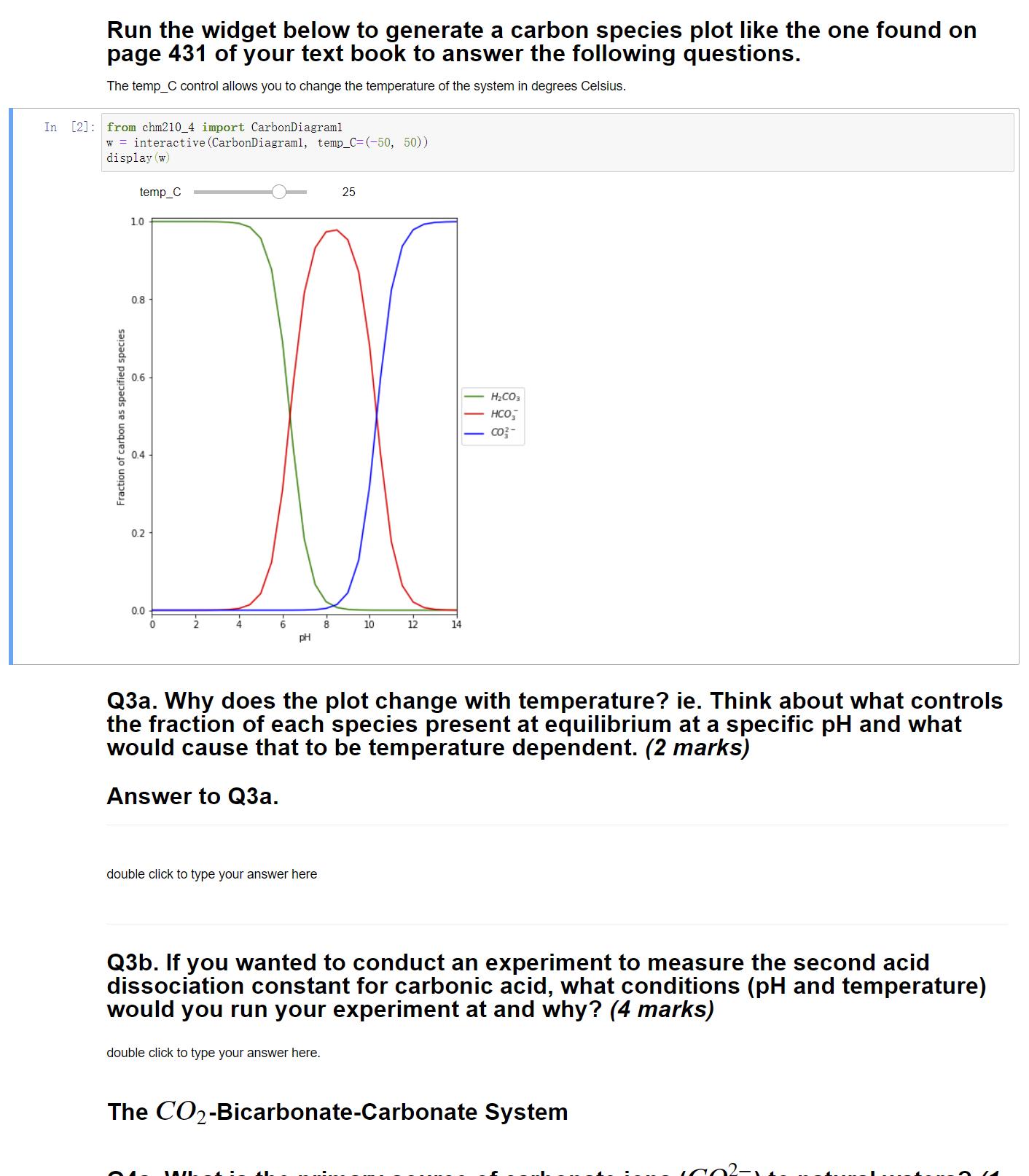
Run the widget below to generate a carbon species plot like the one found on page...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Run the widget below to generate a carbon species plot like the one found on page 431 of your text book to answer the following questions. The temp_C control allows you to change the temperature of the system in degrees Celsius. In [2]: from chm210 4 import CarbonDiagraml w = interactive (CarbonDiagraml, temp_C= (-50, 50)) display (w) temp_C 25 1.0 0.8 0.6 H2CO, HCO, - co3- 04 0.2 0.0 8 10 12 14 pH Q3a. Why does the plot change with temperature? ie. Think about what controls the fraction of each species present at equilibrium at a specific pH and what would cause that to be temperature dependent. (2 marks) Answer to Q3a. double click to type your answer here Q3b. If you wanted to conduct an experiment to measure the second acid dissociation constant for carbonic acid, what conditions (pH and temperature) would you run your experiment at and why? (4 marks) double click to type your answer here. The CO2-Bicarbonate-Carbonate System Fraction of carbon as specified species ||| Run the widget below to generate a carbon species plot like the one found on page 431 of your text book to answer the following questions. The temp_C control allows you to change the temperature of the system in degrees Celsius. In [2]: from chm210 4 import CarbonDiagraml w = interactive (CarbonDiagraml, temp_C= (-50, 50)) display (w) temp_C 25 1.0 0.8 0.6 H2CO, HCO, - co3- 04 0.2 0.0 8 10 12 14 pH Q3a. Why does the plot change with temperature? ie. Think about what controls the fraction of each species present at equilibrium at a specific pH and what would cause that to be temperature dependent. (2 marks) Answer to Q3a. double click to type your answer here Q3b. If you wanted to conduct an experiment to measure the second acid dissociation constant for carbonic acid, what conditions (pH and temperature) would you run your experiment at and why? (4 marks) double click to type your answer here. The CO2-Bicarbonate-Carbonate System Fraction of carbon as specified species |||
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
On page 431 of Physics: Calculus, 2d ed., by Eugene Hecht (Pacific Grove, CA: Brooks/Cole, 2000), in the course of deriving the formula T = 2L/g for the period of a pendulum of length L, the author...
