
Posted Date:
Students also viewed these physics questions
-
The Vietnamese government wants to improve the country's environment. It is offering $200,000 for the next five years to any manufacturing company that reduces its pollution output by 20% over the...
-
CANMNMM January of this year. (a) Each item will be held in a record. Describe all the data structures that must refer to these records to implement the required functionality. Describe all the...
-
Voguish Couches is a small company which makes sofas. The company is owned by the couple Lennox and Stewart. You are an intern working at Voguish Couches. For the sake of simplicity, this assignment...
-
One kg of liquid water at 25oC: (a) Experiences a temperature increase of 1 K. What is U'. in kJ? (b) Experience a change in elevation Z. The change in potential energy EP is the same as Ut for part...
-
What is the mass in milligrams of>? (a) 0.367 mol of HNO3? (b) 245 mmol of MgO? (c) 12.5 mol of NH4NO3? (d) 4.95 mol of (NH4)2Ce (NO3)6 (548.23 g/mol)
-
When an ion channel opens in a cell wall, monovalent (charge \(e\) ) ions flow through the channel at a rate of \(1.0 \times 10^{7} \mathrm{ions} / \mathrm{s}\). a. What is the current through the...
-
1. What benefits of entrepreneurship does Sara Blakely appear to have attained? 2. Is the desire to earn an income a key motivator for Blakely? Explain your answer. 3. What was Blakelys opportunity...
-
If the same data were reported for car insurance, instead of health insurance, would the graph look similar? Why or why not?
-
what contributed to the development of hip hop and how it affected society and/or other popular culture?
-
Two all around enhanced portfolios An and B (all quirky danger has been differentiated away) have 1) expeccted returns of 12% and 9% separately. The Beta for Portfolio An is 1.2 while the Beta for...
-
A sample of gas occupies a volume of 260 ml when the temperature is 85C and the pressure is 750mm Hg. Both the temperature and pressure were changed causing the volume occupied by the gas to become...
-
3. (20 points) Large-scale pathloss a. (12 points) Given are the following system design characteristics. Transmit power = 2W Frequency = 2.4 GHz Pathloss exponent = 3.5 Isotropic receiver antenna...
-
Before and after-tax cost of debt For the following $1,000-par-value bond paying semi-annual interest payments, calculate the before- and after-tax cost of debt. Use the 21% corporate tax rate....
-
Give an exar example to show that the result of the preceding problem need not hold when k is even.
-
Read the article The Relationship Between Cell Phone Use and Academic Performance in a Sample of U.S. College Students Andrew Lepp1, Jacob E. Barkley1, and Aryn C. Karpinski1 ) Based on the assigned...
-
Write each fraction as a percent. 7 50
-
Using the economic balance sheet approach, the Laws economic net worth is closest to: A. $925,000. B. $1,425,000. C. $1,675,000. Raye uses a costbenefit approach to rebalancing and recommends that...
-
Using an economic balance sheet, which of the Laws current financial assets is most concerning from an asset allocation perspective? A. Equities B. Real estate C. Fixed income Raye uses a costbenefit...
-
To address his concern regarding the previous advisers asset allocation approach, Raye should assess the Laws portfolio using: A. a homogeneous and mutually exclusive asset classbased risk analysis....

Study smarter with the SolutionInn App


