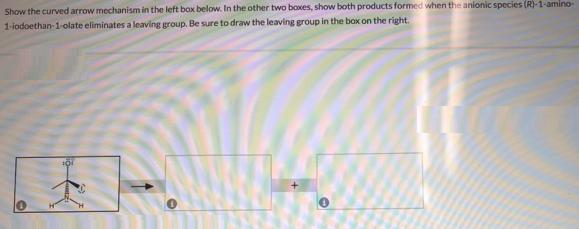
Show the curved arrow mechanism in the left box below. In the other two boxes, show...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Show the curved arrow mechanism in the left box below. In the other two boxes, show both products formed when the anionic species (R)-1-amino- 1-iodoethan-1-olate eliminates a leaving group. Be sure to draw the leaving group in the box on the right. Show the curved arrow mechanism in the left box below. In the other two boxes, show both products formed when the anionic species (R)-1-amino- 1-iodoethan-1-olate eliminates a leaving group. Be sure to draw the leaving group in the box on the right.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Draw curved arrows to show the flow of electrons responsible for the conversion of reactants into products. a. b. c. :O CH-C OCH
-
Two products are formed when methylenecyclohexane reacts with NBS. Explain how each is formed. CH2 CH Br CH2 NBS, peroxide
-
Draw both resonance structures of the enolate formed when each of the following ketones is treated with a strong base: (a) (b) (c) (d) (e)
-
HL Co. uses the high-low method to derive a total cost formula. Using a range of units produced from 1,500 to 7,500, and a range of total costs from $21,000 to $45,000, producing 2,000 units will...
-
A production process operates with 1% nonconforming output. Every hour a sample of 25 units of product is taken, and the number of nonconforming units counted. If one or more nonconforming units are...
-
A metal cylinder with rigid walls contains 2.50 mol of oxygen gas. The gas is cooled until the pressure decreases to 30.0% of its original value. You can ignore the thermal contraction of the...
-
Compute the \(F\) value for a comparison among the three levels representing the average amount of time that swimmers practice weekly ( \( <15\) hours, 15-25 hours, and \(>25\) hours), with the...
-
Goode Company has the following production data for selected months. Compute equivalent units of production for materials and conversion costs, assuming materials are entered at the beginning of...
-
Glucose, during transfusion, flows from a bag through a needle that is 3.0cm long and has an inside diameter of 0.50 mm. If the transfusion rate is to be 0.50 l in 40 min, how high above the needle...
-
You have met with your clients a few times and are now ready to do their retirement needs analysis (RNA). The couple, filing Married Filing Jointly (MFJ), are both 39 years of age and expect to work...
-
A hiring manager has a vacancy on his team. He is interested in backfilling the role as soon as possible. He often says things like he wants "someone younger on the team to balance out the older...
-
In the Challenge Solution, show that shareholders' expected earnings are higher with the new compensation scheme than with the original one.
-
So far, we've researched the viewpoints of leaders in the field of information presentation and human interface design for computer systems. What about experts in other areas? a. Find and interview...
-
Suppose that a market for toothpaste has two sellers, each of which says its product has the best combination of ingredients to protect against tooth decay and to control plaque, tartar, and gum...
-
Consider the two toothpaste producers in Question 5.4. If instead of undertaking an advertising campaign, Firm 1 convinced Firm 2 to form a cartel, would it have been better off? Assume that both...
-
Malaysia's monopoly auto manufacturer produces the Proton, which is protected from imports by a specific tariff, \(t\), on imported goods. The monopoly's profit-maximizing price is \(p^{*}\). The...
-
In the second half of this course you will work in pairs to prepare B2B Sales Presentation. After you have selected a partner to work with, the first step is to choose a product for your...
-
In your readings, there were many examples given for nurturing close family relationships in this ever-evolving technological society we live in Based upon your readings and research describe three...
-
Determine the partial negative charge on the oxygen atom in a C=O bond. The bond length is 1.22 * and the bond dipole moment is 2.30 D.
-
Bupropion hydrochloride is an antidepressant marketed under the trade name Wellbutrin®. Propose a synthesis of bupropion hydrochloride, starting with benzene. Cl CCHCH3 NH2C(CH3)3CI bupropion...
-
The addition of HBr to which of the following alkenes is more highly regioselective? a. b. CH CH CH,CHC=CH, CH 3C CHCH or - CH CH3 or
-
(a) Describe the effect of temperature on the equilibrium constant. (b) Industrial \(\mathrm{CH}_{3} \mathrm{OH}\) is prepared according to the reaction \[ \mathrm{CO}(\mathrm{g})+2...
-
Under atmospheric conditions the acetone-chloroform azeotrope boils at \(64.6^{\circ} \mathrm{C}\) and contains 33.5 mole per cent acetone. The vapour pressures of acetone and chloroform at this...
-
Explain in detail, with neat sketch, the working principle of a fuel cell in producing electrical energy from the chemical energy of a fuel.

Study smarter with the SolutionInn App



