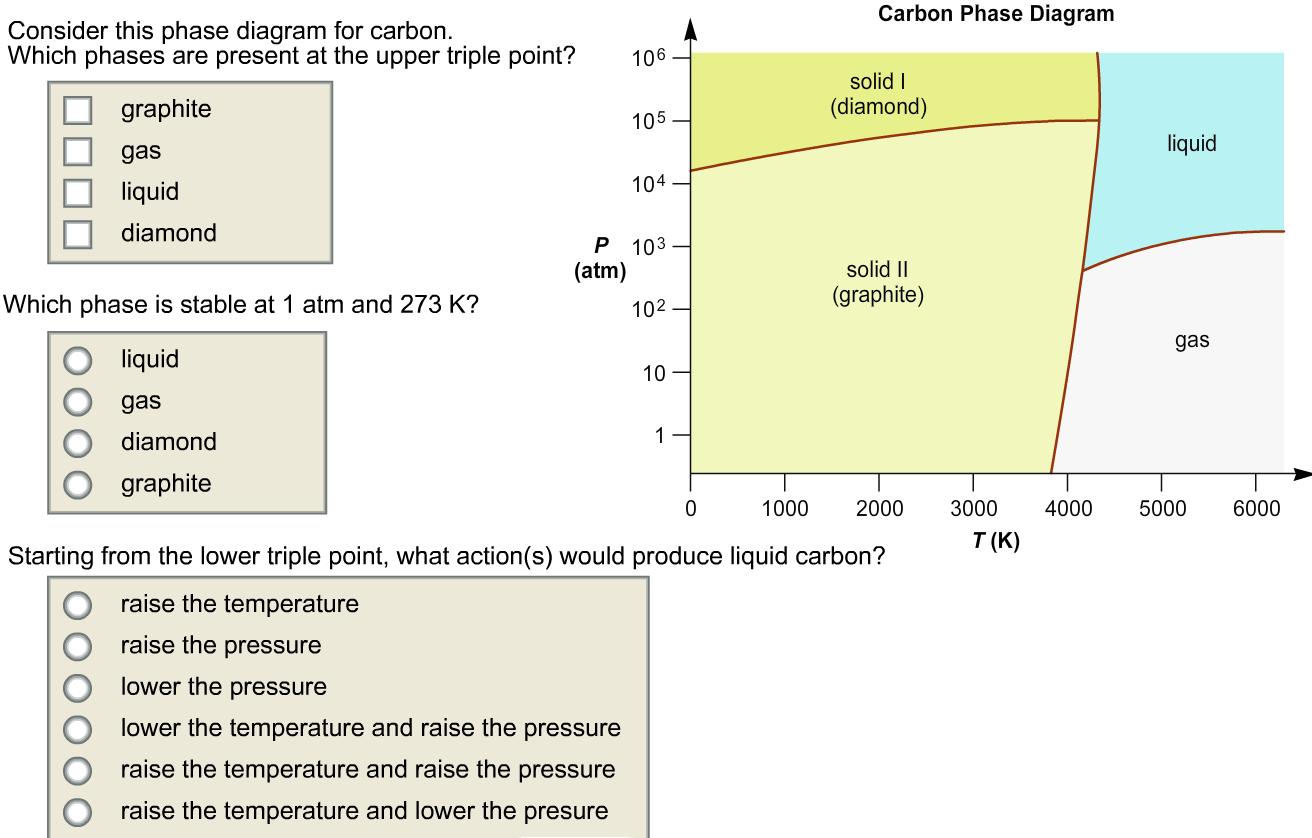
Consider this phase diagram for carbon. Which phases are present at the upper triple point? graphite...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider this phase diagram for carbon. Which phases are present at the upper triple point? graphite gas liquid diamond Which phase is stable at 1 atm and 273 K? liquid gas diamond graphite (atm) 106 105. P 103 lower the pressure lower the temperature and raise the pressure raise the temperature and raise the pressure raise the temperature and lower the presure 104 10² 10 1 Carbon Phase Diagram solid I (diamond) solid II (graphite) 0 Starting from the lower triple point, what action(s) would produce liquid carbon? raise the temperature raise the pressure 1000 2000 liquid gas 3000 4000 5000 T(K) 6000 Consider this phase diagram for carbon. Which phases are present at the upper triple point? graphite gas liquid diamond Which phase is stable at 1 atm and 273 K? liquid gas diamond graphite (atm) 106 105. P 103 lower the pressure lower the temperature and raise the pressure raise the temperature and raise the pressure raise the temperature and lower the presure 104 10² 10 1 Carbon Phase Diagram solid I (diamond) solid II (graphite) 0 Starting from the lower triple point, what action(s) would produce liquid carbon? raise the temperature raise the pressure 1000 2000 liquid gas 3000 4000 5000 T(K) 6000
Expert Answer:
Answer rating: 100% (QA)
TRIPLE POINT THREE 1 2 Ans 3 PHASES PHASES OF UPPER AT AT TRIPLE POINT ANS PRESENT UPPER ... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these chemistry questions
-
Saturated water vapor at 400F is heated at constant pressure until its volume has doubled. Determine the final temperature using the ideal gas equation of state, the compressibility charts, and the...
