s1-2x , O 2x-1 , 2 dynamie system and tent mop T: [0,1]0] T(x)= 2-2x,...
Fantastic news! We've Found the answer you've been seeking!
Question:
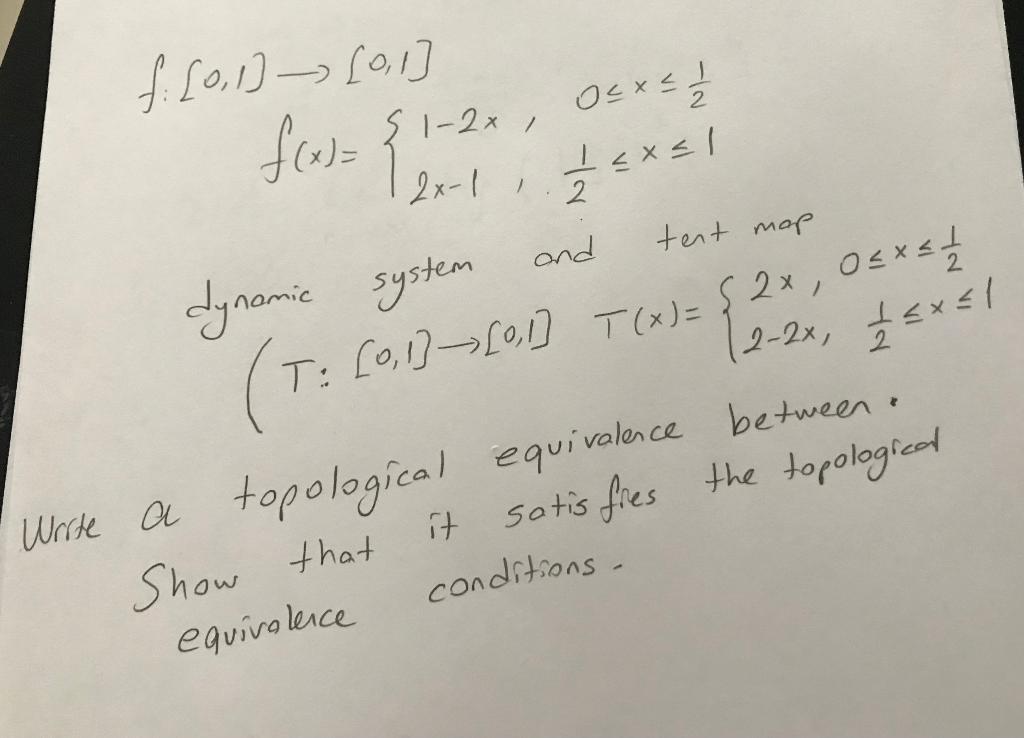
Transcribed Image Text:
s1-2x , Oと×」 2x-1 , 2 dynamie system and tent mop T: [0,1]¬0] T(x)= 2-2x, <x<l Write a topological n. equivalence between it sotis fies the topologicad Show that conditions - equivolence s1-2x , Oと×」 2x-1 , 2 dynamie system and tent mop T: [0,1]¬0] T(x)= 2-2x, <x<l Write a topological n. equivalence between it sotis fies the topologicad Show that conditions - equivolence
Expert Answer:
Answer rating: 100% (QA)
Consider the dynamical system defined by the map f 0 1 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
Q3. (a) (b) For the following boundary value problem, use CTCS scheme with Ax = 0.1 to approximate u (5.0.2) u (1.0.2) and u (2,0.2). 5 2u Ju for d1 dx u(0, t) = u(x,t) = 0 u(x,0) = sin (2x), 0
-
At a particular instant an electron is traveling in the +y direction, with speed 8 x 105 m/s. At the location of the electron there is a magnetic field of magnitude 0.39 T in the -y direction, due to...
-
x / 9 1 / 3 = 2 / 9
-
On January 2, 2013, Parker Corporation invests in the stock of Quarry Corporation. Quarry's book value is $4 million and its assets and liabilities are fairly reported. Quarry reports income of $3...
-
Consider the following hypotheses: H 0 : p < 0.15 H 1 : p > 0.15 Given that n = 150 and = 0.10, calculate b for each of the following conditions. a. p = 0.16 b. p = 0.22
-
Click fraud has become a major concern as more and more companies advertise on the Internet. When Google places an ad for a company with its search results, the company pays a fee to Google each time...
-
As can easily be shown, the probabilities of getting 0 , 1 , or 2 heads with a pair of balanced coins are \(\frac{1}{4}, \frac{1}{2}\), and \(\frac{1}{4}\). What is the probability of getting 2 tails...
-
Fifty-thousand pounds per hour of a 20 wt% aqueous solution of NaOH at 120F is to be fed to an evaporator operating at 3.7 psia, where the solution is concentrated to 40 wt% NaOH. The heating medium...
-
Gold Inc invests $10,000 today in a mutual fund. Gold anticipates leaving this fund alone for 12 years. $ The fund is increased each year-end by specified compound interest rates as follows: years 1...
-
The Ploughman family owns and operates a 640-acre farm that has been in the family for several generations. The Ploughmans always have had to work hard to make a decent living from the farm and have...
-
Golden Wolves Statue Company * Corey had always dreamed of starting his own manufacturing company. Now that he was in an Entrepreneurship class and in his senior year as a Business Major at Alvernia...
-
Imagine that you and several of your classmates have bought the patents and production facilities of a US motor scooter company that has gone bankrupt. Given the growing market for home deliveries,...
-
June and John decide to form a business. They each plan to contribute $20,000 in exchange for a 50 percent interest in the business. They will then take out a bank loan for $30,000 to cover the...
-
Several years ago, Catherines Cattery Supplies signed a contract to deliver 5000 units of a special product each year to one of its customers at a price of $15 per unit. This contract, which runs for...
-
You have recently been appointed as a senior advisor in the domestic and overseas trade division in your countrys ministry of trade and development. Despite a high birth rate and a good education...
-
In the 52 years between 1956 and 2008, Warren Buffetts net worth grew from $100,000 to $62 billion. What was his annual rate of return?
-
Mr. Ansah is a business man he recently moved from the United States of America to Ghana and he was introduced toyou through a mutual friend. Mr. Ansah has made a lot of money from working in the...
-
Using the information in P11-2B, compute the overhead controllable variance and the overhead volume variance. Data From Problem 11-2B: Huang Company uses a standard cost accounting system to account...
-
The following compounds have the same molecular formulas (C4H10). Which one would you expect to have a higher boiling point?
-
Starting with rutile (TiO2), explain how you would obtain pure titanium metal.
-
Classify each of the following species as a weak or strong acid: (a) HNO3, (b) HF, (c) H2SO4, (d) HSO4-, (e) H2CO3, (f) HCO3-, (g) HCl, (h) HCN, (i) HNO2.
-
What is the change in value for this security over the four quarters recorded in the highlowclose graph?
-
Which of the 10 years had the most and least variability between the maximum sales of womens clothing and the minimum for mens, and what were those ranges?
-
What happened to the average sales figures for jewelry over the 10-year period?

Study smarter with the SolutionInn App