-
Competitive information is one of the most common things that businesses research. It is important to note that the internet and easy access to information as made research accessible for even the...
-
Your Way is a fast-food restaurant company with thousands of franchise organizations all around the country. They desperately need you and Just Right Company to develop their latest software...
-
Write a well-developed paragraph that compares/contrasts a character from Good Country People with a character from Seventeen Syllables.
-
What is probability? What do we mean by relative frequency as a probability estimate for events? What is the law of large numbers? How would the law of large numbers apply in this context?
-
For the circuit shown in Figure \(32.4 a\), let the period \(T\) of the oscillation be \(1.0 \mathrm{~s}\) and the emf be zero at \(t=0\). Identify the instant(s) at which the energy stored in the...
-
A surface condenser deals with \(13000 \mathrm{~kg} / \mathrm{h}\) of steam. The leakage air in the system amounts to \(1 \mathrm{~kg}\) per \(2700 \mathrm{~kg}\) of steam. The vacuum in the air pump...
-
Pension plan information for Naperville Window Company is as follows: Assuming no change in actuarial assumptions, what is the pension service cost for2011? January I, 2011 PBO ABO Pension benefits...
-
If an investor invests in a bond that has $1,000 par and pays 7% coupons semi-annually, which matures in 3 years. Assume yield stays at 3% compound semiannually and all received coupon payments will...
-
The trial balance of Norr Ltd at 31 December 20X2 appeared as follows: Additional information 1. During the year a motor vehicle purchased on 31 March 20W9 for 8,000 was sold for 3,000. The sale...
-
1. For the following causal system y(n) = x(n)-x(n-1)-1.2y(n-1)-0.36y(n-2) Determine the transient and steady-state response for the input x(n)=u(n) using the inverse z-transform and the residue...
-
At December 31, 2015, Ashley Co. has outstanding purchase commitments for purchase of 150,000 gallons, at 6.20 per gallon, of a raw material to be used in its manufacturing process. The company...
-
Under what circumstances is it appropriate to record goodwill in the accounts? How should goodwill, properly recorded on the books, be written off in order to conform with IFRS?
-
Manchester United, Inc. sold 10,000 season tickets at 2,000 each. By December 31, 2015, 8 of the 20 home games had been played. What amount should be reported as a current liability at December 31,...
-
Define a restructuring. What costs should not be accrued in a restructuring?
-
The book basis of depreciable assets for Erwin Co. is 900,000 and the tax basis is 700,000 at the end of 2015. The enacted tax rate is 34% for all periods. Determine the amount of deferred taxes to...
-
1. A flask has a mass of 78.23 g when empty and 593.63 g when filled with water. When the same flask is filled with concentrated sulfuric acid, H2SO4, its mass is 1026.57 g. What is the density of...
-
Identify the tax issues or problems suggested by the following situations. State each issue as a question. Jennifer did not file a tax return for 2007 because she honestly believed that no tax was...
-
The random-number generator on calculators randomly generates a number between 0 and 1. The random variable X, the number generated, follows a uniform probability distribution. (a) Draw the graph of...
-
The highest batting average ever recorded in major league baseball was by Ted Williams in 1941 when he hit 0.406. That year, the mean and standard deviation for batting average were 0.2806 and...
-
A certain vehicle emission inspection station advertises that the wait time for customers is less than 8 minutes. A local resident is skeptical and collects a random sample of 49 wait times for...
-
An annealing furnace has a belt that moves material in at a specific velocity, \(U\) (Figure P10.34). Only the region inside the oven is heated and the material sees a radiant heat flux of...
-
As a biochemical engineer you are evaluating a drug delivery system for an artificial protein to combat Alzheimer's. The protein is very large (200,000 molecular weight) and bulky and is sensitive to...
-
Consider the mass transfer example concerning diffusion into a falling film shown in Figure P10.38. A new inventor claims that he can rig the device to operate such that the flux of \(a\) from the...

Study smarter with the SolutionInn App


