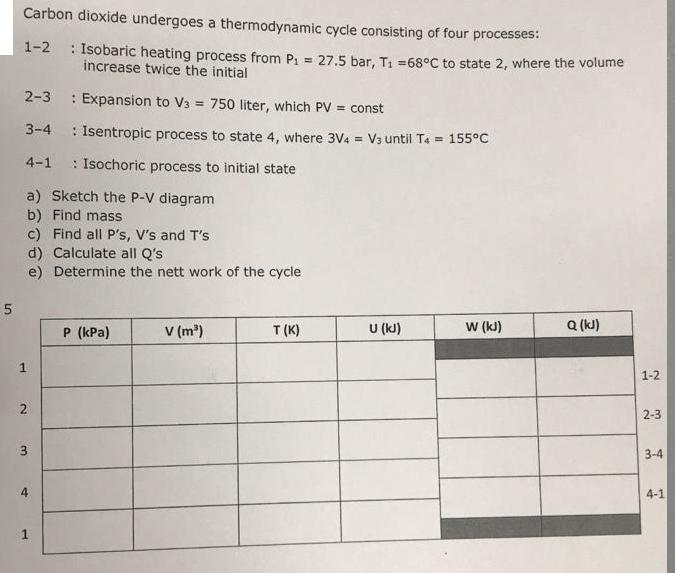
5 Carbon dioxide undergoes a thermodynamic cycle consisting of four processes: 1-2: Isobaric heating process from...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
5 Carbon dioxide undergoes a thermodynamic cycle consisting of four processes: 1-2: Isobaric heating process from P₁ = 27.5 bar, T₁ = 68°C to state 2, where the volume increase twice the initial 2-3 Expansion to V3 = 750 liter, which PV = const 3-4 : Isentropic process to state 4, where 3V4 = V3 until T4 = 155°C 4-1 : Isochoric process to initial state a) Sketch the P-V diagram b) Find mass c) Find all P's, V's and T's d) Calculate all Q's e) Determine the nett work of the cycle 1 2 3 st 4 1 P (kPa) V (m³) T (K) U (kJ) W (kJ) Q (kJ) 1-2 2-3 3-4 4-1 5 Carbon dioxide undergoes a thermodynamic cycle consisting of four processes: 1-2: Isobaric heating process from P₁ = 27.5 bar, T₁ = 68°C to state 2, where the volume increase twice the initial 2-3 Expansion to V3 = 750 liter, which PV = const 3-4 : Isentropic process to state 4, where 3V4 = V3 until T4 = 155°C 4-1 : Isochoric process to initial state a) Sketch the P-V diagram b) Find mass c) Find all P's, V's and T's d) Calculate all Q's e) Determine the nett work of the cycle 1 2 3 st 4 1 P (kPa) V (m³) T (K) U (kJ) W (kJ) Q (kJ) 1-2 2-3 3-4 4-1
Expert Answer:

Related Book For 

Accounting Tools for business decision making
ISBN: 978-0470095461
4th Edition
Authors: kimmel, weygandt, kieso
Posted Date:
Students also viewed these accounting questions
-
Member AB is supported by cable BC and at A by a square rod which fits loosely through the square hole at the end joint of the member as shown. Determine the components of reaction at A and the...
-
An ideal gas characterised by the coefficient c found in relation (5.62) and the coefficient = (c + 1) /c undergoes a refrigeration cycle consisting of four reversible processes (Fig. 7.24): 1 2 :...
-
A gas undergoes a thermodynamic cycle consisting of three processes: Process 12: constant volume, V = 0.028 m3, U2 = U1 =26.4 kJ Process 23: expansion with pV = constant, U3 = U2 Process 31: constant...
-
Listed below are measured amounts of caffeine (mg per 12 oz of drink) obtained in one can from each of 20 brands (7-UP, A&W Root Beer, Cherry Coke, . . . , Tab). Are the statistics representative of...
-
Black Gold Tea Exploration Co. was established on March 15, 2010, to provide oil-drilling services. Black Gold Tea uses field equipment (rigs and pipe) and field supplies (drill bits and lubricants)...
-
Summarize the practical contingency management implications for group size.
-
What stakeholder affected by their behavior was Gilead weighting very lightly when it decided what to tell the FDA about the medicines it wished to have permission to sell?
-
The records of Ellens Boutique report the following data for the month of April. Instructions Compute the ending inventory by the conventional retail inventorymethod. $99,000 Purchases (at cost)...
-
Defining the database management system and identifying the differences between a database and a database management system (DBMS) or relational database management system (RDBMS). Please provide the...
-
On January 1, 2024, Marigold Company purchased 8,568 shares of Swifty Company's common stock for $123,000. Immediately after the stock acquisition, the statements of financial position of Marigold...
-
A 43-year-old woman is admitted to a hospital because of deep abdominal pain in her epigastric region. On examination, it is observed that a retroperitoneal infection erodes an artery that runs along...
-
A stock price is currently $10. It is known that at the end of three months it will be either $11 or $8.5. The risk free interest rate is 5% per annum with continuous compounding. Suppose ST is the...
-
Which is the correct fault tree for the following RBD? a C b d e
-
How does the manipulation of spatial and temporal imagery contribute to the narrative structure and thematic development of literary works, shaping readers' perceptions of setting, atmosphere, and...
-
When does the amplitude of an oscillating system become maximum? Explain.
-
How do reception and reader-response theories investigate the role of the reader in the interpretation and meaning-making of literary texts, considering factors such as reception history, cultural...
-
on May 20 of the current year, Tracey sells a building to Judy for 540,000. Traceys basis in the building is 290,000. This area has a real property tax year that ends on June 30. The taxes are...
-
Access the Federation of Tax Administrators Internet site at www. taxadmin.org/state-tax-forms and indicate the titles of the following state tax forms and publications: a. Minnesota Form M-100 b....
-
Rosenthal Company manufactures bowling balls through two processes: Molding and Packaging. In the Molding Department, the urethane, rubber, plastics, and other materials are molded into bowling...
-
Oklahoma Manufacturing Company uses a standard cost accounting system. In 2012, the company produced 28,000 units. Each unit took several pounds of direct materials and 112 standard hours of direct...
-
Presented on the below are the comparative balance sheets for Lybeck Company at December 31. Additional information:1. Operating expenses include depreciation expense $65,000 and charges from prepaid...
-
A certain radar has range resolution of \(300 \mathrm{~m}\) and is observing a target somewhere in a line of high towers each having RCS \(\sigma_{\text {tower }}=10^{6} \mathrm{~m}^{2}\). If the...
-
An implementation of a double delay line canceler with feedback is shown below. (a) What is the transfer function, \(H(z)\) ? (b) Plot the frequency response for \(K_{1}=0=K_{2}, K_{1}=0.2\), and...
-
Plot the frequency response for the filter described in the previous problem for \(K=-0.5,0\), and 0.5. Data From Problem 2 One implementation of a single delay line canceler with feedback is shown...

Study smarter with the SolutionInn App


