Show all steps when solving. A pump uses 2010 3 ftlb (f)/s in order to move...
Fantastic news! We've Found the answer you've been seeking!
Question:
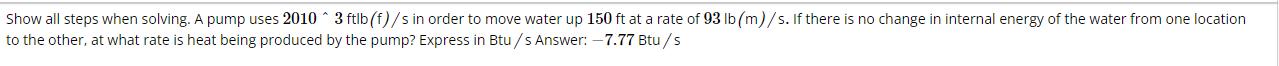
Transcribed Image Text:
Show all steps when solving. A pump uses 2010 3 ftlb (f)/s in order to move water up 150 ft at a rate of 93 lb (m)/s. If there is no change in internal energy of the water from one location to the other, at what rate is heat being produced by the pump? Express in Btu/s Answer: -7.77 Btu/s Show all steps when solving. A pump uses 2010 3 ftlb (f)/s in order to move water up 150 ft at a rate of 93 lb (m)/s. If there is no change in internal energy of the water from one location to the other, at what rate is heat being produced by the pump? Express in Btu/s Answer: -7.77 Btu/s
Expert Answer:
Answer rating: 100% (QA)
To solve this problem we can use the formula for power Power Force Veloc... View the full answer

Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Sunita inherits $45,000 from a distant relative. She decides to invest her inheritance in a diversified, dividend-paying mutual fund. At the time of her initial investment, she purchases 1,500 shares...
-
Suppose that Treasury bills are currently paying 9 percent and the expected inflation is 3 percent. What is the real interest rate?
-
Modify Figure 8-7 to find the concentrations of species in 0.05 M NH3. The only change required is the value of F. How do the pH and fraction of ammonia hydrolysis (= [NH + 4 ]/([NH + 4 ] + [NH 3 ]))...
-
Repeat the calculations of Example 6.3, but for \(80 \mathrm{~mol} \%\) of the liquid distilled. Data From Example 6.3:- Suppose the liquid of Example 6.1 [50 mol% n-heptane (A), 50 mol% n-octane...
-
Ling Company reports the following information for the year ended December 31, 2014: sales revenue $1,000,000, cost of goods sold $700,000, operating expenses $200,000, and an unrealized gain on...
-
Provide a clear definition of SES (Socioeconomic Status), encompassing its components such as income, education, and occupation. Discuss the societal implications of SES and its role in shaping...
-
Overview The milestone for Project One involves applying accounting principles and methods to long-term liabilities and equity. You will also evaluate these financial statement components for...
-
Prove that T= constant X p-I", Cv = 3/2(R) and Cp = 5/2(R) %3D
-
Cullumber Corporation made credit sales of $25,800 which are subject to 7% sales tax. The corporation also made cash sales which totaled $27,071 including the 7% sales tax. (a) Your answer is...
-
Campbell, a single taxpayer, earns $410,000 in taxable income and $2,800 in interest from an investment in State of New York bonds. (Use the U.S. tax rate schedule). Required: If Campbell earns an...
-
The capital expenditures budget includes all but which one of these items? a. Raw materials b. Machinery O c. Land d. Buildings
-
Give rationale of why change management is so difficult for most people Also explain why decision-making is difficult for some people give detailed responses preferably several paragraphs
-
For problems 1 & 2 list the first 4 terms of the sequence. An 1. 2. (-1)+1 00 For problems 3 & 4 determine if the given sequence converges or diverges. If it converges what is its limit? 3. n-7n+3...
-
In a process occurring in a closed system F, the heat transferred from F to the surroundings E is 600 J. If the temperature of E is 300 K and that of F is in the the entropy changes of the...
-
The manager for retail customers, Katie White, wants to hear your opinion regarding one business offer she has received from an entrepreneur who is starting a mobile phone app called Easy Money. The...
-
Fellowes and Associates Chartered Accountants is a successful mid-tier accounting firm with a large range of clients across Canada. In 2011, Fellowes and Associates gained a new client, Health Care...
-
Fellowes and Associates Chartered Accountants is a successful mid-tier accounting firm with a large range of clients across Canada. During the 2011 financial year, Fellowes and Associates gained a...
-
Required You have been working with a national chartered accounting firm for a number of years and you now feel like you are ready to start your own chartered accounting firm. Given the requirements...
-
There is another possible explanation for purchased goodwill appearing in a sole proprietor's statement of financial position. What do you think it might be?
-
Why do the assets need to be revalued in these cases? The business has not been sold.
-
The shown partners have always shared profits and losses in the ratio: Holt 4; Stott 2: Young 1. From 1 January the assets were to be revalued as the profit sharing ratios are to be altered soon. The...

Study smarter with the SolutionInn App


