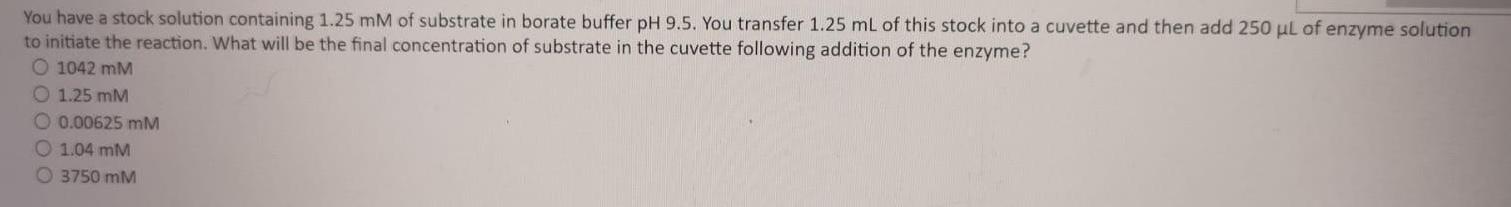
You have a stock solution containing 1.25 mM of substrate in borate buffer pH 9.5. You...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
You have a stock solution containing 1.25 mM of substrate in borate buffer pH 9.5. You transfer 1.25 mL of this stock into a cuvette and then add 250 ul of enzyme solution to initiate the reaction. What will be the final concentration of substrate in the cuvette following addition of the enzyme? O 1042 mM O 1.25 mM O 0.00625 mM O 1.04 mM O 3750 mM You have a stock solution containing 1.25 mM of substrate in borate buffer pH 9.5. You transfer 1.25 mL of this stock into a cuvette and then add 250 ul of enzyme solution to initiate the reaction. What will be the final concentration of substrate in the cuvette following addition of the enzyme? O 1042 mM O 1.25 mM O 0.00625 mM O 1.04 mM O 3750 mM
Expert Answer:
Answer rating: 100% (QA)
Ans 104 mM Explanation Taking125 ml of the stock so... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
You have a stock solution certified by a manufacturer to contain 150.0 z 0.3 mg SO42- /mL. You would like to dilute it by a factor of 100 to obtain 1.500 mg SO42- /mL. Two possible dilution methods...
-
(a) You have a stock solution of 14.8 MNH3.How many milliliters of this solution should you dilute to make 1000.0 mL of 0.250 M NH3? (b) If you take a 10.0-mL portion of the stock solution and dilute...
-
You have a stock portfolio consisting of approximately 100 stockswhere each stock constitutes roughly 1% of the portfolio's total value. The firms constitute a variety of industries and economic...
-
Suppose that inventory at the beginning of the year consists of 5 units of WIDGET01, costing $33. 00 each. During the year, you received three different shipments for WIDGET01 as follows: on January...
-
Refer to the Practice Set data provided in Chapters 2 and 3. Consider the following adjustment data: a. Cleaning supplies on hand at the end of November were $ 50. b. One months combined depreciation...
-
The 2-kg ball is thrown at the suspended 20-kg block with a velocity of 4 m/s. If the coefficient of restitution between the ball and the block is e = 0.8, determine the maximum height h to which the...
-
What is the probability that a player is dealt an initial hand with an 8 and a 3 ?
-
The Beef-up ranch feeds cattle for midwestern farmers and delivers them to processing plants in Topeka, Kansas and Tulsa, Oklahoma. The ranch must determine the amounts of cattle feed to buy so that...
-
3. Use the graph of y = p(x) below to find the value to the right. 8 7 6. 10 5 4 3 2 1 -6 -5 -4 -3 -2 -1 0 1 2 3 4 -1- 2.
-
The director of RCM inc. plans to launch a new product. The initial investment in equipment and other fittings is $800,000. It's been a while since management thinking of launching this new product....
-
5) We want to explore the effect of parameters on a binomial distribution (this is a bit similar to last week's assignment). Start up R/RStudio a) First we'll do our coin example from lecture but use...
-
Ayayai Warehouse distributes suitcases to retail stores and extends credit terms of n/30 to all of its customers. Ayayai Warehouse uses a perpetual inventory system and the earnings approach. At the...
-
a) What amount should Frank report as taxable income? Show all workings. (10 marks) b) Calculate the tax payable by Frank, make note of eligible credits or use the taxtips website mentioned above....
-
and h. Obtain the open-circuit voltage ratio, voltage source Vin. Vin Vout Vin The two two-ports networks shown below are characterized by the hybrid-parameters h (s), and the input impedance Zin,...
-
Study carefully the following links to line graphs here below that shows mortality rates data for neonatal, infant, children under five each per 1,000 live births for United States, Japan, Denmark,...
-
Three +4.0-C point charges are at the three corners of a square of sides 1.00 m. The fourth corner (the lower right hand position) is occupied by a -4.0-C charge. Begin with the sketch; label the...
-
Question 7 (5 points) Redner, Inc. produces two products. Data concerning the two products appear below: Selling price Variable costs Fixed costs Grinding machine time Products A B $70 $95 45 65 15 5...
-
6. (Potential Energy and Conservation of Energy) What should be the spring constant k of a spring designed to bring a 1200-kg car to rest from a speed of 95 km/h so that the occupants undergo a...
-
Write complete balanced half-reactions for (a) oxidation of nitrous acid to nitrate ion in acidic solution, (b) oxidation of N 2 to N 2 O in acidic solution.
-
The precipitation reaction between AgNO3(aq) and NaCl(aq) proceeds as follows: (a) By using Appendix C, calculate ÎH° for the net ionic equation of this reaction. (b) What would you expect...
-
Gold is alloyed (mixed) with other metals to increase its hardness in making jewelry. (a) Consider a piece of gold jewelry that weighs 9.85 g and has a volume of 0.675 cm3. The jewelry contains only...
-
Simple numbers are used in this problem to highlight the concepts covered in the chapter. Assume that the Canberra Company produces one producta bath mat that sells for $10. Canberra uses a...
-
The Trapani Company had the following actual data for 2006 and 2007: The basic production data at standard unit costs for the two years were Fixed factory overhead was budgeted at $98,000 per year....
-
The Carley Company differs from the Marple Company (described in Problem 4-21) in only one respect: it has both variable and fixed manufacturing costs. Its variable costs are $0.14 per litre and its...

Study smarter with the SolutionInn App