-
Ethylene is heated at constant pressure from 5 MPa and 20oC to 200oC. Using the compressibility chart, determine the change in the ethylene's specific volume as a result of this heating.
-
Suppose 1.00 mol of oxygen is heated at constant pressure of 1.00 atm from 10.0C to 25.0C. (a) How much heat is absorbed by the gas? (b) Using the ideal gas law, calculate the change of volume of the...
-
At Blossom Company, events and transactions during 2020 included the following. The tax rate for all items is 20%. (1) Depreciation for 2018 was found to be understated by $148000. (2) A strike by...
-
On April 1, 1996, Taco Bell, the fast-food chain, ran a full-page ad in the New York Times with this news: In an effort to help the national debt, Taco Bell is pleased to announce that we have agreed...
-
Imagine a cylindrical tree of radius a. A wedge is cut from the tree by making two cuts: one in a horizontal plane P perpendicular to the axis of the cylinder and one that makes an angle u with P,...
-
Briefly describe situations in which it would be advantageous to use Form 1040A instead of Form 1040EZ. When would you use Form 1040EZ?
-
The Shannon Community Kitchen provides hot meals to homeless and low-income individuals and families; it is the organizations only program. The Kitchen had the following revenue and expense...
-
May 13 - Received payment from the customer on account due from May 4, less the return and discount General Journal Account Name Debit Credit Cash 4.559 5. 141 Accounts Receivable - Loop 410 Cafe...
-
Peyton Smith enjoys listening to all types of music and owns countless CDs. Over the years, Peyton has gained a local reputation for knowledge of music from classical to rap and the ability to put...
-
Given a normal distribution with mu equals 100 and sigma equals 10 comma probability that Upper X less than 90 or Upper X greater than 115 is
-
An auditor selects a preliminary sample of 100 items out of a population of 1,000 items. The sample statistics generate an arithmetic mean of \($60\), a standard deviation of \($6\), and a standard...
-
An important statistic to consider when using a classical statistical sampling audit plan is the population variability. The population variability is measured by the a. Sample mean. b. Standard...
-
The following list of tests is typical of those usually performed by auditors in gathering evidence to ascertain the fairness of financial statement balances. For each listed test, indicate whether...
-
What is the purpose of obtaining authenticated deposit slips from the bank? Explain the limitations on the effectiveness of this procedure.
-
List two requests an auditor will make of a bank in connection with his or her audit of cash.
-
Sunlight Ltd called for subscriptions for 3 million shares. Theissue price per share is $5.00 to be paid in three parts: the firstpayment of $2.00 is to be made on application, $2.00 is to be paidw 2...
-
DC has unused FTC carryover from 2017 in the separate category for GC income as the result of income generated by a foreign branch. The income was foreign source general category income. In 2018 the...
-
The pressure gage on a 2.5-m3 oxygen tank reads 500 kPa. Determine the amount of oxygen in the tank if the temperature is 28oC and the atmospheric pressure is 97 kPa. 2 500 kPa 02 V 25 m T 28C FIGURE...
-
A gas mixture consists of 6 kmol of H2 and 2 kmol of N2. Determine the mass of each gas and the apparent gas constant of the mixture.
-
Methane (CH4) is burned with stoichiometric amount of air during a combustion process. Assuming complete combustion, determine the air-fuel and fuel-air ratios.
-
Using divisibility rules, determine if 342,887 is divisible by 9 .
-
Using divisibility rules, determine if 87,762 is divisible by 10 .
-
Can 43,568 pieces of mail be separated into 6 bins with the same number of pieces of mail per bin?

Study smarter with the SolutionInn App


